Chapter10 Temperature and Heat Chapter10 Temperature and Heat














- Slides: 28

Chapter-10 Temperature and Heat

Chapter-10 Temperature and Heat 1 Temperature and its Measurement 2 Heat and Specific Heat Capacity 5 The Flow of Heat Everyday Phenomenon: Solar Collectors and the Greenhouse Effect

Temperature

Mercury Clinical Thermometer

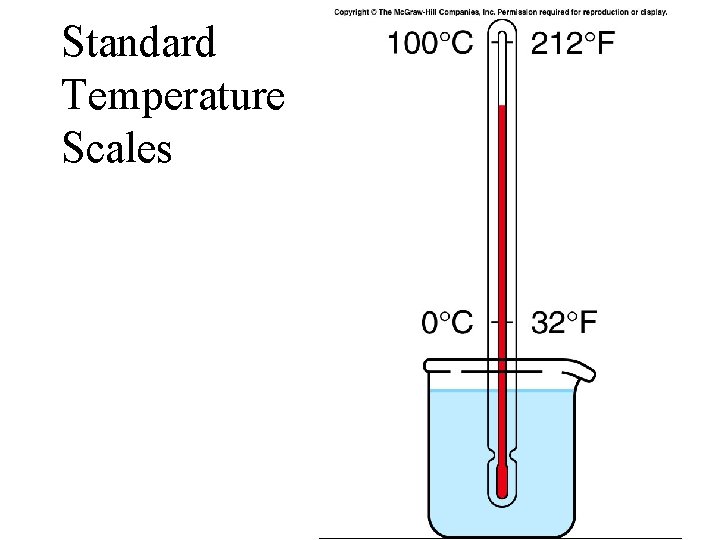
Standard Temperature Scales

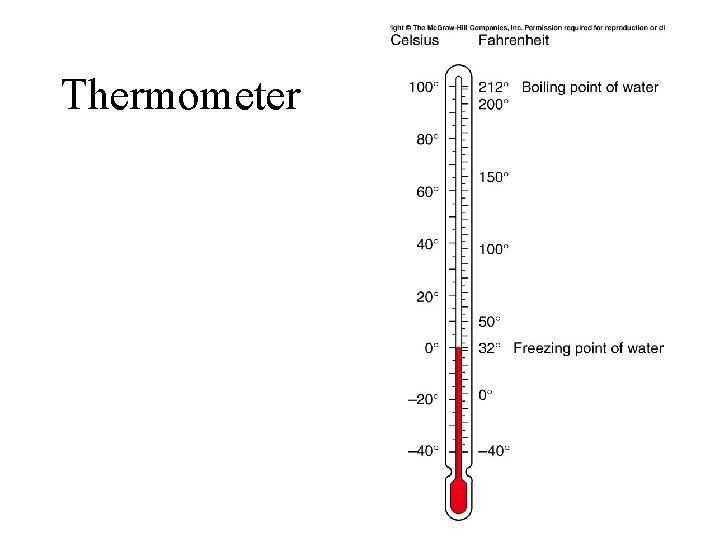
Thermometer

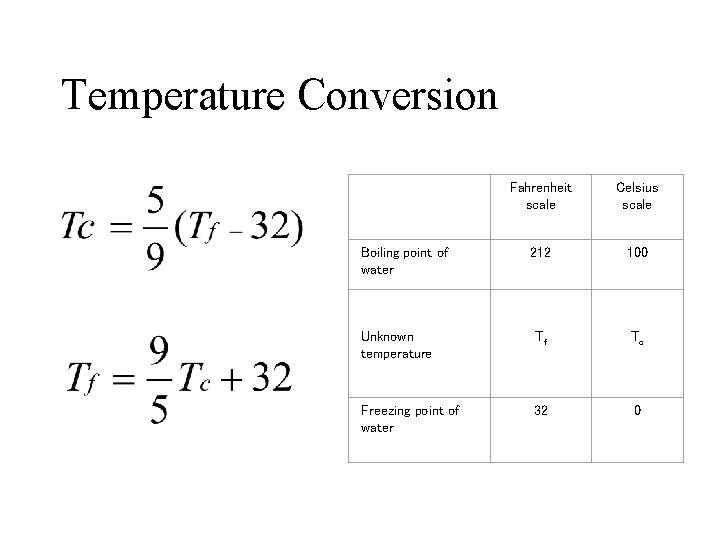
Temperature Conversion Fahrenheit scale Celsius scale 212 100 Unknown temperature Tf Tc Freezing point of water 32 0 Boiling point of water

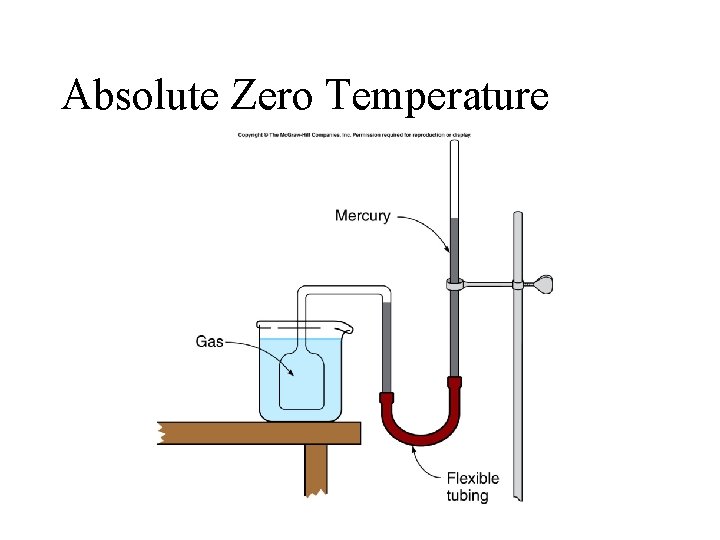
Absolute Zero Temperature

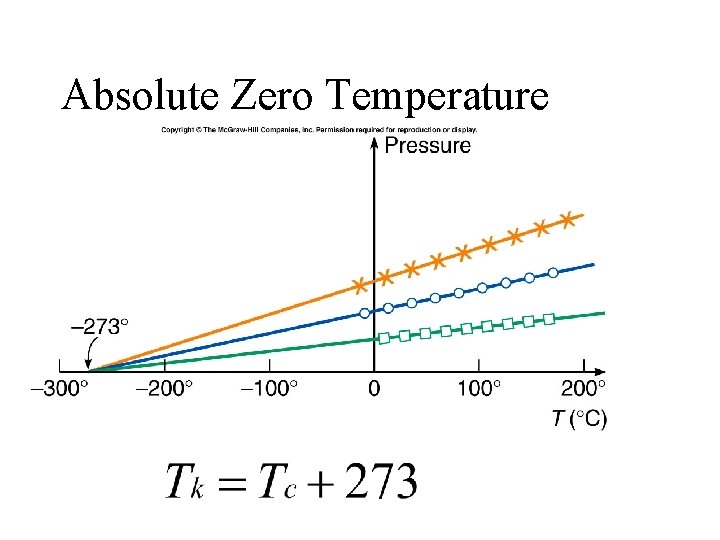
Absolute Zero Temperature

E 4

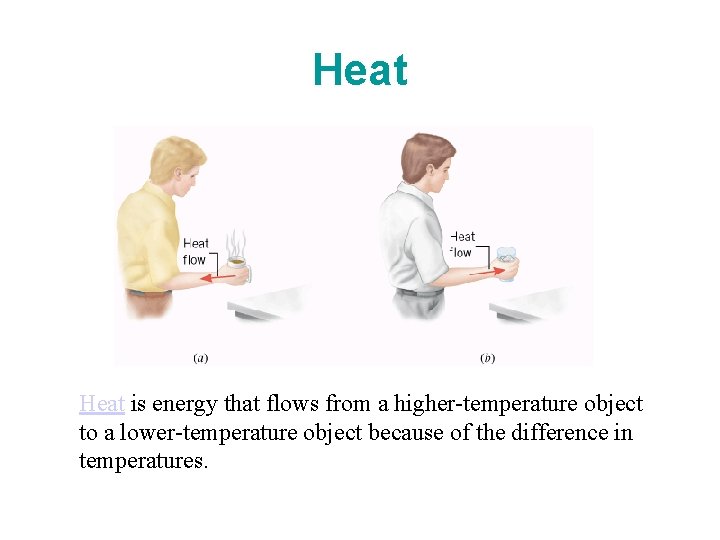
Heat is energy that flows from a higher-temperature object to a lower-temperature object because of the difference in temperatures.

Units for Heat SI unit for heat is the joule, J. Calorie is another unit for heat. It comes with a lower case and an upper case. Nutritionists use the word “Calorie, ” with a capital C, to specify the energy content of foods. For example, a regular 12 -oz can of soda has about 140 Calories. The cgs unit of heat is the calorie, with a lower case. One calorie (1 cal) is defined as the amount of heat needed to raise the temperature of one gram of water by one Celsius degree. 1 food Calorie = 1000 calories = 1 kcal 1 calorie = 4. 186 J.

Specific Heat Capacity Specific heat capacity of a material is the quantity of heat needed to change a unit mass of the material by a unit change in temperature. It is a property of the material.

Heat Q The heat Q that must be supplied or removed to change the temperature of a substance of mass m by an amount DT is, where c is the specific heat capacity of the substance. Unit for Specific Heat Capacity: SI: J/(kg · C°) cgs: cal/(g. C°)

Calorimetry

CP 4

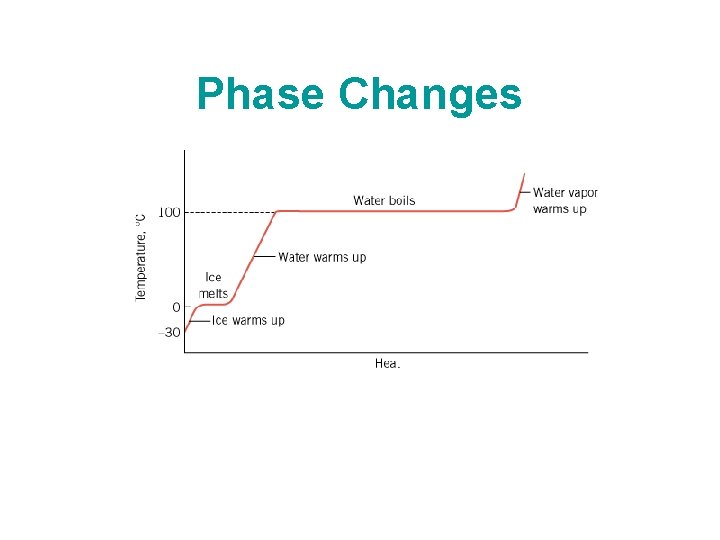
Phase Changes

Phase Changes

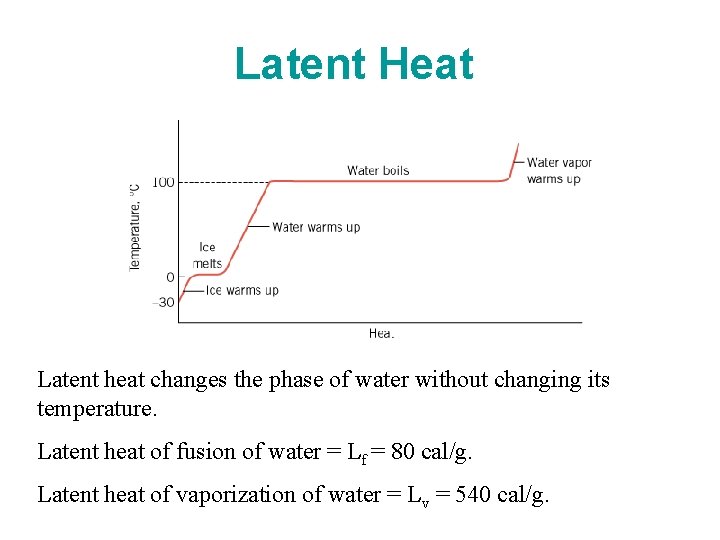
Latent Heat Latent heat changes the phase of water without changing its temperature. Latent heat of fusion of water = Lf = 80 cal/g. Latent heat of vaporization of water = Lv = 540 cal/g.

E 8

10. 5 The Flow of Heat can flow via conduction, convection, and radiation.

Conduction When a metal block and a wooden block, both at room temperature, are picked up, the metal block feels cooler, due to conduction of heat. Conduction is the process whereby heat is transferred directly through a material.

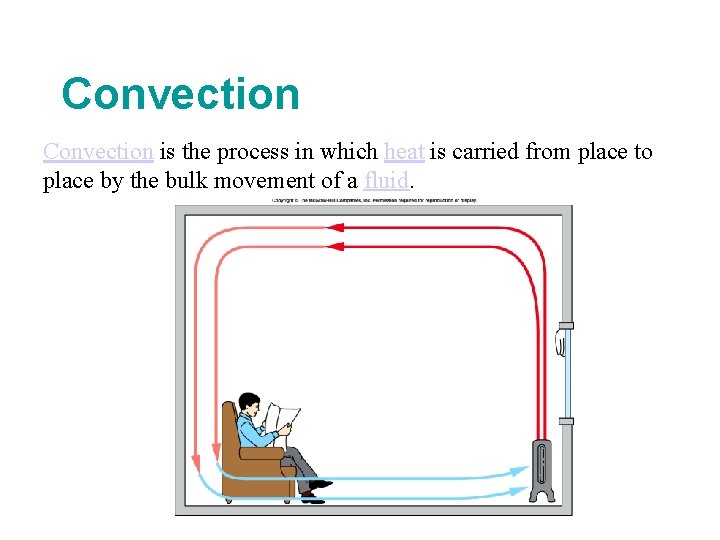
Convection is the process in which heat is carried from place to place by the bulk movement of a fluid.

Radiation is the process in which energy is transferred by means of electromagnetic waves. Heat transfer by radiation can take place through vacuum. This is because electromagnetic waves are involved in radiation and they can propagate through empty space.

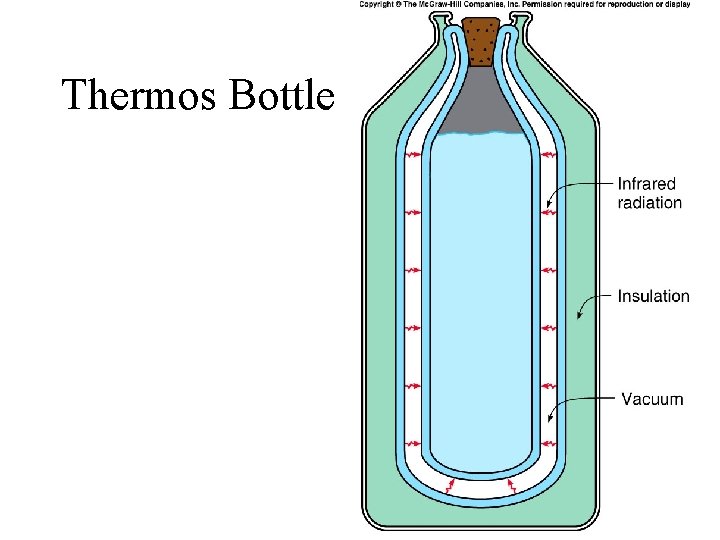
Thermos Bottle
Q 28

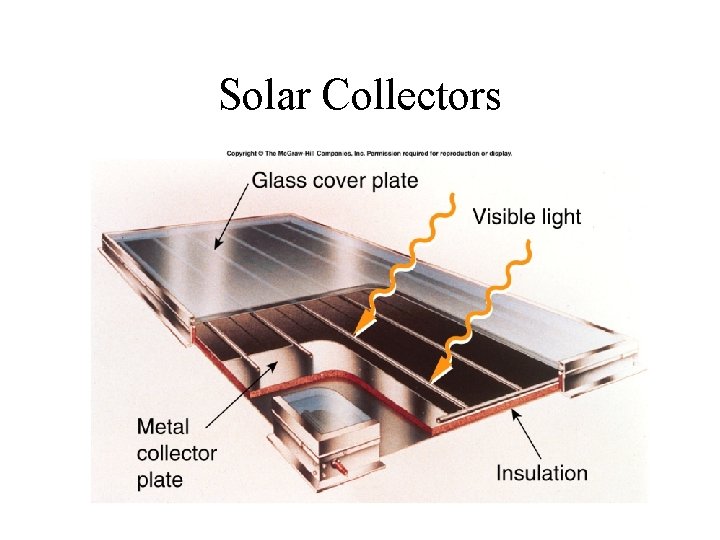
Solar Collectors

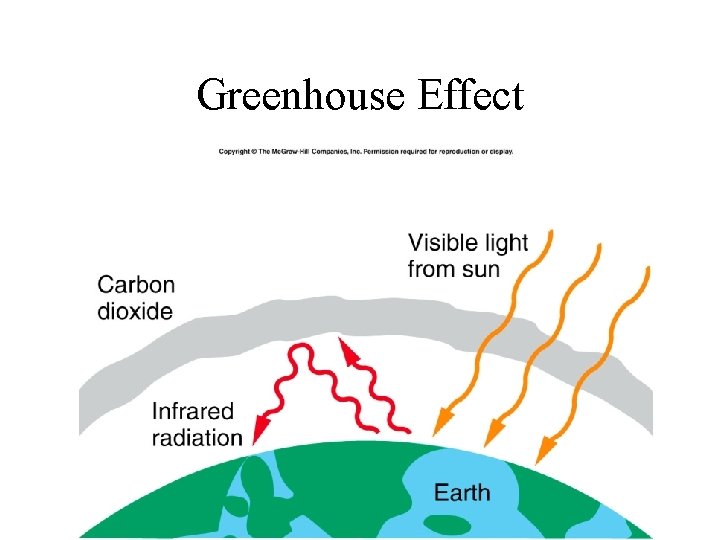
Greenhouse Effect