Fisiopatologia diagnosi e classificazione della mielofibrosi Barosi Giovanni

Fisiopatologia, diagnosi e classificazione della mielofibrosi Barosi Giovanni Laboratorio di Epidemiologia Clinica e Centro per lo Studio della Mielofibrosi. Fondazione IRCCS Policlinico S. Matteo, Pavia Catania, 7 Novembre 2008

Key Questions in Primary Myelofibrosis Biological key questions 1. What genetic event preceding or concomitant to JAK 2 V 617 F mutation contributes to the development of PMF? 2. Mechanism for constitutive mobilization of CD 34+ cells in PMF 3. Mechanism for constitutive mobilization of endothelial progenitor cells and angiogenesis in PMF 4. Role of TGF beta in myelofibrosis and myeloproliferation of PMF Operational (Clinical) key questions 1. JAK 2 mutation load and prognosis 2. Definition of resistance to HU in PMF Therapeutic key questions 1. Efficacy of molecularly targeted therapies

Toward a model of malignancy for primary myelofibrosis
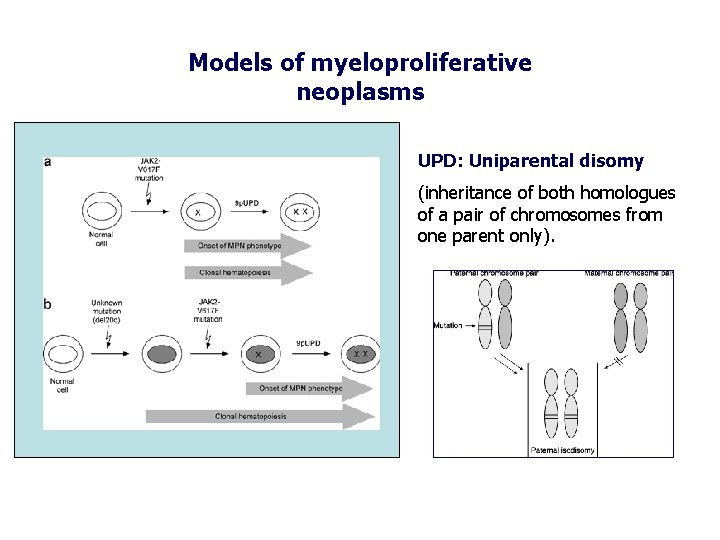
Models of myeloproliferative neoplasms UPD: Uniparental disomy (inheritance of both homologues of a pair of chromosomes from one parent only).

Evidence that a preceding genetic event is necessary for developing the MPN disease 1. In certain MPN patients, the clonality of expanded myeloid progenitors is found to be larger than JAK 2 V 617 F clone (JAK 2 V 617 F being a late genetic event). Kralovics, R. et al. Blood 2006; 108: 1377 -1380
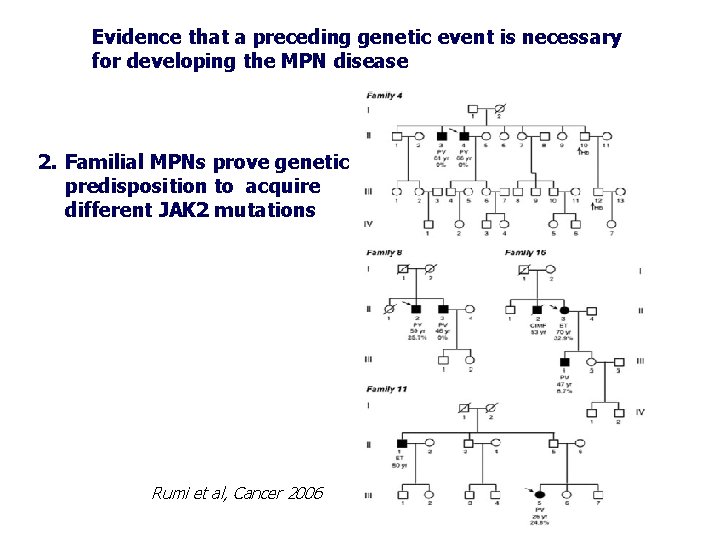
Evidence that a preceding genetic event is necessary for developing the MPN disease 2. Familial MPNs prove genetic predisposition to acquire different JAK 2 mutations Rumi et al, Cancer 2006
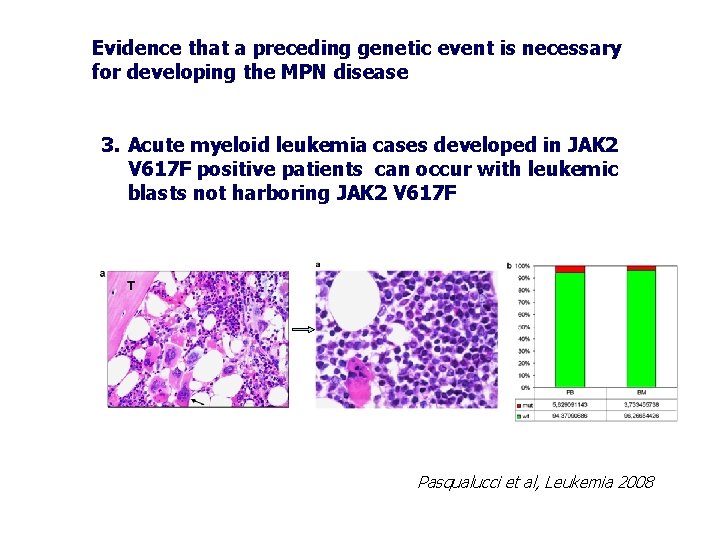
Evidence that a preceding genetic event is necessary for developing the MPN disease 3. Acute myeloid leukemia cases developed in JAK 2 V 617 F positive patients can occur with leukemic blasts not harboring JAK 2 V 617 F Pasqualucci et al, Leukemia 2008
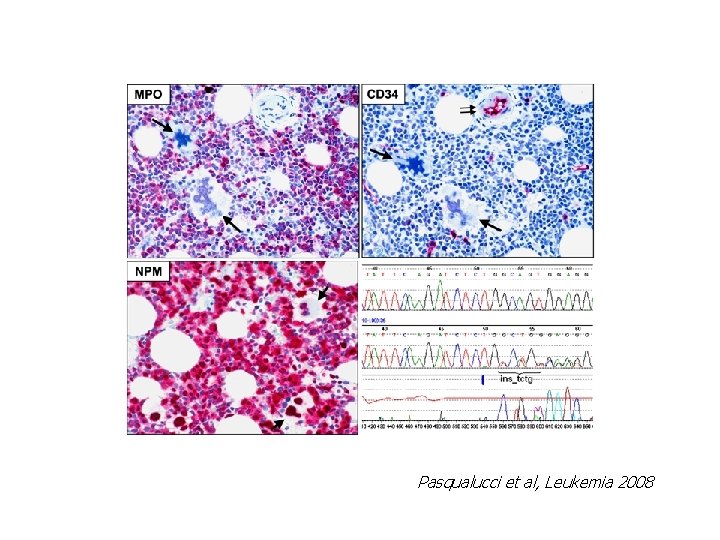
Pasqualucci et al, Leukemia 2008

Other JAK 2 -independent molecular events influence the phenotype of PMF Epigenetic alterations • SOCS • CXCR 4 mi. RNA
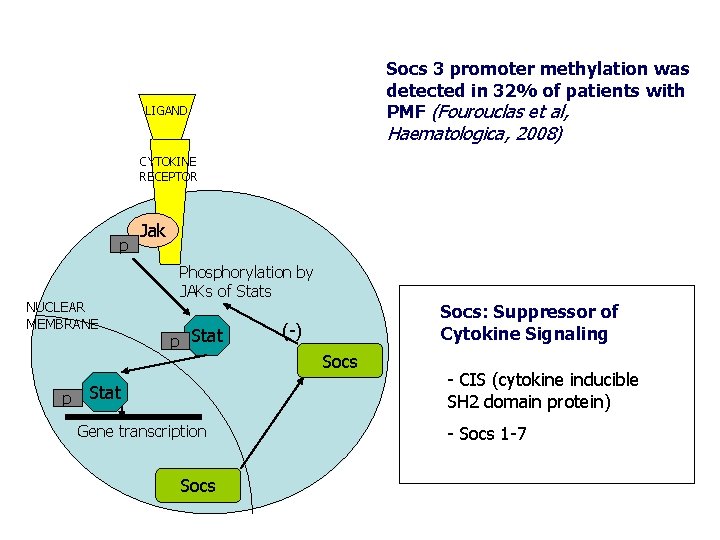
Socs 3 promoter methylation was detected in 32% of patients with PMF (Fourouclas et al, LIGAND Haematologica, 2008) CYTOKINE RECEPTOR p NUCLEAR MEMBRANE Jak Phosphorylation by JAKs of Stats p Stat Gene transcription Socs: Suppressor of Cytokine Signaling (-) Socs - CIS (cytokine inducible SH 2 domain protein) - Socs 1 -7
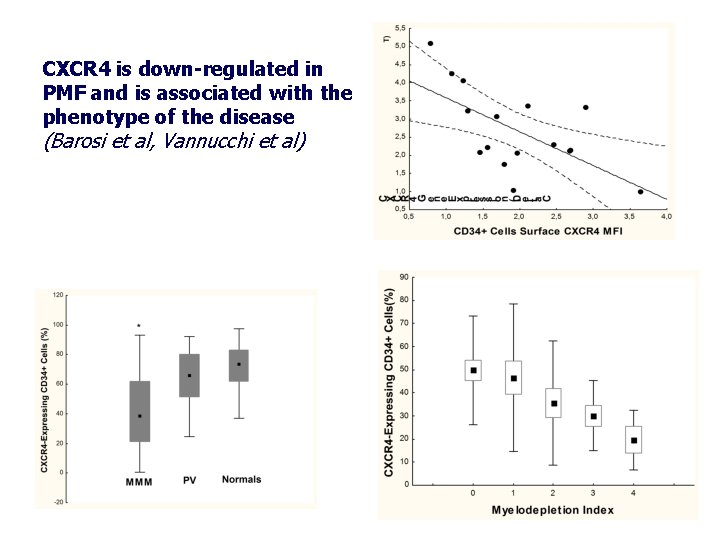
CXCR 4 is down-regulated in PMF and is associated with the phenotype of the disease (Barosi et al, Vannucchi et al)
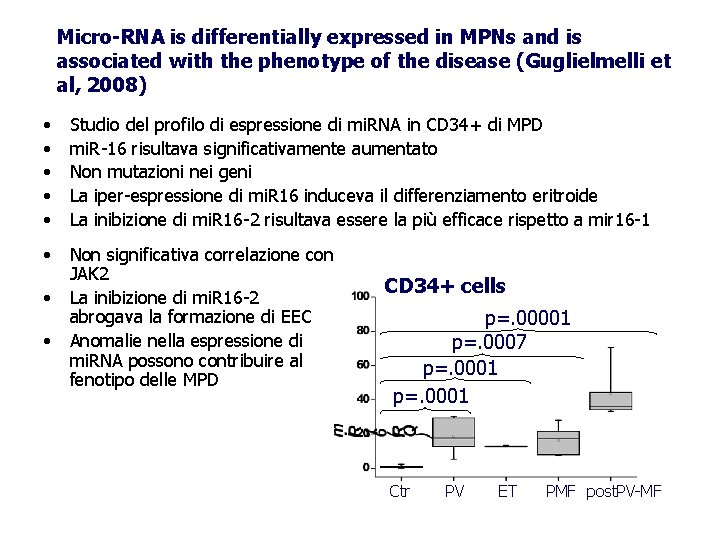
Micro-RNA is differentially expressed in MPNs and is associated with the phenotype of the disease (Guglielmelli et al, 2008) • • • Studio del profilo di espressione di mi. RNA in CD 34+ di MPD mi. R-16 risultava significativamente aumentato Non mutazioni nei geni La iper-espressione di mi. R 16 induceva il differenziamento eritroide La inibizione di mi. R 16 -2 risultava essere la più efficace rispetto a mir 16 -1 • Non significativa correlazione con JAK 2 La inibizione di mi. R 16 -2 abrogava la formazione di EEC Anomalie nella espressione di mi. RNA possono contribuire al fenotipo delle MPD • • CD 34+ cells p=. 00001 p=. 0007 p=. 0001 Ctr PV ET PMF post. PV-MF

Endothelial progenitor cells
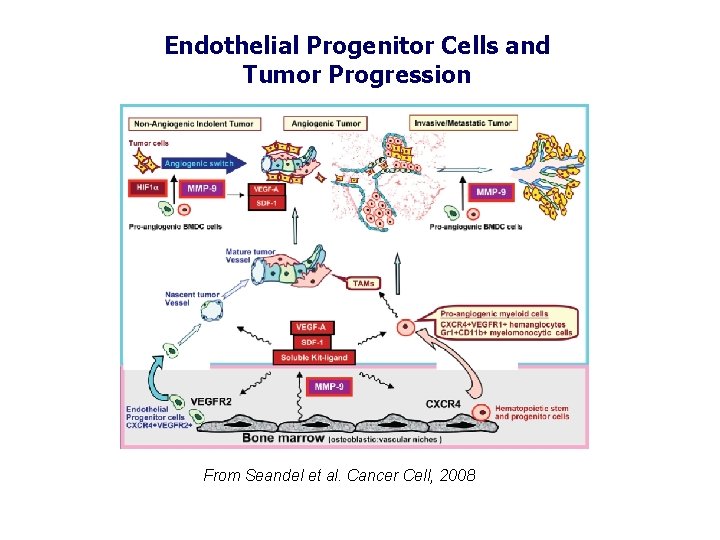
Endothelial Progenitor Cells and Tumor Progression From Seandel et al. Cancer Cell, 2008
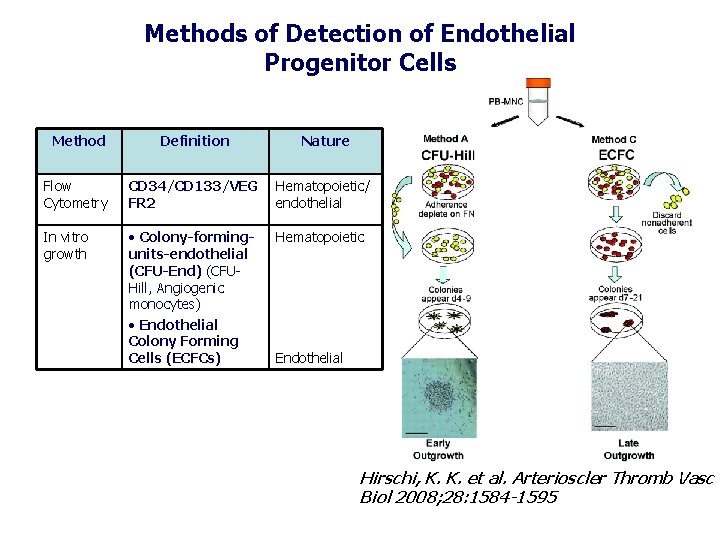
Methods of Detection of Endothelial Progenitor Cells Method Definition Nature Flow Cytometry CD 34/CD 133/VEG FR 2 Hematopoietic/ endothelial In vitro growth • Colony-formingunits-endothelial (CFU-End) (CFUHill, Angiogenic monocytes) • Endothelial Colony Forming Cells (ECFCs) Hematopoietic Endothelial Hirschi, K. K. et al. Arterioscler Thromb Vasc Biol 2008; 28: 1584 -1595
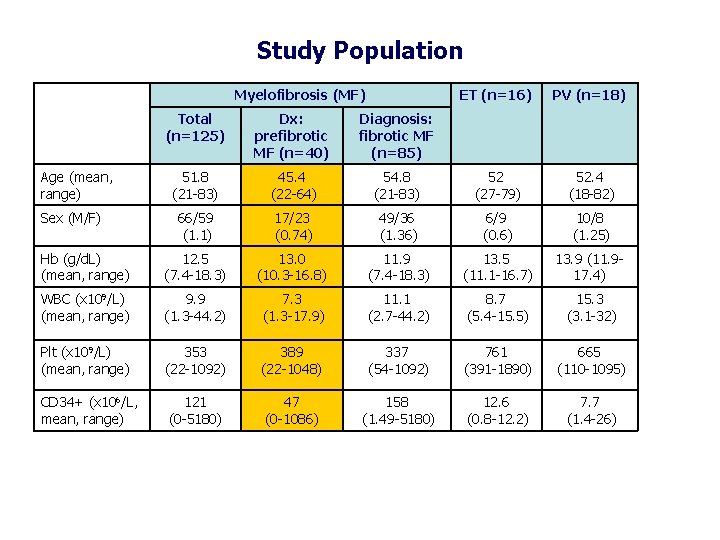
Study Population Myelofibrosis (MF) ET (n=16) PV (n=18) Total (n=125) Dx: prefibrotic MF (n=40) Diagnosis: fibrotic MF (n=85) 51. 8 (21 -83) 45. 4 (22 -64) 54. 8 (21 -83) 52 (27 -79) 52. 4 (18 -82) 66/59 (1. 1) 17/23 (0. 74) 49/36 (1. 36) 6/9 (0. 6) 10/8 (1. 25) Hb (g/d. L) (mean, range) 12. 5 (7. 4 -18. 3) 13. 0 (10. 3 -16. 8) 11. 9 (7. 4 -18. 3) 13. 5 (11. 1 -16. 7) 13. 9 (11. 917. 4) WBC (x 109/L) (mean, range) 9. 9 (1. 3 -44. 2) 7. 3 (1. 3 -17. 9) 11. 1 (2. 7 -44. 2) 8. 7 (5. 4 -15. 5) 15. 3 (3. 1 -32) Plt (x 109/L) (mean, range) 353 (22 -1092) 389 (22 -1048) 337 (54 -1092) 761 (391 -1890) 665 (110 -1095) CD 34+ (x 106/L, mean, range) 121 (0 -5180) 47 (0 -1086) 158 (1. 49 -5180) 12. 6 (0. 8 -12. 2) 7. 7 (1. 4 -26) Age (mean, range) Sex (M/F)
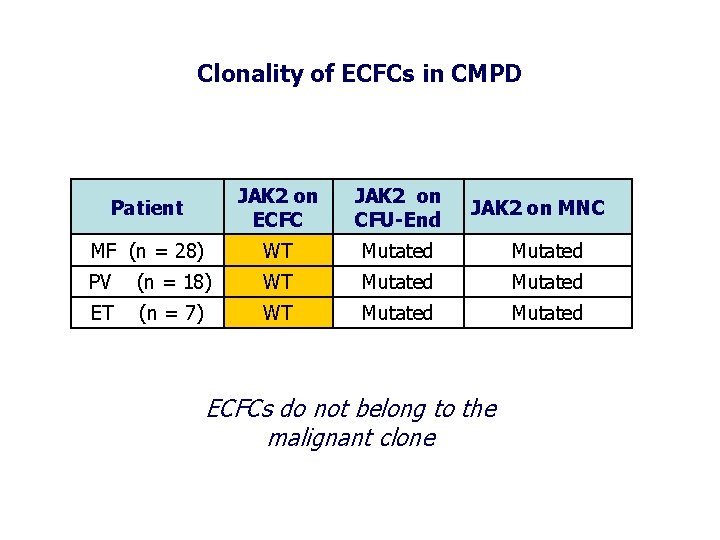
Clonality of ECFCs in CMPD Patient JAK 2 on ECFC JAK 2 on CFU-End MF (n = 28) WT Mutated PV (n = 18) WT Mutated ET (n = 7) WT Mutated JAK 2 on MNC ECFCs do not belong to the malignant clone

ECFCS in Myelofibrosis ECFCs are not associated with: • Hemoglobin level • Disease duration • Spleen size • JAk 2 mutational status or JAK 2 allele burden
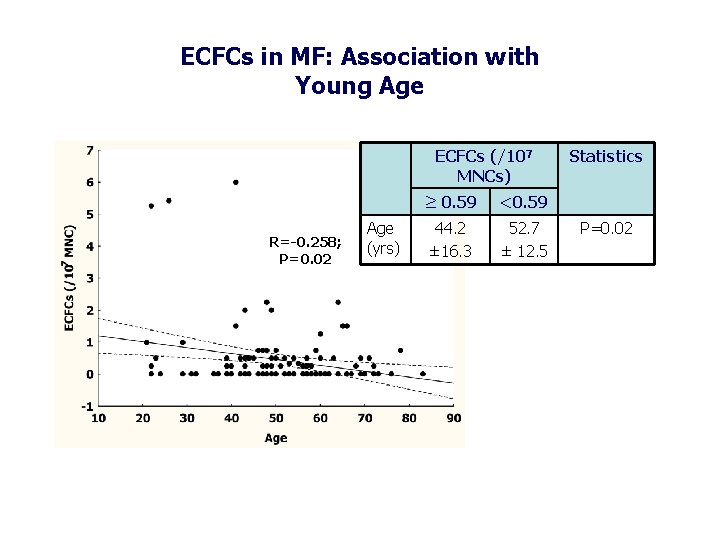
ECFCs in MF: Association with Young Age ECFCs (/107 MNCs) R=-0. 258; P=0. 02 Age (yrs) ≥ 0. 59 <0. 59 44. 2 ± 16. 3 52. 7 ± 12. 5 Statistics P=0. 02
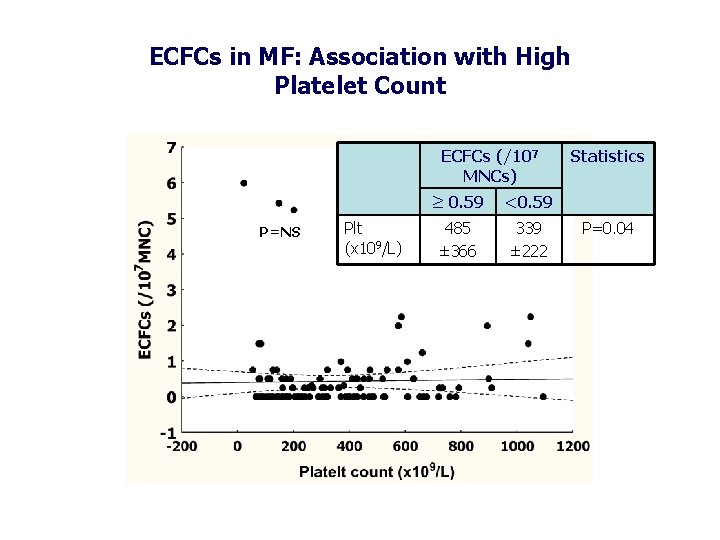
ECFCs in MF: Association with High Platelet Count ECFCs (/107 MNCs) P=NS Plt (x 109/L) ≥ 0. 59 <0. 59 485 ± 366 339 ± 222 Statistics P=0. 04
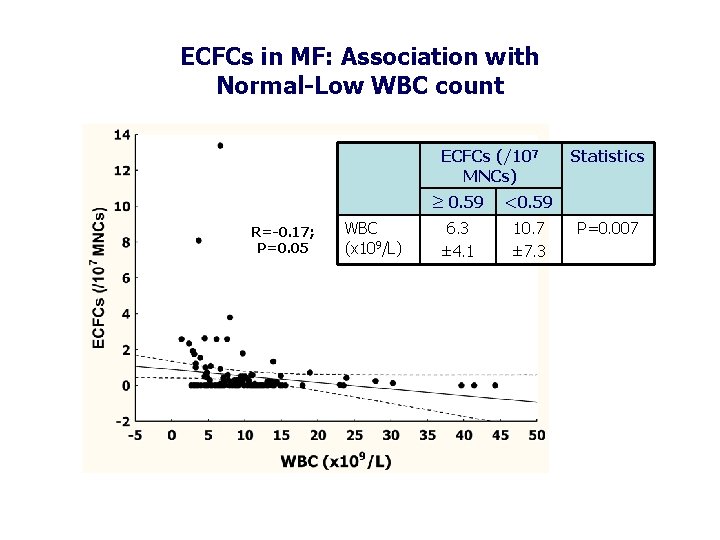
ECFCs in MF: Association with Normal-Low WBC count ECFCs (/107 MNCs) R=-0. 17; P=0. 05 WBC (x 109/L) ≥ 0. 59 <0. 59 6. 3 ± 4. 1 10. 7 ± 7. 3 Statistics P=0. 007
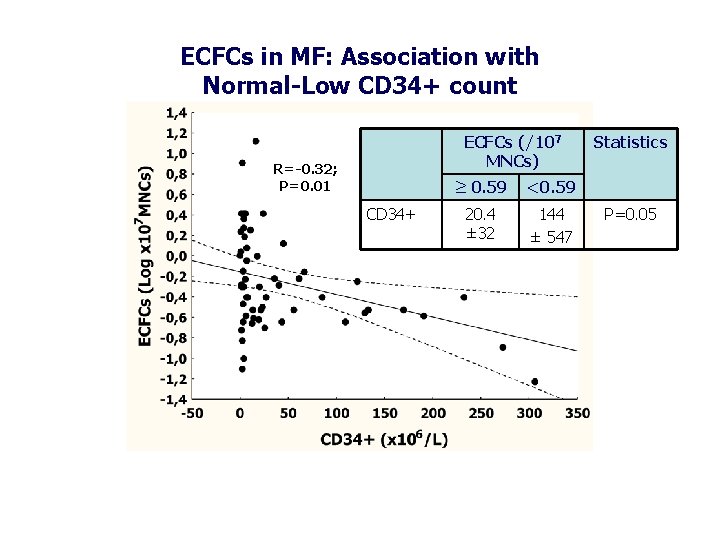
ECFCs in MF: Association with Normal-Low CD 34+ count ECFCs (/107 MNCs) R=-0. 32; P=0. 01 CD 34+ ≥ 0. 59 <0. 59 20. 4 ± 32 144 ± 547 Statistics P=0. 05
ECFCs in MF: Association with a Diagnosis of Prefibrotic Myelofibrosis MF (N= 125) Samples with at least one colony grown Frequency (/107 MNCs) (mean, range) Samples with a frequency higher than 0. 56/107 MNCs Statistics Total Dx: prefibrotic MF (n=40) Dx: fibrotic MF (n=85) 58 (46. 4%) 21 (52. 5%) 37 (43. 5%) NS 0. 52 1. 04 (0 -13. 38) 0. 28 (0 -3. 8) P=0. 007 22 (17. 6%) 12 (30%) 10 (11. 7%) P=0. 1

Conclusion • With the current assay method, ECFCs in peripheral blood are measurable in a limited number of normal subjects and patients with CMPDs • The highest frequency of ECFCs is measured in patients with Myelofibrosis • High frequency of ECFCs is associated with young age, high number of platelet count, normal or low number of WBC, normal CD 34+ cells and a diagnosis of prefibrotic myelofibrosis

Hypothesis: EPCs mark an early phase of the disease (pre-angiogenic and premetastatic phase) • No hemopoietic progenitor cells mobilization (no myeloid metaplasia) • EPCs respond to angiogenic stimuli (hypoxia? ) and migrate to extramedullary sites • Hemopoietic progenitor cells mobilization (myeloid metaplasia) Angiogenic switch • Intense angiogenesis

WHO (2008) criteria for primary myelofibrosis (PMF): diagnosis requires meeting all 3 major and 2 minor criteria Major criteria 1. Presence of magakaryocyte proliferation and atypia*, usually accompanied by either reticulin and/or collagen fibrosis, or in the absence of significant reticulin fibrosis, the megakaryocyte changes must be accompanied by an increased bone marrow cellularity characterized by granulocytic proliferation and often decreased erythropoiesis (i. e. prefibrotic cellular-phase disease) 2. Not meeting WHO criteria for polycythemia vera, BCR-ABL 1+ chronic myelogenous leukemia, myelodysplastic syndrome, or other myeloid neoplasms 3. Demonstration of JAK 2 V 6127 F or other clonal marker (e. g. MPL W 515 K/L), or in the absence of a clonal marker, no evidence that bone marrow fibrosis or other changes are secondary to infection, autoimmune disorder or other chronic inflammatory condition, hairy cell leukemia or other lymphoid neoplasm, metastatic malignancy, or toxic (chronic) myelopathies Minor criteria 1. Leukoerythroblastosis 2. Increased in serum lactate dehydrogenase level 3. Anemia 4. Splenomegaly ______________________________________ * Small to large megakaryocytes with an aberrant nuclear /cytoplasmic ratio and hyperchromatic, bulbous, or irregularly folded nuclei and dense clustering

The Italian Diagnostic Criteria of MMM NECESSARY CRITERIA Diffuse bone marrow fibrosis B. Absence of Philadelphia chromosome or BCR-ABL rearrangement in peripheral blood cells A. OPTIONAL CRITERIA 1. Splenomegaly of any grade 2. Anisopoikilocytosis with tear-drop erythrocytes 3. Presence of circulating immature myeloid cells 4. Presence of circulating erythroblasts 5. Presence of clusters of megakaryoblasts and anomalous megakaryocytes in bone marrow sections 6. Myeloid metaplasia DIAGNOSIS OF MMM: The two necessary criteria plus any other 2 optional criteria when splenomegaly is present, or any other 4 optional criteria when splenomegaly is absent Barosi et al. BJH 1999
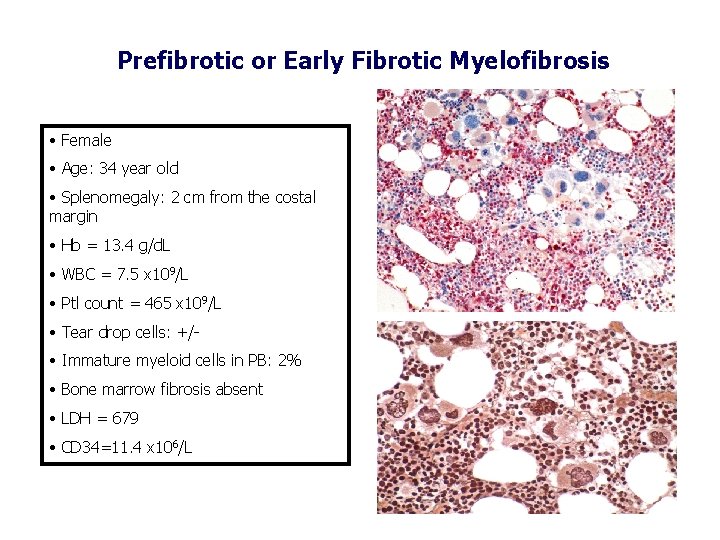
Prefibrotic or Early Fibrotic Myelofibrosis • Female • Age: 34 year old • Splenomegaly: 2 cm from the costal margin • Hb = 13. 4 g/d. L • WBC = 7. 5 x 109/L • Ptl count = 465 x 109/L • Tear drop cells: +/ • Immature myeloid cells in PB: 2% • Bone marrow fibrosis absent • LDH = 679 • CD 34=11. 4 x 106/L

New Therapeutic Targets in Myelofibrosis • Non-JAK 2 Inhibitors • JAK 2 Inhibitors Class I (JAK 2 selective) Class II (non JAK 2 selective)

Non-JAK 2 Inhibitors - Published Studies Molecular Target Name Company Design and phase DNA Methylation 5 Azacitidine Pharmion Reference 5 -day Mesa et al, Leukemia schedule/Phase II 2008 7 -day Quintas-Cardama et schedule/Phase II al, Leukemia 2008 Proteasome Bortezomib (Velcade) Millenium Phase II Mesa et al. Leukemia Farnesyltra nsferase Tipifarnib (Zarnestra) Johnson & Johnson Phase II Mesa et al, Leukemia VEGFR-2 PTK/ZK Novartis/ Shering Phase I Giles et al, Leukemia 2008 2007 Research 2007
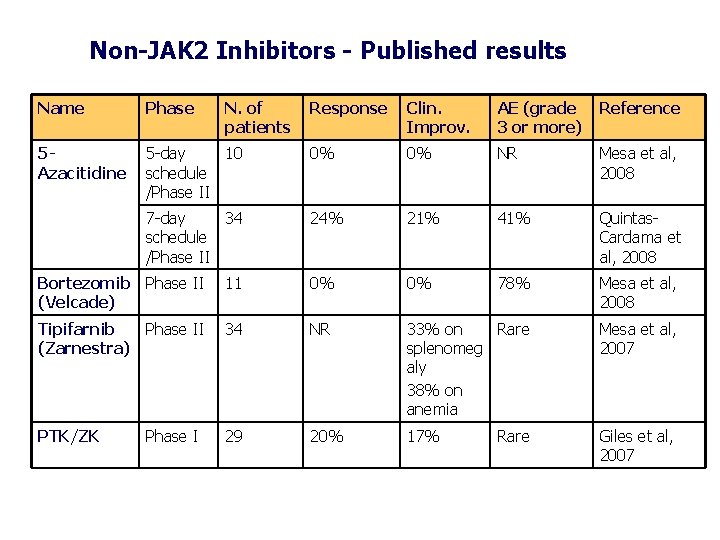
Non-JAK 2 Inhibitors - Published results Name Phase 5 Azacitidine N. of patients Response Clin. Improv. AE (grade 3 or more) Reference 5 -day 10 schedule /Phase II 0% 0% NR Mesa et al, 2008 7 -day 34 schedule /Phase II 24% 21% 41% Quintas. Cardama et al, 2008 Bortezomib Phase II (Velcade) 11 0% 0% 78% Mesa et al, 2008 Tipifarnib Phase II (Zarnestra) 34 NR 33% on Rare splenomeg aly 38% on anemia Mesa et al, 2007 PTK/ZK 29 20% 17% Giles et al, 2007 Phase I Rare

Lessons From Non-JAK 2 Inhibiting Molecular Targets in Myelofibrosis • Targeting non disease-specific molecular pathways results in null or moderate response • Preclinical analysis of therapeutic agents may not predict clinical acitivity -Bortezomib has activity in thrombopoietin-driven murine model of myelofibrosis (Wagner-Ballon, 2007), but no activity in humans. • Clinical activity is not correlated with the documented biological acitivty - No difference in hypomethylation with 5 -AZA between responders and non-responders (Quintas-Cardama, 2008)
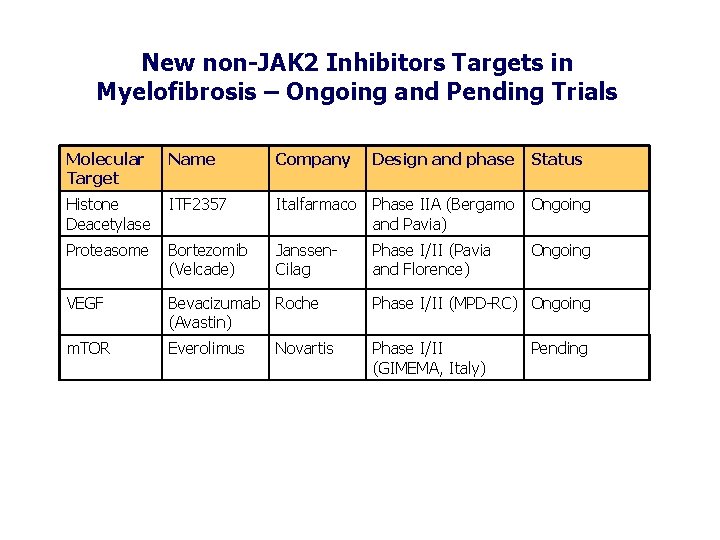
New non-JAK 2 Inhibitors Targets in Myelofibrosis – Ongoing and Pending Trials Molecular Target Name Company Histone Deacetylase ITF 2357 Italfarmaco Phase IIA (Bergamo and Pavia) Ongoing Proteasome Bortezomib (Velcade) Janssen. Cilag Ongoing VEGF Bevacizumab Roche (Avastin) Phase I/II (MPD-RC) Ongoing m. TOR Everolimus Phase I/II (GIMEMA, Italy) Novartis Design and phase Phase I/II (Pavia and Florence) Status Pending
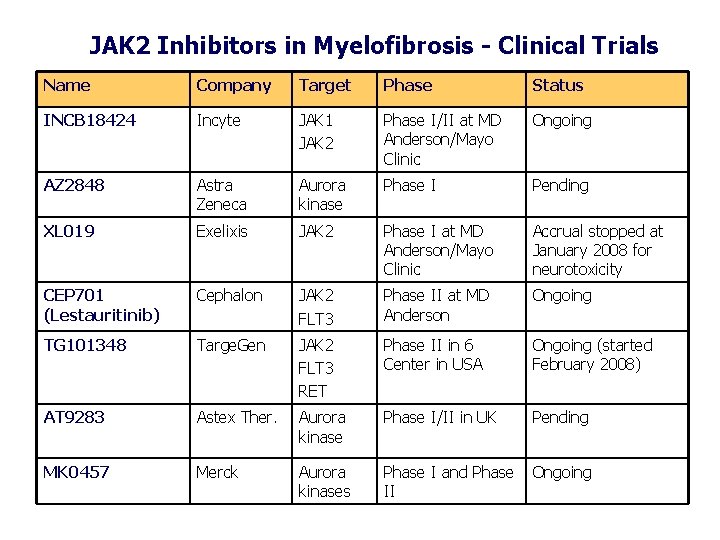
JAK 2 Inhibitors in Myelofibrosis - Clinical Trials Name Company Target Phase Status INCB 18424 Incyte JAK 1 JAK 2 Phase I/II at MD Anderson/Mayo Clinic Ongoing AZ 2848 Astra Zeneca Aurora kinase Phase I Pending XL 019 Exelixis JAK 2 Phase I at MD Anderson/Mayo Clinic Accrual stopped at January 2008 for neurotoxicity CEP 701 (Lestauritinib) Cephalon JAK 2 FLT 3 Phase II at MD Anderson Ongoing TG 101348 Targe. Gen JAK 2 FLT 3 RET Phase II in 6 Center in USA Ongoing (started February 2008) AT 9283 Astex Ther. Aurora kinase Phase I/II in UK Pending MK 0457 Merck Aurora kinases Phase I and Phase II Ongoing

The First Results with a JAK 2 Inhibitor in Myelofibrosis- INCB 18424 Rationale: INCB 18424 is a small molecule that inhibits specifically JAK 2 Design: Phase I/II trial in patients with PMF and post PV/ET MF (both JAK 2 V 617 F and wild type). Dose escalating from 25 mg PO BID to 50 mg PO BID Response: • Six patients had a marked reduction in splenomegaly and symptomatic improvement • Serial JAK 2 testing had gradual reduction in the percentage of PCR product with the V 617 F mutation Toxicity: No significant toxicity Verstovsek et al, ASH 2007

Inhibitors of JAK 2 Signal Transduction • Toxicities related to inhibition of JAKs other than JAK 2 • First results highly encouraging • Most appropriate candidates • Optimum dose and schedule
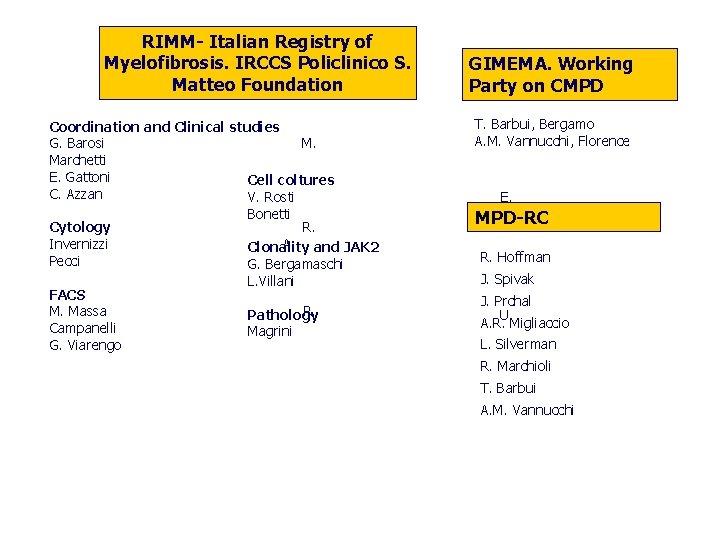
RIMM- Italian Registry of Myelofibrosis. IRCCS Policlinico S. Matteo Foundation Coordination and Clinical studies G. Barosi M. Marchetti E. Gattoni Cell coltures C. Azzan V. Rosti Bonetti Cytology R. Invernizzi A. and JAK 2 Clonality Pecci G. Bergamaschi L. Villani FACS M. Massa R. Pathology Campanelli Magrini G. Viarengo GIMEMA. Working Party on CMPD T. Barbui, Bergamo A. M. Vannucchi, Florence E. MPD-RC R. Hoffman J. Spivak J. Prchal U. A. R. Migliaccio L. Silverman R. Marchioli T. Barbui A. M. Vannucchi
- Slides: 37