CONTEMPORARY PHARMACEUTICAL COMPOUNDING Dr J Domenech From International












































































































- Slides: 110

CONTEMPORARY PHARMACEUTICAL COMPOUNDING Dr. J. Domenech From International Journal of Pharmaceutical Compounding

Role of the Compounding Pharmacist • “Individualizing Drug Therapy”

IJPC First Issue Cover


OUTLINE • Introduction • Compounded Pharmaceuticals • U. S. Pharmacopeia • FDA and Contemporary Compounding • Current USP Compounding Activities • New Drug Delivery Systems • Summary

INTRODUCTION • History of Pharmacy Compounding in the United States • Reasons for the Growth of Compounding • Special Patient Populations • Examples of Pharmaceutical Compounding

History of Pharmacy Compounding in the U. S. • In the past, Compounding Was Pharmacy • 1900 s gave way to commercially prepared pharmaceuticals • Many strengths/dosage forms available • Economics changed all that • Limited strengths/dosage forms • “One Size Fits All” approach

Reasons for the Growth of Pharmacy Compounding: • Limited dosage forms • Limited strengths • Home health care • Hospice • Nonavailable drug products/combinations • Discontinued Drugs • Drug Shortages • Orphan drugs • Veterinary compounding • New therapeutic approaches • Special Patient Populations

SPECIAL PATIENT POPULATIONS • Pediatrics • Geriatrics • Bioidentical Hormone Replacement Therapy • Pain Management • Dental Patients • Environmentally & Cosmetic Sensitive • Sports Injuries • Veterinary Compounding • Small, Large, Herd, Exotic, Companion



MEETING PATIENTS NEEDS • Traditional Dosage Forms • New Dosage Forms

COMPOUNDED DOSAGE FORMS Oral Solids (Capsules, Tablets) Oral Liquids (Solutions, Susp, Emulsions) Topicals (Creams, Ointments, Gels) Suppositories, Inserts Injectables Many, many others….

NEWER COMPOUNDED DOSAGE FORMS • Oral • Topical • Parenteral • Specialty

RAPID-DISSOLVING TABLETS • Active Drug • Lactose • PEG 3350 qs 70 mg 30 mg • Actual size depends upon mold. • ‘Bridging’ mechanism

Compounded Gummy Bears

GUMMY GELS • Fentanyl citrate 1. 884 mg • Chewable gummy gel base 23. 35 g • Bentonite 500 mg • Aspartame 500 mg • Acacia powder 500 mg • Citric acid monohydrate 650 mg • Flavor concentrate 10 -12 drops

VETERINARY ORAL PASTE • Ingredient #1 • PEG 300 • PEG 3350 35 • Prop Glycol • Molasses • Peanut Butter • Hydrog Veg Oil #2 65 25 - #3 25 25 50 50 - #4 25 35 65

ORAL PASTES • VANCOMYCIN PASTE • (VANC PASTE) • • Vancomycin 500 mg • Aspartame 200 mg • Flavor qs • Sodium benzoate 200 mg • Methylcellulose 2% Gel qs 100 m. L

Compounded Lollipops

LOLLIPOPS • Sodium chloride 46. 56 g • Potassium chloride 3 g • Calcium lactate 6. 12 g • Magnesium citrate 2. 04 g • Sodium bicarbonate 22. 44 g • Sodium phosphate monobasic 3. 84 g • Silica gel 3. 6 g • Flavor qs • PEG 1450 qs

Compounded Popsicles

POPSICLES • NYSTATIN POPSICLES • • --------------------- • Nystatin powder 2, 500, 000 u • Sorbitol 70% solution 20 m. L • Syrup 50 m. L • Flavoring (banana, etc. ) 5 m. L • Purified water qs 300 m. L

TROCHES/LOZENGES • TESTOSTERONE 2 MG TROCHES • • Testosterone 24 mg • Citric acid 300 mg • Stevia powder 250 mg • Saccharin sodium 30 mg • Polyethylene glycol 1450 20 g • Citrus flavor qs

SUBLINGUAL DROPS • TESTOSTERONE 10 mg/0. 1 m. L SL • • Testosterone • Saccharin • Silica gel • Tangerine oil • Almond oil qs 1 g 100 mg 200 mg qs 10 m. L

Compounded PLO Gels

TOPICAL PLO GELS • PROMETHAZINE HCL 50 mg/m. L PLO GEL • • -------------------------Promethazine HCl 5 g • Purified water 4 m. L • Lecithin: Isopropyl palmitate 22 m. L • Pluronic F 127 30% Gel qs 100 m. L

TOPICAL PLO GELS • Capsaicin 75 mg • Ketamine HCl 2 g • Ketoprofen 10 g • Ethoxy diglycol 10 m. L • Lecithin: Isopropyl palmitate 22 m. L • Pluronic F 127 30% gel qs 100 m. L

RAPID-PENETRATING TOPICALS • PROGESTERONE 50 mg/m. L CLEAR SOLUTION • • Progesterone 5 g • Benzyl alcohol 20 m. L • Alcohol, absolute 20 m. L • DMSO 20 m. L • Propylene glycol qs 100 m. L

LIPID CRYSTALS CREAM • ANTHRALIN 1% IN LIPID CRYTALS • • Anthralin • Glyceryl laurate • Glyceryl myristate • Citric acid • Sodium hydroxide • Purified water 1 g 7 g 21 g 1 g qs 140 mg 100 g



Compounding Parenterals

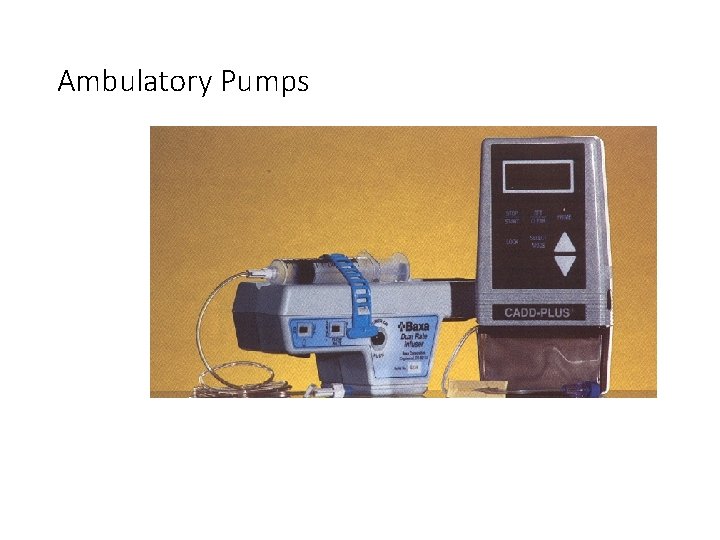
AMBULATORY PUMP INFUSION SOLUTION • CEFTAZIDIME 20 mg/m. L • • --------------------- • Ceftazidime 2. 5 g • Sterile water for injection qs • 0. 9% Sodium chloride inj qs 125 m. L

Ambulatory Pumps

INTRATHECAL INJECTION • Fentanyl citrate 314 μg • Bupivacaine HCl 100 mg • Baclofen 500 μg • 0. 9% Sodium chloride inj. qs 20 m. L

SPONGE DISKS • VANCOMYCIN SPONGE DISKS • • ---------------------- • Vancomycin HCl 5 mg • Sponge (collagen or gelatin) qs

IMPLANTABLE BEADS • TOBRAMYCIN IMPREGNATED POLYMETHYLMETHACYLRATE BEADS • • ---------------------- • Tobramycin sulfate 1. 2 g • Palacos Bone cement 40 g

IONTOPHORETIC SOLUTION • Dexamethasone • Sterile water for injection sodium phosphate 400 mg qs 100 m. L

Iontophoresis Unit

Iontophoresis Unit

Iontophoresis Unit

Inside Iontophoresis Unit

Size of a Dupel Iontophoresis Unit

PHONOPHORESIS PREPARATIONS • HYDROCORTISONE 10% PHONOPHORESIS GEL • • Hydrocortisone 10 g • Carbopol 940 1. 25 g • Propylene glycol 15 m. L • Methylparaben 200 mg • Propyleparaben 100 mg • Purified water qs 100 m. L • Sodium hydroxide 10% Sol qs

Compounding Oral Inhalation Solutions

Compounded Oral Inhalation Solutions

U. S. PHARMACOPEIA Setting Standards for Drugs in the U. S. since 1906

Pharmacopeia Development

Pharmacopeia • Pharmakon drug • poiein to make • Used together in Pharmacopeia means any recipe or formula or other standards required to make or prepare a drug. • 1580 Bergamo, Italy…. . first used in connection with a local book of drug standards.

Pharmacopeias • Local, City and National Pharmacopeias in Europe • The London, Edinburgh and Dublin Pharmacopeias were official until 1864 • Replaced by the British Pharmacopoeia • How about in the U. S. ?

Pharmacopoeias of the U. S. • 1778 Lititz Pharmacopeia • First Pharmacopeia in the U. S. • Published in Lititz, Pennsylvania for use by the Military Hospital of the U. S. Army • 1808 Massachusetts Medical Society • published a 272 page pharmacopeia with information on 536 drugs and preparations

Pharmacopoeias of the U. S. • Jan 1817 • • Dr. Lyman Spalding Submitted a plan Medical Society of the County of New York Creation of a national pharmacopeia ---Divided U. S. into 4 geographical districts Medical schools and societies were to develop a pharmacopeia and appoint delegates to a general convention to be held in Washington, DC

Pharmacopoeias of the U. S. • Jan 1820 First U. S. Pharmacopeial Convention • Only 2 districts submitted plans • These were reviewed, consolidated and adopted. • Dec 1820 First U. S. Pharmacopeia was published • 272 pages containing 217 drugs/preparations

USP I • Preface: (in part) • It is the object of the Pharmacopeia to select from among substances which possess medicinal power, those, the utility of which is most fully established and best understood; and to form from them preparations and compositions, in which their powers may be exerted to the greatest advantage……. .

USP AND CONTEMPORARY COMPOUNDING

U. S. PHARMACOPEIA AND FDAMA • 1985 USP Convention • Resolution 4 • Compounding Information in the USP • Resolution 5 • Standards for Repackaged and Compounded Parenterals • 1990 USP Convention • Established the Expert Advisory Panel on Pharmacy Compounding

Resolution #4 • Be it resolved that the COR examine the desirability and feasibility of developing, with a view to inclusion in the USP, the following types of information: • 1. The short-term stability of drugs when dissolved in common diluents and stored in common standardized containers and/or delivery systems at room, refrigerator and freezer temperatures;

Resolution #4 (cont’d) • 2. p. Ka and minimum solubility of drugs in common diluents; and • 3. p. H, osmolality and osmolarity of reconstituted injectables and liquid dosage forms.

Resolution #5 • Be it resolved that the COR be charged with the responsibility for providing standards and test methods; specifications for packaging, labeling, and storage; guidelines for appropriate documentation; and, where necessary, procedures for compounding parenteral preparations.

PSD Subcommittee • Expert Advisory Panel on Pharmacy Compounding was formed to advise the PSD Subcommittee • Also, the Review Panel on Pharmacy Compounding Practices was formed to assist the Expert Advisory Panel by providing immediate expert review on materials produced by the Panel

Expert Advisory Panel • Oct 1993 First meeting • Organized into 2 groups • General Chapter Group • to prepare a general informational chapter on compounding • Monograph Group • develop monographs for specific preparations • those widely compounded but not available commercially

U. S. PHARMACOPEIA AND FDAMA • 1993 -2000 Expert Advisory Panel Activities • I. General Chapter Group • <795> Pharmacy Compounding • II. Monograph Group • develop monographs for specific preparations

FDA AND CONTEMPORARY COMPOUNDING

FDA ACTIVITIES • Mid 1990 s FDA began investigating a number of pharmacies that were compounding large quantities of selected drug products. • Manufacturing under the guise of compounding • “New Drugs”

Food and Drug Administration Activities • FDA considered compounded preparations as “New Drugs” and subject to the New Drug Provisions • • IND NDA Safety Efficacy • Enforcement Activities

FDAMA 97 Passage • Pharmacy professional organizations • U. S. Congress • FDAMA 97 • Compounding provisions

FOOD AND DRUG ADMINISTRATION MODERNIZATION ACT • New Drug Requirements • shall not apply to a drug product if the drug product is compounded for an individual patient based on the unsolicited receipt of a valid prescription order…. . if the compounding is by: • a licensed pharmacist • a licensed physician

FOOD AND DRUG ADMINISTRATION MODERNIZATION ACT • Anticipatory Compounding • Physician-Patient-Pharmacist “Triad”

FOOD AND DRUG ADMINISTRATION MODERNIZATION ACT • Compounding must be done using the following sources of ingredients: • USP/NF monographs • Commercial products • Bulk Drug Substances List (being developed)

FOOD AND DRUG ADMINISTRATION MODERNIZATION ACT • Compounding cannot be done from: • Drugs on the “Negative List” • drugs that have been withdrawn due to safety or efficacy reasons • List was developed

FOOD AND DRUG ADMINISTRATION MODERNIZATION ACT • Cannot compound regularly or in inordinate amounts any drug products that are essentially copies of commercially available products

FOOD AND DRUG ADMINISTRATION MODERNIZATION ACT • Cannot compound a drug product that “presents demonstrable difficulties for compounding that reasonably demonstrate an adverse effect on the safety and effectiveness of that drug product”. (list)

FOOD AND DRUG ADMINISTRATION MODERNIZATION ACT • Memorandum of Understanding • Distribution of inordinate amounts interstate • Handling of complaints

FOOD AND DRUG ADMINISTRATION MODERNIZATION ACT • Advertising • The pharmacy, pharmacist or physician cannot advertise or promote the compounding of any particular drug, class of drug, or type of drug.

FDA Modernization Act of 1997 • FDA Advisory Committee on Compounding • Function: to advise the FDA in the areas of bulk drug substances, safety and efficacy and difficult-to-compound products. • FDA Pharmacy Compounding Steering Committee (Internal to FDA)

FDA Modernization Act of 1997 Three Lists • Products not to be compounded because they were withdrawn from the market based on safety and efficacy concerns • Bulk drug substances of proven quality accepted for use in pharmacy compounding • Difficult-to-compound products

IMPLEMENTATION OF FDAMA • Ongoing since 1997 • FDA Steering Committee (Internal) • FDA Compounding Advisory Committee (External) • Work with USP

USP I • Preface: (in part) • It is the object of the Pharmacopeia to select from among substances which possess medicinal power, those, the utility of which is most fully established and best understood; and to form from them preparations and compositions, in which their powers may be exerted to the greatest advantage……. .

FDAMA IMPLEMENTATION AND THE USP • <1161>Pharmacy Compounding Practices became <795 Pharmacy Compounding • Monographs of accepted bulk drug substances are being developed • <1206> Sterile Preparations-Pharmacy Practices has been recommended as guidelines for sterile preparations compounding…being renumbered as <797> • New chapters being written

Current USP Compounding Activities

USP 2000 -2005 • New structure from Committee of Revision to Expert Committees • Compounding Pharmacy Expert Committee • General Chapters, incl <795> • Nonsterile preparation monographs • Parenteral Products--Compounding and Preparation Expert Committee • General Chapters, incl <1206> • Sterile preparation monographs

USP Convention 2000 • Resolution • Continue to develop and institute, in collaboration with other organizations as appropriate, specific initiatives focused on the development of appropriate compounding guidelines and monographs for non-commercially available, but commonly prescribed, medicines and dosage forms for use in special populations, notably neonatal, pediatric, geriatric, and terminally ill patients.

U. S. PHARMACOPEIA AND FDAMA • Activities to date: • 15 official compounding monographs • 8 more stability studies underway • 6 formulas being processed through PF • 2 official chapters and 2 additional chapters in process: • Pharmacy Calculations • Good Compounding Practices

CURRENT ACTIVITIES OF PHARMACY COMPOUNDING EXPERT COMMITTEE • Survey of compounding pharmacists in hospitals, community pharmacies and long-term care facilities (August 2000) • List of over 150 preparations, mostly pediatric, that need to be considered. • 2000 Resolution:

U. S. PHARMACOPEIA • 2001 • Recent survey listed over 1000 other preparations need monographs • Well over 5, 000 different formulations routinely compounded


FDAMA and the 9 th District • Early 2001 • the Ninth Circuit ruled that the FDAMA section dealing with compounding was invalid in the 9 th Circuit District (NV, CA, WA, OR, MT, ID, AZ, AK, HI) but still in effect in the rest of the US.

FDAMA and the 9 th District • April 29, 2002 • U. S. Supreme Court ruled the advertising restrictions unconstitutional and the section not severable. • Entire 503 a now is thrown out and nonenforceable

SUMMARY • Pharmacy compounding is now legally recognized by the FDA, the Supreme Court, Congress, etc. as a necessary component of quality health care • Emphasis on quality of compounding is increasing with documentation of quality being recommended and required • Clinical pharmacy becomes more of a reality with compounding pharmacy




A LOOK INTO THE NEAR FUTURE New Compounded Drug Delivery Systems (DDS)

Future Trends • Adhesive Site-Specific DDS • Antibody-Based DDS • Biocompatible Microsphere DDS • Biodegradable Polymers DDS • Biologic-Based DDS • Electromagnetic/Radiation- Activated DDS

Future Trends • Immunomodulator DDS • Implant-Enhanced DDS • Microorganism-Containing • Lipid Microcylinders • Liposome Enhancements • Living-Cell Therapies Microcapsule DDS

Future Trends • Magnetic System DDS • Maze-Escape DDS • Monoclonal Antibody DDS • Novel Nasal DDS • New Osmotic DDS • Transmucosal DDS • Polymer Drug Complex DDS

Future Trends • Pulsatile DDS • Resealed Erythrocyte DDS • Respiratory DDS • Self-Assembling Controlled-Release DDS • Programmed Skin-Surface DDS

NANOTECHNOLOGY: The Ultimate Alchemy

NANOTECHNOLOGY • The art and science of building molecular structures so they are sufficiently large and complex to function as machines or devices • Atomically precise, functional machine systems developed on the scale of the nanometer • Builds objects atom by atom, molecule by molecule

POTENTIAL PRODUCTS • Activated Pharmaceuticals (Magic Bullets) • Cell-herding machines to stimulate rapid wound healing • Nanosurgeons to repair damaged cellular parts • Nanocruisers to attack viruses and bacteria

FORECASTS: 2 -5 YEARS • Inexpensive handheld biosensors built on the basis of nanoscale ion channel switches • Simple detection of diseases, within minutes, from a small sample of saliva or blood

FORECASTS • DNA vaccines will begin to be available in the next 5 -10 years • Superior and safer than traditional vaccines • Ability to directly mimic body components and can “rebuild” worn, defective, damaged, diseased cells/tissues/organs • Blood products, artificial skin products, bioartificial organs, blood vessels

FORECASTS • IF a breakthrough to a universal assembler occurs during the next 1015 years, an entirely new field of “nanomedicine” and “nanopharmacy” will emerge by 2020.

NANOMEDICINE • Monitoring, repair, construction and control of human biological systems at the molecular level, using engineered nanodevices and nanostructures.

NANOPHARMACY Preparation and delivery of ultra-small pharmaceuticals, therapeutic substances and delivery systems.

NANOPHARMACY AND NANOPHARMACEUTICALS • Motors consisting of, for example, ATPase molecules with a metallic substrate and a chemical “propeller” on the other. As the ATP breaks down, the biomotor moves. • This motor may be able to compound tiny quantities of drugs and pump them directly to the target tissues.

NANOPHARMACY AND NANOPHARMACEUTICALS • The uses of biomolecular motors could be used for sensing or placing in living cells as a pharmacy to deliver medicine when required.

NANOPHARMACY AND NANOPHARMACEUTICALS • New formulations and routes for drug delivery • Pharmaceuticals based on an individuals genome

CONCLUSIONS • We must live in today and prepare for tomorrow • Compounding pharmacists roles in “individualizing drug therapy” is preparing the foundation for the “NANOPHARMACY” of tomorrow.