Technology transfer of Biopharmaceutical product 2016 08 29

생물 의약품 기술이전: Technology transfer of Biopharmaceutical product 2016 -08 -29





참고문헌 • ISPE Good Practice Guide: Technology Transfer (Sept. 2003) • Annex 7 WHO guidelines on transfer of technology in pharmaceutical manufacturing, WHO Technical Report Series, No. 961, 2011 • ICH Q 5 E Comparability of Biotechnolgical/Biological Products subject to Changes in their Manufacturing Process • ICH Q 7 A GMPs for Active Pharmaceutical Ingredients • Analytical chemistry in a GMP environment a practical guide James M Miller • Technology Transfer Herwig Kapeller, Novarits Pharmaceuticals Corps. ISPE Sep. 10, 2009

생물의약품의 기술 이전 유형 1. 2 “a logical procedure that controls the transfer of any process together with its documentation and professional expertise between development and manufacture or between manufacture sites” 1. 5 Transfer of technology requires a documented, planned approach using trained and knowledgeable personnel working within a quality system, with documentation of data covering all aspects of development, production and quality control. Usually there is a sending unit (SU), a receiving unit and the unit managing the process, which may or may not be a separate entity. ○ 이 지침에서의 기술이전은 의약품의 개발에서 시판에 이 르기까지의 지식과 경험을 토대로 제조소 간 제제개발 및 제조·품질관리에 대한 전문기술 및 문서 등을 이전하는 논 리적 절차를 말한다. Annex 7 WHO guidelines on transfer of technology in pharmaceutical manufacturing
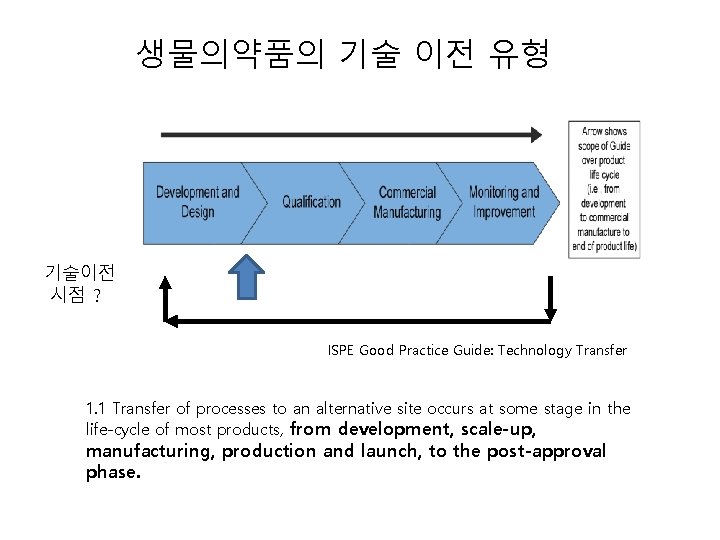
생물의약품의 기술 이전 유형 기술이전 시점 ? ISPE Good Practice Guide: Technology Transfer 1. 1 Transfer of processes to an alternative site occurs at some stage in the life-cycle of most products, from development, scale-up, manufacturing, production and launch, to the post-approval phase.
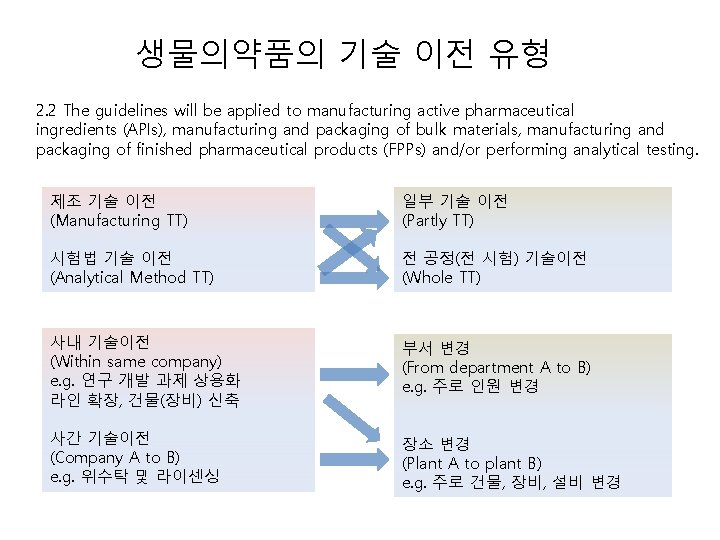
생물의약품의 기술 이전 유형 2. 2 The guidelines will be applied to manufacturing active pharmaceutical ingredients (APIs), manufacturing and packaging of bulk materials, manufacturing and packaging of finished pharmaceutical products (FPPs) and/or performing analytical testing. 제조 기술 이전 (Manufacturing TT) 일부 기술 이전 (Partly TT) 시험법 기술 이전 (Analytical Method TT) 전 공정(전 시험) 기술이전 (Whole TT) 사내 기술이전 (Within same company) e. g. 연구 개발 과제 상용화 라인 확장, 건물(장비) 신축 부서 변경 (From department A to B) e. g. 주로 인원 변경 사간 기술이전 (Company A to B) e. g. 위수탁 및 라이센싱 장소 변경 (Plant A to plant B) e. g. 주로 건물, 장비, 설비 변경
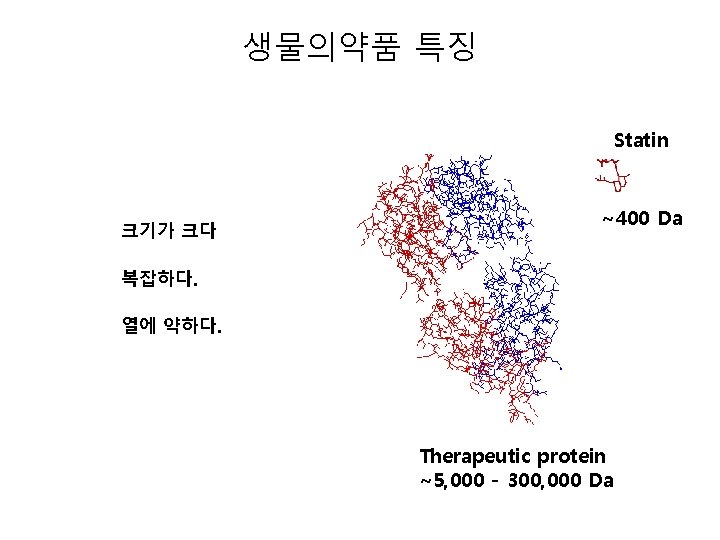
생물의약품 특징 Statin 크기가 크다 ~400 Da 복잡하다. 열에 약하다. Therapeutic protein ~5, 000 - 300, 000 Da
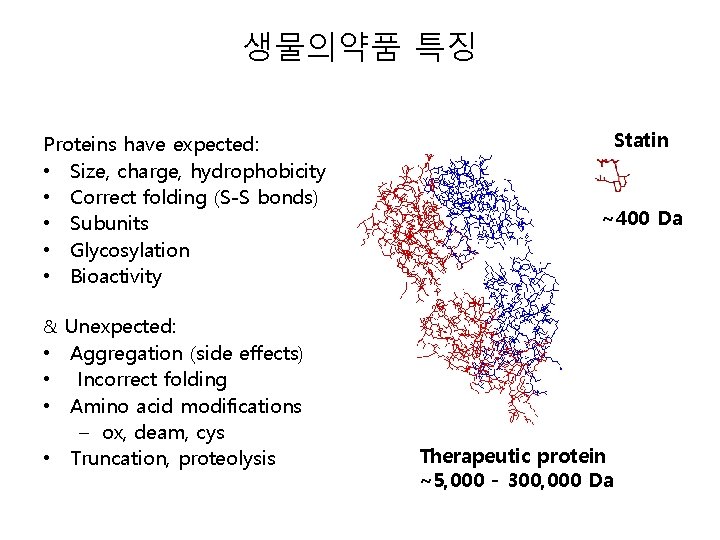
생물의약품 특징 Proteins have expected: • Size, charge, hydrophobicity • Correct folding (S-S bonds) • Subunits • Glycosylation • Bioactivity Statin ~400 Da & Unexpected: • Aggregation (side effects) • Incorrect folding • Amino acid modifications – ox, deam, cys • Truncation, proteolysis Therapeutic protein ~5, 000 - 300, 000 Da
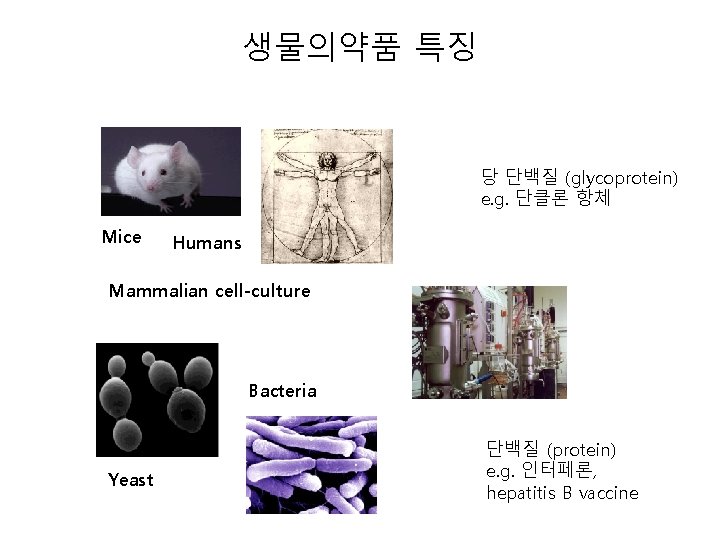
생물의약품 특징 당 단백질 (glycoprotein) e. g. 단클론 항체 Mice Humans Mammalian cell-culture Bacteria Yeast 단백질 (protein) e. g. 인터페론, hepatitis B vaccine

생물의약품 특징 Bacteria Mycoplasma Mice Fungi Humans Mammalian cell-culture Bacteria Yeast Viruses TSE agents


생물의약품 공정의 특징 • Cell banking system - Master Cell Bank (MCB) - Working Cell Bank (WCB) - End of Production Cells (EOP) • Drug substance manufacturing and release - Cultivation - Purification - Virus inactivation/removal step • Drug product formulation and release - Formulation Sterile filtration
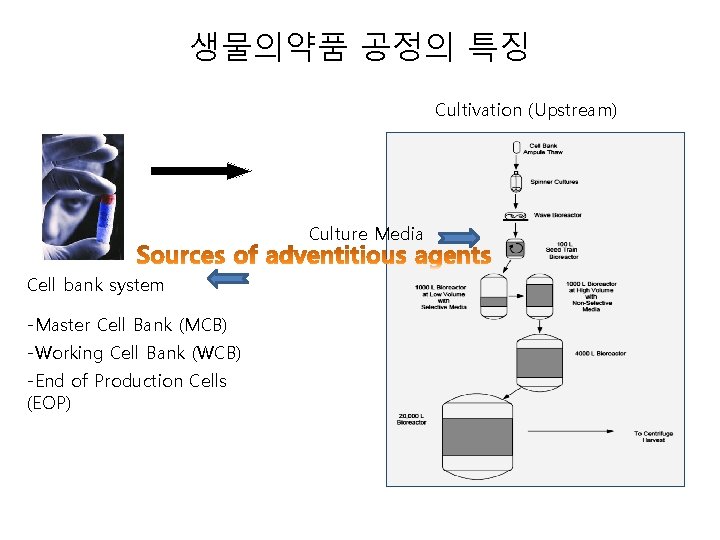
생물의약품 공정의 특징 Cultivation (Upstream) Culture Media Cell bank system -Master Cell Bank (MCB) -Working Cell Bank (WCB) -End of Production Cells (EOP)
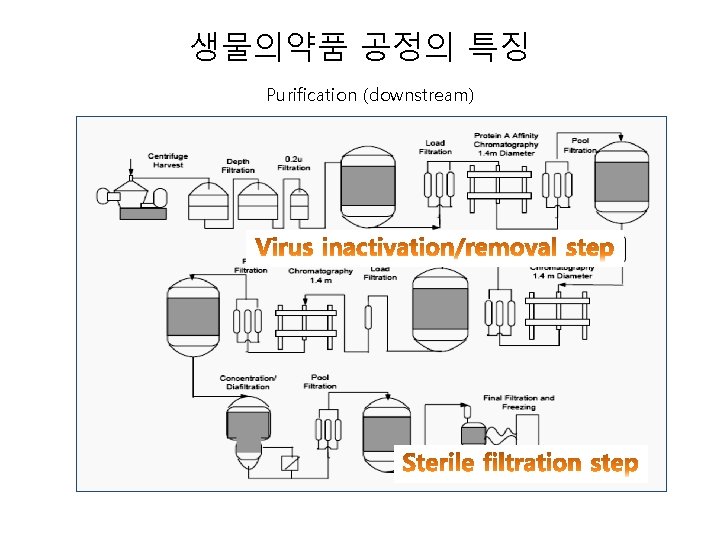
생물의약품 공정의 특징 Purification (downstream)

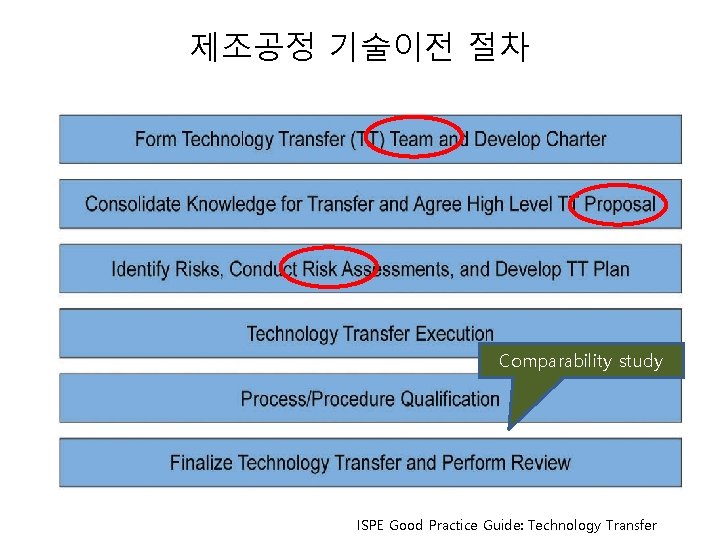
제조공정 기술이전 절차 Comparability study ISPE Good Practice Guide: Technology Transfer




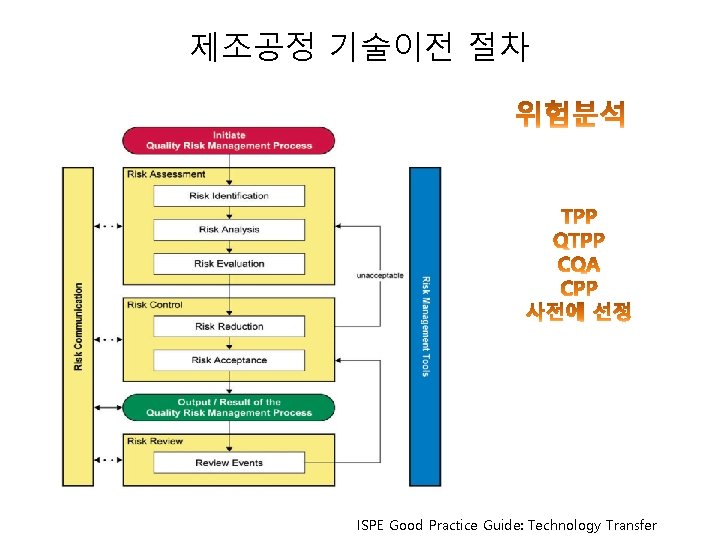
제조공정 기술이전 절차 ISPE Good Practice Guide: Technology Transfer



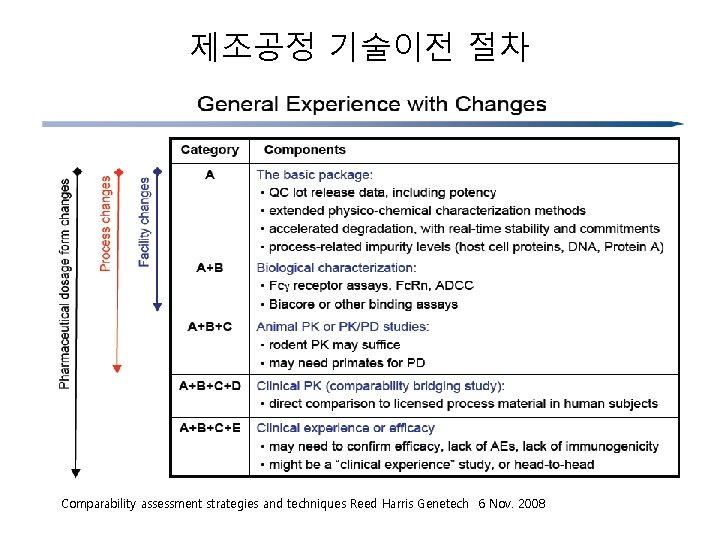
제조공정 기술이전 절차 Comparability assessment strategies and techniques Reed Harris Genetech 6 Nov. 2008

제조공정 기술이전 절차 Comparability Protocol : REMICADE (Centocor) Leiden, specific The Netherlands Malvern process change Malvern, PA, USA Change in column bed heights for two ♦ General Scale-up of seed and production cellfacility♦ in changes from licensed Leiden chromatography steps culture processes ♦ Closed system requiring less stringent gowning procedures ♦ Scale-up of downstream process ♦ Change in cell separation device used during production ♦ Increase use ofcell CIPculture and SIP cycles ♦ Virus filtration in one part ♦ ♦Change in storage conditions for Increased automation harvest supernatant Case studies in the use of Comparability protocols to support changes in. REMICADE bulk manufacturing Michele Myers.

제조공정 기술이전 절차 ♦ In-process testing results at all steps of the process ♦ Cell culture – Viability at thaw in pre-culture – Accumulated generations throughout pre-culture and production cell culture process – Specific productivity in production cell culture process – Product quality throughout the production cell culture process ♦ Harvest Storage – Product quality throughout storage ♦ Viral and DNA clearance studies for those chromatography steps in which column bed heights were changed ♦ Impurity clearance throughout the purification process
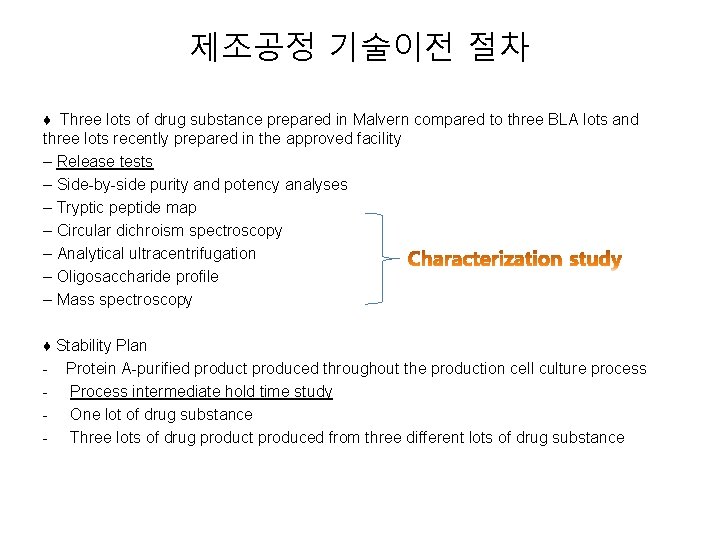
제조공정 기술이전 절차 ♦ Three lots of drug substance prepared in Malvern compared to three BLA lots and three lots recently prepared in the approved facility – Release tests – Side-by-side purity and potency analyses – Tryptic peptide map – Circular dichroism spectroscopy – Analytical ultracentrifugation – Oligosaccharide profile – Mass spectroscopy ♦ Stability Plan - Protein A-purified product produced throughout the production cell culture process - Process intermediate hold time study - One lot of drug substance - Three lots of drug product produced from three different lots of drug substance

제조공정 기술이전 절차 ♦ July 2000: Agency comments on the comparability plan were received – List validation studies related to process transfer – Describe requalification and revalidation plans for lot release and other critical assays to be implemented – Include bioburden and LAL testing of all intermediates generated during downstream processing and Protein A-purified product stability – Include images showing comparability of Regulatory contact • IEF gels • AUC traces • Tryptic peptide maps • SDS-PAGE gels • CD traces • SE-HPLC plots

제조공정 기술이전 절차 Case: An Increase in PRCA Associated with EPREX® § Beginning in 1998, an increased, though still rare, incidence of PRCA was linked to epoetin products, particularly EPREX brand. § Antibodies neutralize all r. Hu. EPOs and patient EPO • • One case per 5, 000 patient years Marketed in over 90 countries EPREX and PRCA § Erythropoietin – a natural protein that stimulates the production of red blood cells (erythrocytes) § Recombinant Erythropoietin – manufactured form of the natural protein (generic name is epoetin) used to treat anemia. § EPREX § Pure Red Cell Aplasia (PRCA) – severe and rare form of anemia ® – a brand of epoetin marketed outside of the U. S.
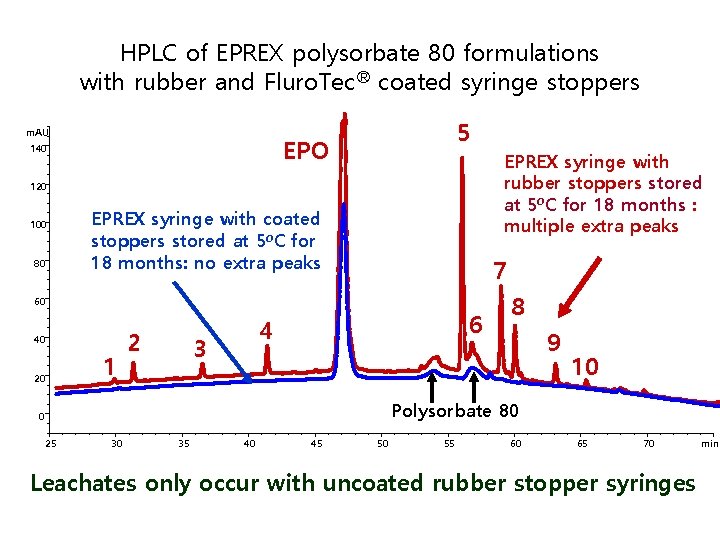
HPLC of EPREX polysorbate 80 formulations with rubber and Fluro. Tec® coated syringe stoppers m. AU 5 EPO 140 EPREX syringe with rubber stoppers stored at 5 o. C for 18 months : multiple extra peaks 120 100 80 EPREX syringe with coated stoppers stored at 5 o. C for 18 months: no extra peaks 7 60 40 20 1 2 6 4 3 8 9 10 Polysorbate 80 0 25 30 35 40 45 50 55 60 65 70 Leachates only occur with uncoated rubber stopper syringes min

제조공정 기술이전 절차 4년간 100여명 조사 결과 § Uncoated rubber 와 formulation에 쓰이는 Polysorbate 80 반응하여 유출물 발생 § 이 유출물이 adjubant로 작용하여 anti-EPO antibodies 생성을 유발 함 § Fluro. Tec coated stoppers 사용으로 유출물을 줄인 후로는 PRCA 발병율이 baseline으 로 다시 감소 Immunogenicity of Therapeutic Proteins: A Case Report Fred Bader Johnson & Johnson

생물의약품 시험법 특징 시험법 및 규격 설정의 참고문헌 USP, EP, 대한약전 etc. : Official compendia (Monographs/ General chapters) ICH Q 6 B Specifications: Test Procedures and Acceptance Criteria for Biotechnological/Biological Products, 1999 생물공학 의약품/생물학적 제제의 규격설정의 가이드 (Guidance on the Specifications of Biotechnological /Biological products, 2004) 식약청 고시 의약품등 기준및 시험방법 심사의뢰서 심사규정 개정 생기?

생물의약품 시험법 특징 1. 확인 시험 (Identity tests) 성상, SDS-PAGE, Western blotting, Peptide mapping N-terminal sequence, IEF, Monosaccharide id. etc. 2. 순도 시험 (Purity tests) SDS-PAGE, HPLC (RP, IEX, SEC), HCP, HCD etc. 3. 함량 시험 (Content tests) UV(A 280), Sialic acid content, ELISA etc. 4. 역가 시험 (Potency tests) Cell-based assay, Enzymatic assay, animal assay, etc. ※ 공정서 시험 p. H, 불용성 이물, 불용성 미립자, 엔도톡신, 무균 이상독성부정 (마우스, 기니픽), 삼투압, 마이코플라즈마, etc.
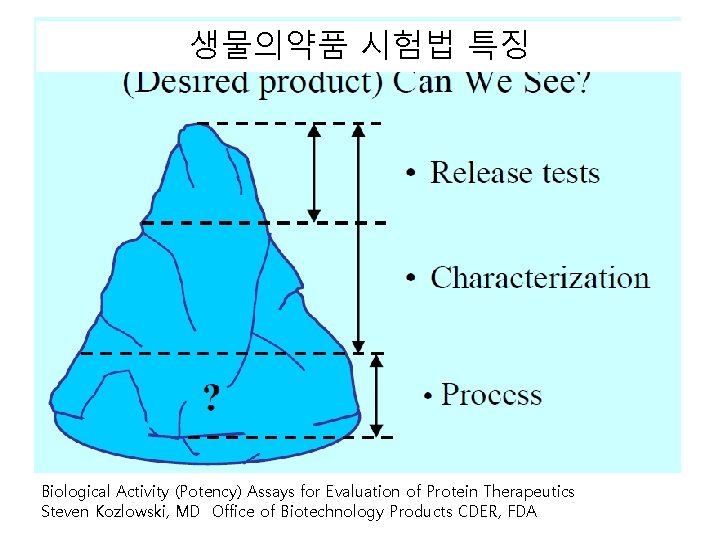
생물의약품 시험법 특징 Biological Activity (Potency) Assays for Evaluation of Protein Therapeutics Steven Kozlowski, MD Office of Biotechnology Products CDER, FDA
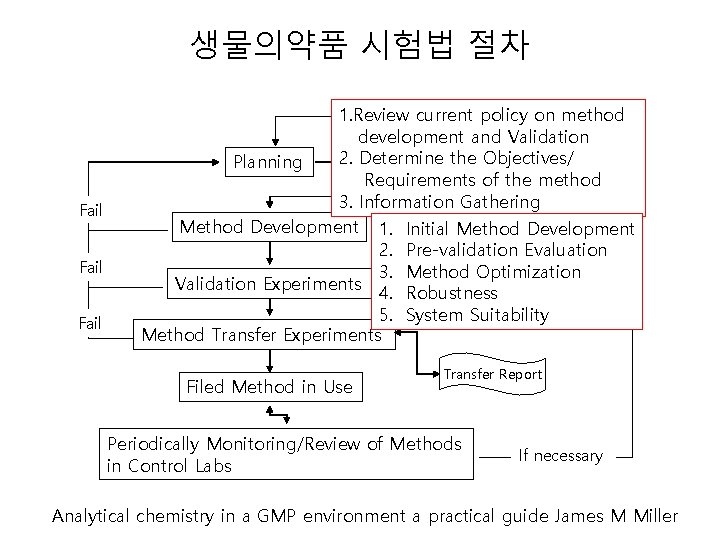
생물의약품 시험법 절차 Fail 1. Review current policy on method development and Validation 2. Determine. Development the Objectives/ Planning Plan Project Requirements of the method 3. Information Gathering Method Development 1. Initial Method Development Report Draft Procedure 2. Pre-validation Evaluation 3. Method Optimization Validation Experiments 4. Robustness Validation Report 5. System Suitability Method Transfer Experiments Filed Method in Use Transfer Report Periodically Monitoring/Review of Methods in Control Labs If necessary Analytical chemistry in a GMP environment a practical guide James M Miller




Potential problems with Student’s t test as an equivalence test Student’s t test often is applied to compare results from a new analytical test method with those from another test method. the following hypotheses are set up in Student’s t test. 1. Student’s t test assumes the mean values of two populations are equal and requires the experimenter to prove that they are different. 2. Experimenter is attempting to prove that the mean values of two populations are different with high confidence rather than attempting to prove they are the same. Reference Pharmaceutical Technology SEPTEMBER 2005 Analytical Method Equivalency An Acceptable Analytical Practice

Two One-Sided T test as an equivalence test Ha is represented by the region where the true population difference between two sets of data is within the lower and upper acceptance limits. 1) more likely to conclude that there is a lack of equivalence in the presence of too much variation, the opposite of the effect seen with the two sample Student t test. 2) Two one-sided test is not limited to the bioequivalence problem but is easily extended to other applications such as method transfers or the comparison of manufacturing process results after process modifications. 3) The test also has been used to determine equivalence between robotic and manual assays as well as content uniformity methods for tablets of small molecules*. Reference Pharmaceutical Technology SEPTEMBER 2005 Analytical Method Equivalency An Acceptable Analytical Practice * 6. K. R. Lung et al. , “Statistical Method for the Determination of Equivalence of Automated Test Procedures, ” J. Automated Methods and Management in Chemistry 25 (6), 123– 127 (2003).
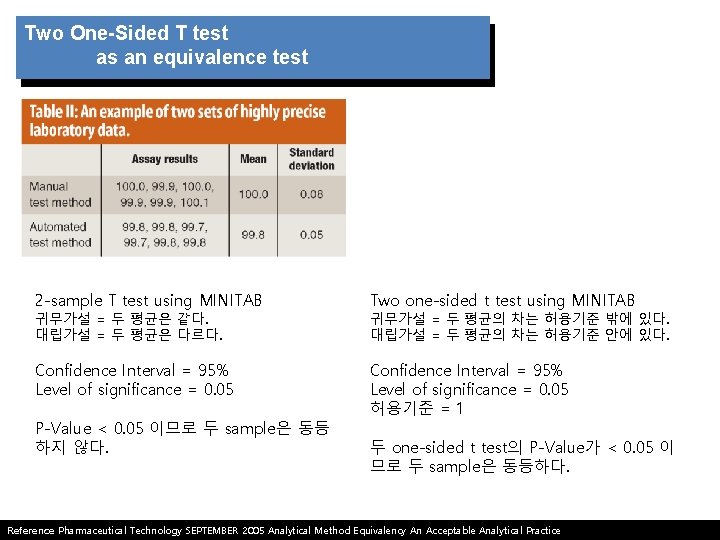
Two One-Sided T test as an equivalence test 2 -sample T test using MINITAB Two one-sided t test using MINITAB Confidence Interval = 95% Level of significance = 0. 05 허용기준 = 1 귀무가설 = 두 평균은 같다. 대립가설 = 두 평균은 다르다. P-Value < 0. 05 이므로 두 sample은 동등 하지 않다. 귀무가설 = 두 평균의 차는 허용기준 밖에 있다. 대립가설 = 두 평균의 차는 허용기준 안에 있다. 두 one-sided t test의 P-Value가 < 0. 05 이 므로 두 sample은 동등하다. Reference Pharmaceutical Technology SEPTEMBER 2005 Analytical Method Equivalency An Acceptable Analytical Practice
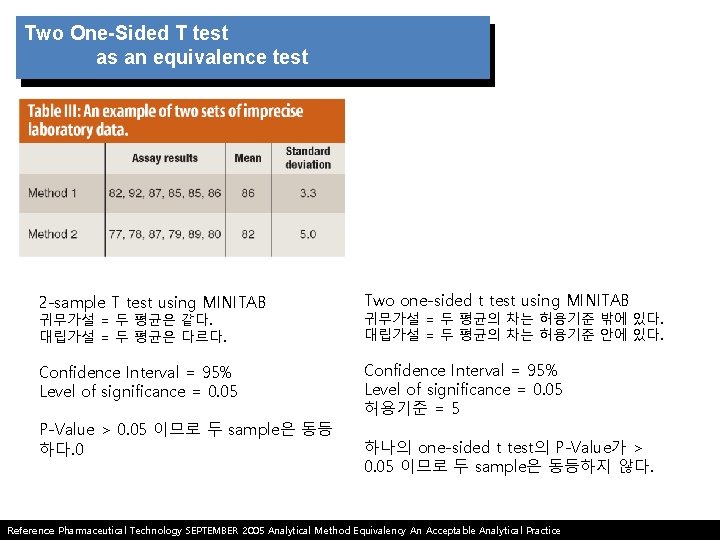
Two One-Sided T test as an equivalence test 2 -sample T test using MINITAB Two one-sided t test using MINITAB 귀무가설 = 두 평균은 같다. 대립가설 = 두 평균은 다르다. 귀무가설 = 두 평균의 차는 허용기준 밖에 있다. 대립가설 = 두 평균의 차는 허용기준 안에 있다. Confidence Interval = 95% Level of significance = 0. 05 허용기준 = 5 P-Value > 0. 05 이므로 두 sample은 동등 하다. 0 하나의 one-sided t test의 P-Value가 > 0. 05 이므로 두 sample은 동등하지 않다. Reference Pharmaceutical Technology SEPTEMBER 2005 Analytical Method Equivalency An Acceptable Analytical Practice


생물의약품 시험법 특징 역가시험 종류 • Biological Potency assays (bioassay): ► Cellular (i. e. , proliferation activity, signal transduction activation, etc) ► Animal (i. e. , LD 50) • Acellular Potency Assays: � ► Biochemical (i. e. , enzyme activity in vitro) ► Binding (i. e. , ELISA, SRP, etc) • The ideal potency assay should be : ► Reflective of the Product’s mechanism of action (Mo. A) ► Indicative of lot-to-lot consistency during Product Release ► Robust and have consistently low variability and high specificity




- Slides: 53