Basic Principles of GMP Sterile Pharmaceutical Products Annex

Basic Principles of GMP Sterile Pharmaceutical Products Annex 6. TRS 902, 2002 Module 14 | Slide 1 of 62 January 2006 STOP

Sterile Production Objectives l To review basic GMP requirements in the manufacture of sterile pharmaceutical products l To review air classifications for activities related to the manufacture of sterile products l To review the different types of sterilization methods l To review quality assurance aspects in the manufacture and control of sterile products l To consider current issues applicable in your country Module 14 | Slide 2 of 62 January 2006 STOP

Sterile Production GMP Requirements for Sterile Products l Additional rather than replacement l Specific points relating to minimizing risks of contamination ä microbiological ä particulate matter ä pyrogen Module 14 | Slide 3 of 62 January 2006 STOP

Sterile Production General Considerations l l Production in clean areas Appropriate standard of cleanliness Filtered air supplied Airlocks for entry ä personnel and/or equipment ä materials l Separate areas for operations ä component preparation (containers and closures) ä product preparation 1. 1 – 1 -2 ä filling, sterilization, etc. Module 14 | Slide 4 of 62 January 2006 STOP

Sterile Production Premises l Design ä avoid unnecessary entry of supervisors and control personnel ä operations observed from outside l In clean areas, all exposed surfaces ä smooth, impervious, unbroken ä minimize shedding and accumulation of particles, microorganisms ä permit cleaning and disinfection ä no uncleanable recesses, ledges, shelves, cupboards, equipment ä sliding doors undesirable 9. 1 – 9. 6 ä false ceilings sealed Module 14 | Slide 5 of 62 January 2006 STOP

Sterile Production Premises (continued) l In clean areas, all exposed surfaces (2) ä proper installation of pipes and ducts, no recesses, no unsealed openings ä sinks and drains avoided, and excluded in Grade A and B areas – where installed, design, location, maintenance – effective cleanable traps – air breaks preventing backflow – floor channels open and easily cleanable 9. 6. Module 14 | Slide 6 of 62 January 2006 STOP

Sterile Production Premises (continued) l Changing rooms ä designed as airlocks ä effective flushing with filtered air ä separate rooms for entry and exit desirable ä hand washing facilities ä interlocking system for doors ä visual and/or audible warning system l Use filtered air supply to maintain pressure cascade l Pressure differential approximately 10 to 15 Pascals l Zone of greatest risk – immediate environment 9. 7 – 9. 9 Module 14 | Slide 7 of 62 January 2006 STOP

Sterile Production Premises (continued) l Pathogenic, highly toxic, radioactive materials l Pressure cascade may be different l Decontamination procedures – air, equipment, garments l Qualification including airflow patterns ä no risk to the product l Warning system to indicate failure in air supply l Pressure indicators – results regularly recorded l Restricted access – e. g. use of barriers 9. 9 – 9. 12 Module 14 | Slide 8 of 62 January 2006 STOP

Sterile Production Equipment l Conveyer belts l Effective sterilization of equipment l Maintenance and repairs from outside the clean area ä if taken apart, resterilized before use ä use clean instruments and tools l Planned maintenance, validation and monitoring ä equipment, air filtration systems, sterilizers, water treatment systems 10. 1 – 10. 5 Module 14 | Slide 9 of 62 January 2006 STOP

Sterile Production Equipment (continued) l Water treatment plants and distribution system ä design, construction, maintenance ä operation and design capacity ä testing programme l Water for Injection (WFI) ä produced, stored, distributed – prevention of growth of microorganisms ä constant circulation at temperature above 70, or not more than 4 degrees Celsius 10. 6 Module 14 | Slide 10 of 62 January 2006 STOP

Sterile Production Environmental Monitoring - I Microbiological l Air samples l Surface swabs l Personnel swabs Module 14 | Slide 11 of 62 January 2006 STOP

Sterile Production Environmental Monitoring – II Physical l Particulate matter l Differential pressures l Air changes, airflow patterns l Clean up time/recovery l Filter integrity l Temperature and relative humidity l Airflow velocity Module 14 | Slide 12 of 62 January 2006 STOP

Sterile Production Sanitation l Frequent, thorough cleaning of areas necessary l Written programme l Regular monitoring to detect resistant strains of microorganisms l Chemical disinfection l Monitoring of disinfectants and detergents l Dilutions ä clean containers, stored for defined periods of time ä Sterilized before use, when used in Grade A or B areas 3. 1 – 3. 2 Module 14 | Slide 13 of 62 January 2006 STOP

Sterile Production Sanitation (continued) l Monitoring of clean areas l Monitoring of personnel and surfaces after critical operations l Frequent monitoring in areas where aseptic operations are carried out ä settle plates, volumetric air samples, surface sampling (swabs and contact plates) ä sampling methods should not contaminate the area l Results considered when batch release is done 3. 3 Module 14 | Slide 14 of 62 January 2006 STOP
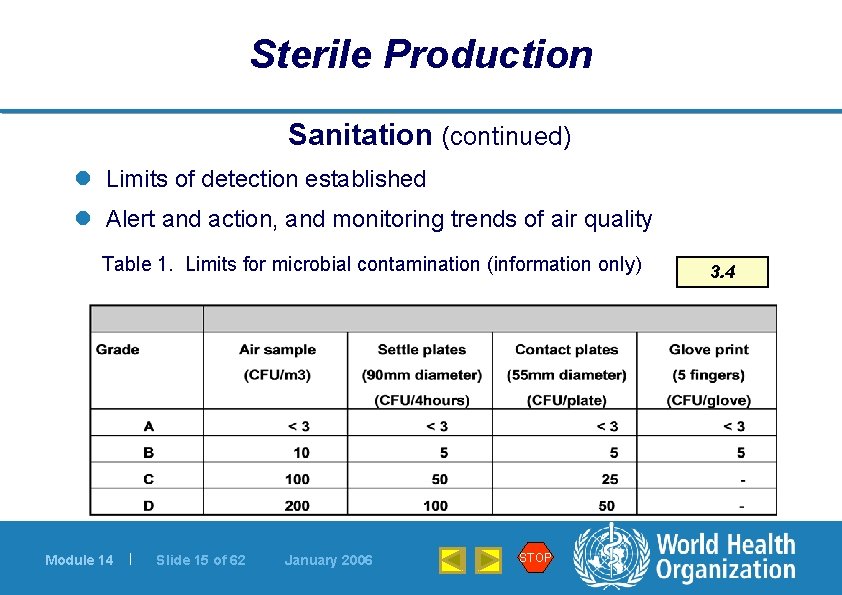
Sterile Production Sanitation (continued) l Limits of detection established l Alert and action, and monitoring trends of air quality Table 1. Limits for microbial contamination (information only) Module 14 | Slide 15 of 62 January 2006 STOP 3. 4

Sterile Production Personnel l Minimum number of personnel in clean areas ä especially during aseptic processing l Inspections and controls from outside l Training to all including cleaning and maintenance staff ä initial and regular ä manufacturing, hygiene, microbiology l Special cases ä supervision in case of outside staff 8. 1 – 8. 3 ä decontamination procedures (e. g. staff who worked with animal tissue materials) Module 14 | Slide 16 of 62 January 2006 STOP

Sterile Production Personnel (continued) l High standards of hygiene and cleanliness l Periodic health checks l No shedding of particles l No introduction of microbiological hazards l No outdoor clothing l Changing and washing procedure l No watches, jewellery and cosmetics Module 14 | Slide 17 of 62 January 2006 8. 4 – 8. 6 STOP

Sterile Production Personnel (continued) l Clothing of appropriate quality: ä Grade D – hair, beard, moustache covered – protective clothing and shoes ä Grade C – hair, beard, moustache covered – single or 2 -piece suit (covering wrists, high neck), shoes – no fibres to be shed ä Grade A and B 8. 7 ä headgear, beard and moustache covered, masks, gloves ä not shedding fibres, and retain particles shed by operators Module 14 | Slide 18 of 62 January 2006 STOP

Sterile Production Personnel (continued) l Outdoor clothing not in change rooms leading to Grade B and C rooms l Change at every working session, or once a day (if supportive data) l Change gloves and masks at every working session l Disinfect gloves during operations l Washing of garments – separate laundry facility l No damage, and according to validated procedures 8. 8 – 8. 9 Module 14 | Slide 19 of 62 January 2006 STOP

Sterile Production Group session 1 l You are asked to visit a factory producing the following product lines: ä injections in ampoules and vials, including insulin, vaccines and heat-stable pharmaceuticals ä sterile eye ointment l Describe the type of facility you would expect to find l List the typical rooms, their purpose and air classification Module 14 | Slide 20 of 62 January 2006 STOP

Sterile Production Possible Issues l Poor design of the building l Poor design of the systems, e. g. water, HVAC l Flow of personnel l Flow of material l No validation or qualification l Old facilities not complying with current requirements Module 14 | Slide 21 of 62 January 2006 STOP

Sterile Production Possible Issues (continued) l Particulate levels/microorganisms l Differential pressures l Air changes l Temperature/humidity Module 14 | Slide 22 of 62 January 2006 STOP

Sterile Production Two categories of manufacturing operations l Terminally sterilized ä prepared, filled and sterilized l Aseptic preparation ä some or all stages 1. 3 Module 14 | Slide 23 of 62 January 2006 STOP

Sterile Production Manufacture of sterile preparations l Classification of clean areas l Manufacturing operation in an appropriate environment cleanliness level l Minimize risks – particulate and microbiological contamination – product and material l Meet classification "at rest" ä (That is "completed installation, equipment installed and operating, but no operating personnel present") 4. 1 Module 14 | Slide 24 of 62 January 2006 STOP

Sterile Production Manufacture of sterile preparations l For sterile pharmaceutical preparations: l Grade A ä local zone, high risk operations, e. g. filling, aseptic connections ä usually UDAF systems used l Grade B ä background environment to Grade A (in case of aseptic preparation and filling) l Grade C and Grade D 4. 1 ä Clean areas for less critical operations Module 14 | Slide 25 of 62 January 2006 STOP
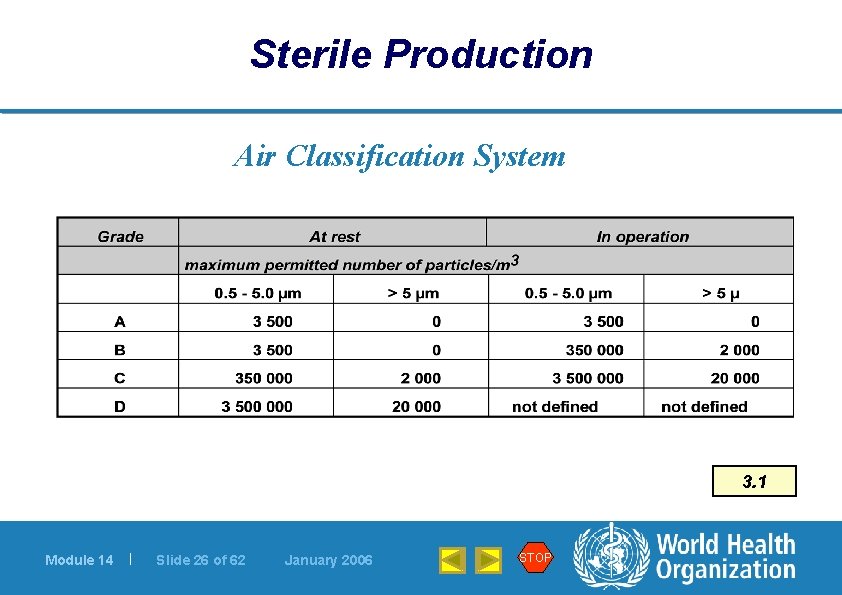
Sterile Production Air Classification System 3. 1 Module 14 | Slide 26 of 62 January 2006 STOP

Sterile Production Manufacture of sterile preparations l To reach Grade B, C and D, the number of air changes should be appropriate to the size of the area, number of personnel, equipment present l Minimum of 20 air changes per hour l Clean-up time about 15 – 20 minutes l Good airflow pattern in the area l HEPA filtered air l Suitable methods to determine particulate matter and micro ä e. g. EU, ISO, Japan, USA Module 14 | Slide 27 of 62 January 2006 4. 1 – 4. 2. STOP

Sterile Production Manufacture of sterile preparations l Control particulate during operation l Monitoring during operation l Alert and action limits for particulate and micro l Action taken when exceeded l Area grades should be proven (e. g. validation runs, media fills, environment, time limits - based on microbiological contamination/bioburden found) 4. 3 – 4. 5 Module 14 | Slide 28 of 62 January 2006 STOP
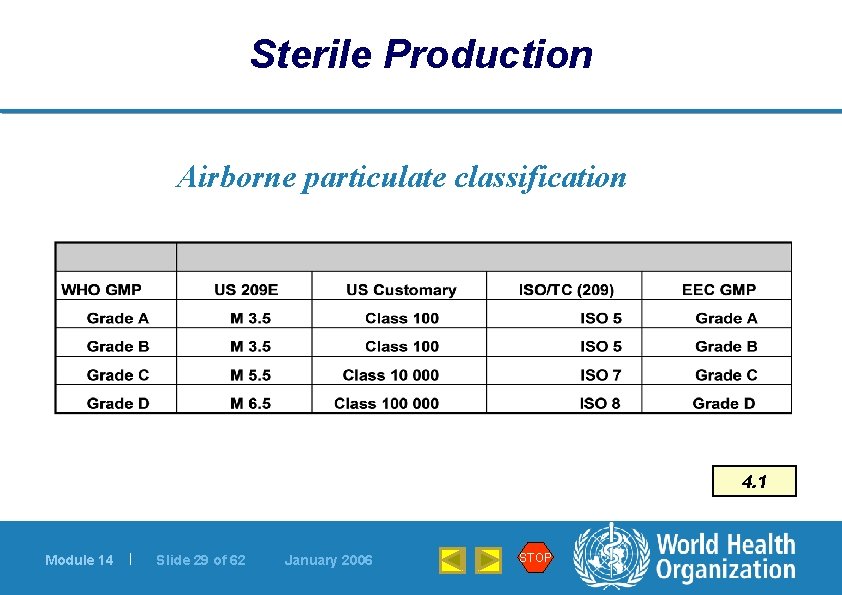
Sterile Production Airborne particulate classification 4. 1 Module 14 | Slide 29 of 62 January 2006 STOP

Sterile Production Processing l Minimize contamination - all stages including before sterilization and during processing l No unsuitable materials, e. g. live microbiological organisms l Minimize activities ä staff movement controlled and methodical ä avoid shedding of particles l Temperature and humidity comfortable l Containers and materials in the area 4. 15 – 4. 16, 4. 20 – 4. 21 Module 14 | Slide 30 of 62 January 2006 STOP

Sterile Production Processing l Validation – should not compromise the processes l Aseptic process validation: sterile media fill (“broth fills”) ä simulate actual operation – intimate as closely as possible ä simulate worst expected condition ä use appropriate medium/media ä sufficient number of units, e. g. equal to batch size (small batches) – acceptable limit – investigations ä revalidation: periodic and after change l New processing procedures validated ä revalidation after significant changes 4. 17, 4. 18, 4. 28 ä and regular intervals Module 14 | Slide 31 of 62 January 2006 STOP

Sterile Production Processing l Water sources, water treatment systems and treated water l Monitored regularly ä chemicals ä biological contamination ä endotoxin l Water specification l Records of results and action taken 4. 19 Module 14 | Slide 32 of 62 January 2006 STOP

Sterile Production Processing l Components, bulk product containers and equipment ä fibre generation ä no recontamination after final cleaning ä stage properly identified ä sterilized when used in aseptic areas l Used in clean areas, passed through double-ended sterilizers or use triple wrapping l Gas used to purge solution or blanket a product – passed through a sterilizing filter 4. 22 – 4. 23 Module 14 | Slide 33 of 62 January 2006 STOP

Sterile Production Processing l Bioburden monitored ä products: before sterilization ä working limits established ä solutions to be filtered before filling (especially LVP) ä pressure release outlets – hydrophobic microbiological air filters l Starting materials – microbiological contamination should be minimal l Monitored as per specification 4. 26, 5. 3 Module 14 | Slide 34 of 62 January 2006 STOP

Sterile Production Processing l Time intervals: components, bulk containers, equipment l Washing and drying and sterilization; and sterilization and use ä as short as possible ä time limit validated l Time intervals: product l Start of preparation of solution and sterilization (filtration) ä as short as possible ä maximum time set for each product 4. 23 - 4. 24 Module 14 | Slide 35 of 62 January 2006 STOP

Sterile Production Group session 2 l Considering the same factory as in the previous group session, discuss the process of sterilization l List all the items that will need to be sterilized (and indicate the choice of sterilization process) l What are the key features you should find in each sterilization situation? l Discuss the relevance, need, and the extent of qualification and validation required Module 14 | Slide 36 of 62 January 2006 STOP

Sterile Production Possible Issues l Autoclave - no pressure gauge l Autoclave - no temperature recorder l Autoclave - superheated steam l Clean room - pressure differentials l Exposure for settle plates l Interlocks turned off l Rusty Laminar airflow cabinets l HEPA filters not checked regularly Module 14 | Slide 37 of 62 January 2006 STOP

Sterile Production Sterilization l Methods of sterilization ä moist or dry heat ä irradiation (ionizing radiation) ä sterilizing gaseous agents (e. g. ethylene oxide) ä filtration with subsequent aseptic filling l Whenever possible: terminal sterilization by heat in their final container - method of choice 5. 1 – 5. 2 Module 14 | Slide 38 of 62 January 2006 STOP

Sterile Production Sterilization l Validation ä all sterilization processes ä special attention when non-pharmacopoeial methods are used ä non-aqueous or oily solutions l Before the method is adopted – its suitability and efficacy demonstrated with desired conditions ä all parts of the load ä each type of load ä physical measurements and biological indicators (where appropriate) ä verified at least annually and after change 5. 4 – 5. 5 ä records maintained Module 14 | Slide 39 of 62 January 2006 STOP

Sterile Production Sterilization l For effective sterilization l Whole of the material subjected to the treatment l Biological indicators l Additional method of monitoring l Storage and use, quality checked through positive control l Risk of contamination 5. 6 - 5. 7 Module 14 | Slide 40 of 62 January 2006 STOP

Sterile Production Sterilization l Differentiation between sterilized and not-yet-sterilized products l Each basket/tray or other carrier, properly labelled ä name of material ä batch number ä sterilization status l Use of autoclave tape l Sterilization records for each run – approved as part of the batch release procedure 5. 8 - 5. 9 Module 14 | Slide 41 of 62 January 2006 STOP

Sterile Production Terminal Sterilization by heat l Sterilization by moist heat l Sterilization by dry heat l Sterilization by radiation l Sterilization by gases and fumigants 6 Module 14 | Slide 42 of 62 January 2006 STOP

Sterile Production Terminal Sterilization by heat l Recording of each cycle, e. g. time and temperature chart ä temperature: validated coolest part ä check from second independent probe ä additional chemical or biological indicators l Heating phase: sufficient time for the whole load ä determined for each load l Cooling phase: after sterilization cycle ä precautions to prevent contamination 6. 2 – 6. 3 ä sterilized cooling fluid/gas Module 14 | Slide 43 of 62 January 2006 STOP

Sterile Production Terminal Sterilization by moist heat (heating in an autoclave) l Water-wettable materials only, and aqueous formulations l Temperature, time and pressure monitored l Temperature recorder independent of the controller l Independent temperature indicator l Drain – temperature recorded from this position l Regular leak test when vacuum is part of the cycle l Material allows for removal of air and penetration of steam l All parts of the load in contact with steam l Quality of the steam – no contamination 6. 4 – 6. 6 Module 14 | Slide 44 of 62 January 2006 STOP

Sterile Production Terminal Sterilization by dry heat l For non-aqueous liquids, dry powders l Air circulation in the chamber l Positive pressure in chamber to prevent entry of non-sterile air l HEPA filtered air supplied l When removing pyrogens, challenge tests ä validation (using endotoxins) 6. 7 Module 14 | Slide 45 of 62 January 2006 STOP

Sterile Production Terminal Sterilization by radiation l Suitable for heat-sensitive materials and products ä confirm suitability of method for material ä ultraviolet irradiation not acceptable l Contracting service – ensure validation status, responsibilities l Measurement of dose during procedure l Dosimeters independent of dose rate ä quantitative measurement ä number, location and calibration time-limit l Biological indicators only as additional control l Radiation sensitive colour discs 6. 8 – 6. 10 Module 14 | Slide 46 of 62 January 2006 STOP

Sterile Production Terminal Sterilization by radiation (2) l Information forms part of the batch record l Validation to cover effects of variation in density of packages l Handling procedures to prevent misidentification of irradiated and non-irradiated materials l Each package to have a radiation-sensitive indicator l Total radiation dose administered within a predetermined period of time 6. 10 – 6. 13 Module 14 | Slide 47 of 62 January 2006 STOP

Sterile Production Terminal Sterilization by gases and fumigants l Only when no other method is suitable l E. g. ethylene oxide, hydrogen peroxide vapour l Validation: also prove the gas has no damaging effect on product l Time and conditions for degassing (specified limits) - residue l Direct contact with microbial cells essential ä nature and quantity of packaging materials l Humidity and temperature equilibrium 6. 14 – 6. 20 Module 14 | Slide 48 of 62 January 2006 STOP

Sterile Production Terminal Sterilization l Monitoring of each cycle with biological indicators ä time, pressure ä temperature, humidity ä gas concentration Sterilization by gases and fumigants (2) l Post-sterilization storage – controlled manner ä ventilated conditions ä defined limit of residual gas ä validated process l Safety and toxicity issues Module 14 | Slide 49 of 62 January 2006 6. 21 STOP

Sterile Production Terminally sterilized products 4. 6 – 4. 7 Module 14 | Slide 50 of 62 January 2006 STOP

Sterile Production Terminally sterilized products 4. 8 – 4. 9 Module 14 | Slide 51 of 62 January 2006 STOP

Sterile Production Aseptic processing and sterilization by filtration Aseptic processing l Objective is to maintain the sterility of a product, assembled from sterile components l Operating conditions so as to prevent microbial contamination l What do you think are the aspects that require careful attention? 7. 1 – 7. 2 Module 14 | Slide 52 of 62 January 2006 STOP

Sterile Production Aseptic processing and sterilization by filtration Aseptic processing (2) l Careful attention to: l Environment l Personnel l Critical surfaces l Container/closure sterilization l Transfer procedures l Maximum holding period before filling Module 14 | Slide 53 of 62 January 2006 7. 3 STOP

Sterile Production Aseptic preparation 4. 10, 4. 11, 4. 14 Module 14 | Slide 54 of 62 January 2006 STOP

Sterile Production Aseptic preparation Module 14 | Slide 55 of 62 January 2006 4. 10 – 4. 13 STOP

Sterile Production Sterilization by Filtration l Through a sterile filter of 0, 22 µm or less, into previously sterilized containers ä remove bacteria and moulds ä not all viruses or mycoplasmas l Consider complementing with some degree of heat treatment l Double filter layer or second filtration advisable, just before filling - no fibre shedding or asbestos filters l Filter integrity testing immediately after use ä also before use if possible 7. 4 – 7. 7 Module 14 | Slide 56 of 62 January 2006 STOP

Sterile Production Sterilization by Filtration (2) l Validation to include ä time taken to filter a known volume ä pressure difference to be used across the filter l Significant differences to be noted and investigated, recorded in batch records l Integrity of gas and air vent filters checked after use, other filters at appropriate intervals 7. 7 Module 14 | Slide 57 of 62 January 2006 STOP

Sterile Production Sterilization by Filtration (3) l Same filter not used for more than one working day, unless validated l No filter interaction with product, e. g. ä removal of ingredients ä releasing substances into product 7. 8 – 7. 9 Module 14 | Slide 58 of 62 January 2006 STOP

Sterile Production Quality Control l Samples for sterility testing should be representative l From parts of the batch, most at risk ä aseptic filling - at beginning and end of batch filling, and after interruptions ä heat sterilized – coolest part of the load l Sterility of the batch ensured through validation ä validated sterilization cycle ä media fill l Sterility test procedure as per pharmacopoeia, and validated for each product l Batch processing records, sterility testing records, environmental records should be reviewed 2. 1 -2. 2 Module 14 | Slide 59 of 62 January 2006 STOP

Sterile Production Quality Control l Endotoxin testing for injectable products ä water for injection, intermediate and finished product l Always for large volume infusion solutions l Pharmacopoeia method, validated for each product l Failure of the test – investigation l Corrective action 2. 3 Module 14 | Slide 60 of 62 January 2006 STOP

Sterile Production Finishing of products l l l Containers closed by means of validated methods Samples checked for integrity Maintenance of vacuum (where applicable) checked Parenteral products inspected individually Visual inspection under suitable and controlled conditions ä illumination and background ä eyesight checks of operators ä allowed frequent breaks l Other methods ä validated, and equipment performance checked at intervals ä results recorded 11. 1 – 11. 3 Module 14 | Slide 61 of 62 January 2006 STOP

Sterile Production Group session 3 l Considering the same factory as in the previous group sessions, devise a plan for monitoring of the facility l List the parameters to be tested, tests to be used, acceptance criteria and frequency of testing Module 14 | Slide 62 of 62 January 2006 STOP
- Slides: 62