Colloids and their role in Pharmaceutical Industry Colloids



















- Slides: 23

Colloids and their role in Pharmaceutical Industry

Colloids A mixture in which very small particles of one substance are distributed evenly throughout another substance.

History This term was first used in 1862 to distinguish colloids from crystalloids. Colloids have been studied by scientists since the early 1800’s. to

Introduction The word “colloid” was derived from the Greek, “kolla” for glue. Colloid is short synonym for colloidal system. The heterogeneous biphasic system. Size of colloidal particle ranges from 1 nm to 100 nm.

Classification There are two criteria's for classification of colloids. 1. Based on the state of Aggregation of phase 2. Based on the interaction of phase

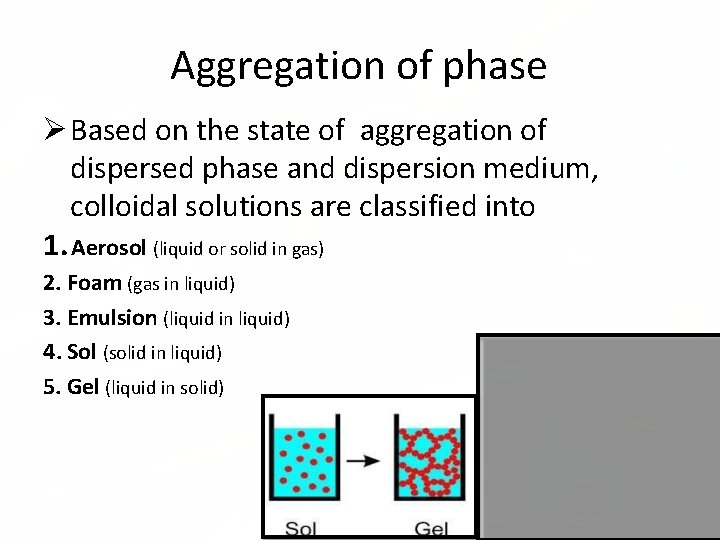
Aggregation of phase Based on the state of aggregation of dispersed phase and dispersion medium, colloidal solutions are classified into 1. Aerosol (liquid or solid in gas) 2. Foam (gas in liquid) 3. Emulsion (liquid in liquid) 4. Sol (solid in liquid) 5. Gel (liquid in solid)

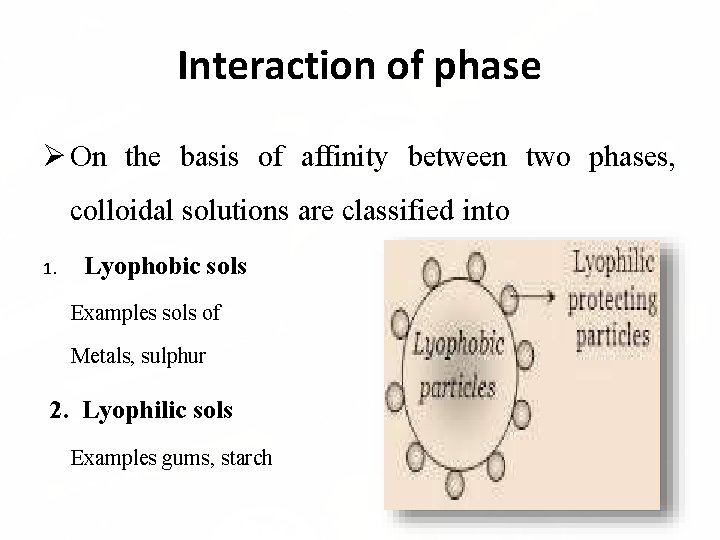
Interaction of phase On the basis of affinity between two phases, colloidal solutions are classified into 1. Lyophobic sols Examples sols of Metals, sulphur 2. Lyophilic sols Examples gums, starch

Preparation Two methods are generally used for the preparation of colloidal solutions. 1. By Condensation 2. By Dispersion

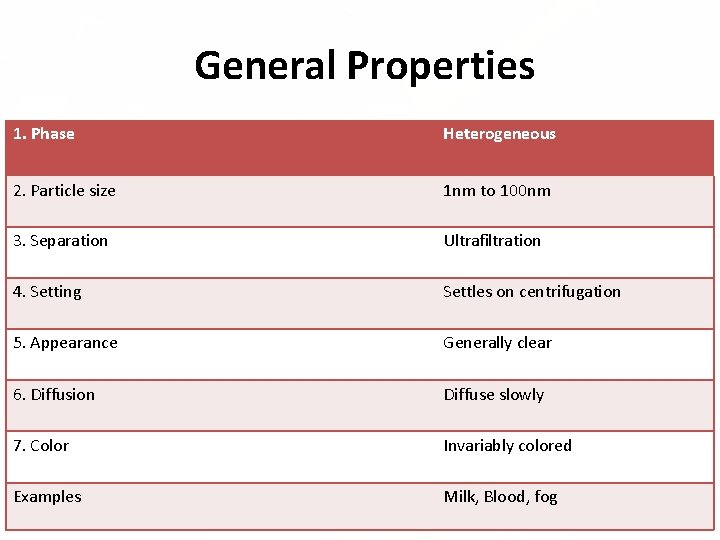
General Properties 1. Phase Heterogeneous 2. Particle size 1 nm to 100 nm 3. Separation Ultrafiltration 4. Setting Settles on centrifugation 5. Appearance Generally clear 6. Diffusion Diffuse slowly 7. Color Invariably colored Examples Milk, Blood, fog

Kinetic properties Brownian motion Rapid, random, zigzag movement of colloidal particles through dispersion medium. This movement is due to bombardment of colloidal particles by the molecules of dispersion medium.

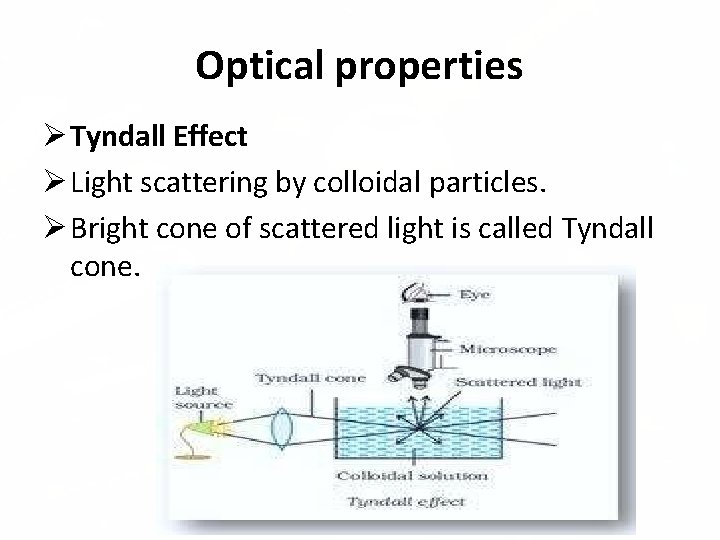
Optical properties Tyndall Effect Light scattering by colloidal particles. Bright cone of scattered light is called Tyndall cone.

Electrical properties Charges on colloidal particles: Colloidal particles always carry some charge otherwise the colloidal solution would be unstable. It may be positively or negatively charged. Due to acidic and basic groups

Purification The colloidal solutions prepared by any method contain different impurities. They can be separated by 1. Dialysis 2. Ultrafiltration

Role in pharmaceutics Therapy Colloidal system are used as therapeutic agents in different areas. Colloidal medicines are more effective and are easily absorbed in our system. Example , penicillin and streptomycin.

Eye lotions There are various eye lotions that are prepared by colloidal solutions. Argyrol and protargyrol

Colloidal sulphur It is used as disinfectants. It is a homeopathic remedy used to treat skin conditions such as psoriasis, eczema etc.

Colloidal copper It is used in treatment of cancer. It is used as a remedy for burns, arthritis, parasites, viral and bacterial infections.

Colloidal Gold It is diagnostic agent for paresis. It is used as mineral supplement to augment the body’s immune system.

Colloidal silver It is an unrivalled anti-biotic. It destroy all types of virus, bad fungi and bacteria. It promotes healing.

Colloidal electrolytes Increase solubility, stability and taste of compounds Used as additives in drugs E. g. Quaternary Ammonium salts

colloidal Silica Prevents flocculation. Stability to drugs. e. g. silicon dioxide

Natural colloids Plasma protein. Gelatin coating over tablets and granules. For protection.
