INCOMPATIBILITIES N SANTHI PRIYA ASSISTANT PROFESSOR DEPARTMENT OF

INCOMPATIBILITIES N. SANTHI PRIYA ASSISTANT PROFESSOR DEPARTMENT OF PHARMACEUTICS CHALAPATHI INSTITUTE OF PHARMACEUTICAL SCIENCES

DEFINITION • The undesired change taking place in the physical , chemical or therapeutic properties of the medicament of a prescription are mixed together , is termed as INCOMPATIBILITY • While handling a prescription containing combinations many a times problems asscociated with therapeutic , physical and chemical properties of the drugs arise

UNINTENTIONAL INCOMPATIBILITY • TOLERATED – interaction is minimized but composition of the prescription is not altered • ADJUSTED – interaction is prevented by addition or substitution of constituents without affecting the medicinal action of the preparation INTENTIONAL INCOMPATIBILITY The prescriber wants that the prescription , be dispensed as it is

3 CLASSES • THERAPEUTIC INCOMPATIBILITY • PHYSICAL INCOMPATIBILITY • CHEMICAL INCOMPATIBILITY

THERAPEUTIC INCOMPATIBILITY • When the response to one or more drugs in the patient is of a nature or intensity different from that intended , it is known as therapeutic incompatibility REASONS 1. Dosage errors 2. wrong dose or dosage form 3. Contraindicated drug 4. Synergistic or antagonistic drugs 5. Drug interactions

DOSAGE ERROR • Dispensing of an overdose of prescription • Any experienced pharmacist should be able to detect such an error while checking the prescription Ex: ü Atropine sulphate-0. 06 gm ü Phenobarbital-0. 015 gm ü Asprin-0. 300 gm If the quantity of atropine sulphate for a single capsule is more than the recommended maximum dose, The physician should be contacted EX-2 Codeine phosphare-0. 5 gm ONE POWDER TO BE TAKEN AT BED TIME

Wrong dose or dosage form • Possibility of dispensing the wrong drug because of confusingly similar names • Ex: 1. Prednisone and prednisolone 2. Protamine and protamide 3. Digoxin and disgitoxin

Contraindicated drugs • Certain drugs are contraindicated in a particular disease or when a particular patient is allergic to it 1. Corticosteroids-contraindicated to patients having peptic ulcer 2. Morphine, barbiturates-dangerous in severe asthma 3. Pencillin and sulphur drugs - allergic to patients 4. Vasoconstrictors-should not be given to hypertensive patients

Example-contraindication Sulphadiazine-0. 25 gm Sulpharmerazine-0. 25 gm Ammonium chloride -0. 5 gm Ammonium chloride being a urinary acidifier , would cause deposition of sulphonamide crystals in kidney Hence this contraindicated combination should be avoided

Synergistic or antagonistic drugs Synergistics: • Penicillin derivatives+probenicid • Aspirin +paracetamol Antagonistics: Amphetamine X barbiturate Stimulants X sedatives Cholinergic drugs X anticholinergic drugs Purgatives X antidiarrhoeals Antacids X antibiotics

CHEMICAL INCOMPATIBILITY • Chemical changes which occour due to the interaction of the prescribed substances leading to the formation of a harmful or dangerous product • They often occour by 1. OXIDATION – REDUCTION 2. HYDROLYSIS 3. COMBINATION OF BOTH

Oxidation – reduction 1. Oxidation -Loss of electrons 2. Reduction - Gain of electrons Oxidization of prescriptions • On exposure to air • Higher storage conditions • Light • Over dilution • Incorrect p. H adjustments • Presence of catlysis

• Trace metal ion catalysis - complexing agents like disodium edetate , sodium calcium edetate • Autooxidation of fats and oils , phenols, aldehydes, vitamins - antioxidants like propyl gallate, thymol, BHT, BHA, hydroquinone • Light Reduces siver, mercury, gold salts to metallic form
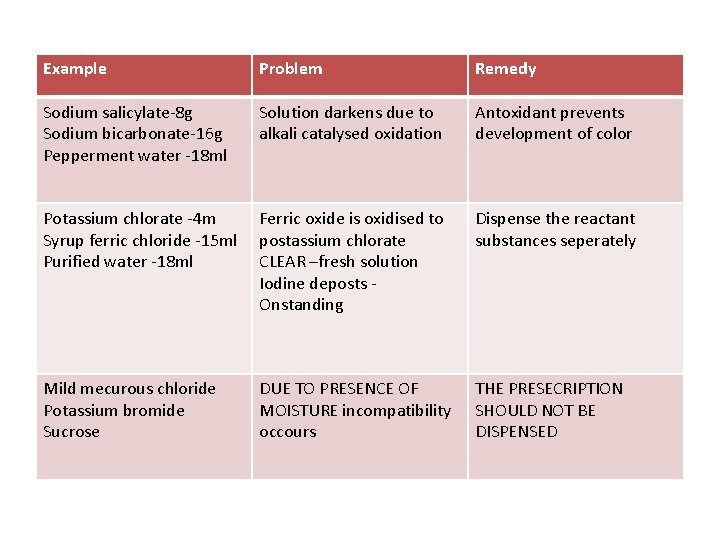
Example Problem Remedy Sodium salicylate-8 g Sodium bicarbonate-16 g Pepperment water -18 ml Solution darkens due to alkali catalysed oxidation Antoxidant prevents development of color Potassium chlorate -4 m Syrup ferric chloride -15 ml Purified water -18 ml Ferric oxide is oxidised to postassium chlorate CLEAR –fresh solution Iodine deposts Onstanding Dispense the reactant substances seperately Mild mecurous chloride Potassium bromide Sucrose DUE TO PRESENCE OF MOISTURE incompatibility occours THE PRESECRIPTION SHOULD NOT BE DISPENSED

Acid base reactions I. Precipitation II. Gas formation III. Color development or color change
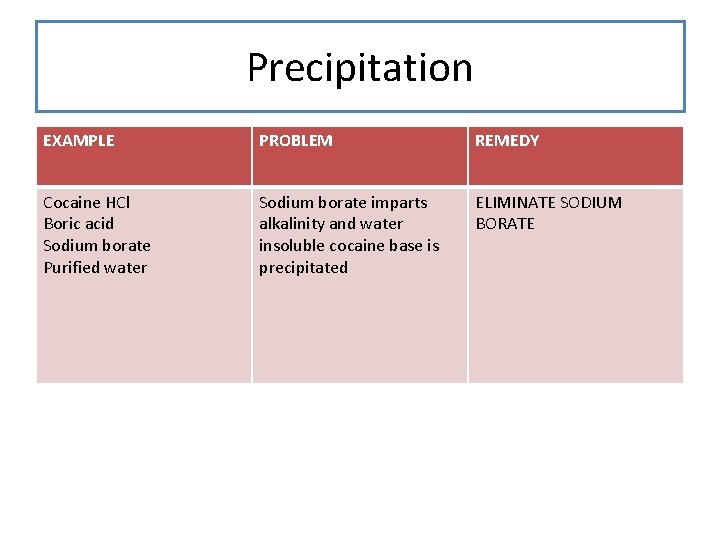
Precipitation EXAMPLE PROBLEM REMEDY Cocaine HCl Boric acid Sodium borate Purified water Sodium borate imparts alkalinity and water insoluble cocaine base is precipitated ELIMINATE SODIUM BORATE
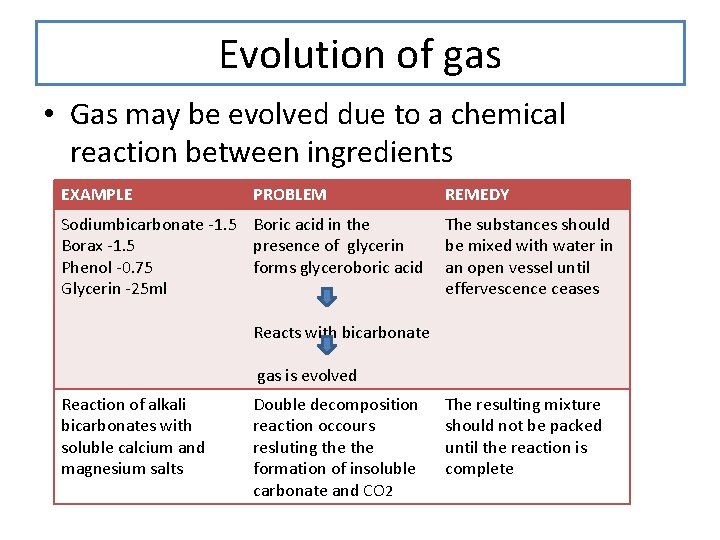
Evolution of gas • Gas may be evolved due to a chemical reaction between ingredients EXAMPLE PROBLEM Sodiumbicarbonate -1. 5 Boric acid in the Borax -1. 5 presence of glycerin Phenol -0. 75 forms glyceroboric acid Glycerin -25 ml REMEDY The substances should be mixed with water in an open vessel until effervescence ceases Reacts with bicarbonate gas is evolved Reaction of alkali bicarbonates with soluble calcium and magnesium salts Double decomposition reaction occours resluting the formation of insoluble carbonate and CO 2 The resulting mixture should not be packed until the reaction is complete

Breaking of structure of disperse systems • Gelling agents like carbomers , sodium cmc lose viscoisty by change in p. H outside an optimum range • Carbomer p. H< 5 and p. H> 10 liquify • Alginic acid at p. H< 3 precipitates sodium alginate to form sodium derivative

Change in or development of color • Dyes – color -IONIZATION DEPENDING ON p. H DYE CHANGE WITH p. H GENTIAN VIOLET ( ANTISEPTIC) IS BASIC PURPLE IN COLOR ØIn acidic Green to Yellow PHENOPHTHALEIN ( LAXATIVE ) ØIN ACIDIC COLORLESS ØIN ALKALINE RED

HYDROLYSIS DRUGS PRONE TO HYDROLYSIS : Procaine Sulphonamides Chlorothiazide Barbituric acid Aspirin Alkaloids Penicillin ADDITIVES PRONE TO HYDROLYSIS : GELATIN- SUCROSE FLAVORING OILS CHLOROBUTANOL Hydrolysis promoters : • Heat catalysts • Hydrogen ions • Hydroxyl ions Ex: ESTERS, AMIDES, METALS like Zn, Fe
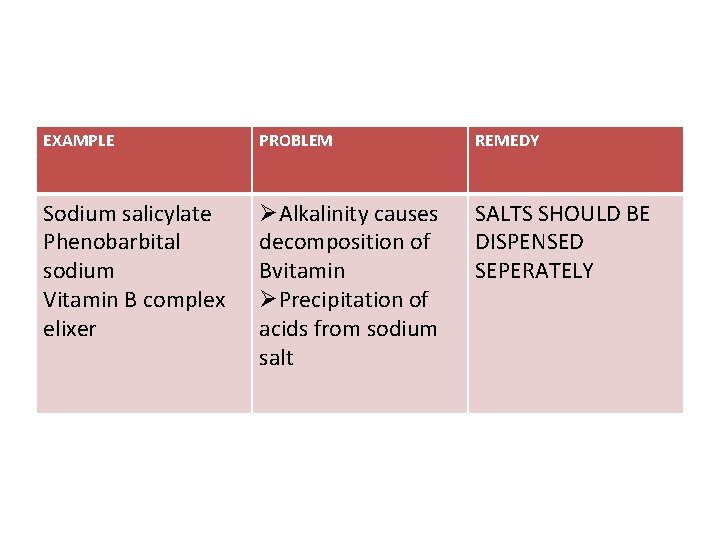
EXAMPLE PROBLEM REMEDY Sodium salicylate Phenobarbital sodium Vitamin B complex elixer ØAlkalinity causes decomposition of Bvitamin ØPrecipitation of acids from sodium salt SALTS SHOULD BE DISPENSED SEPERATELY
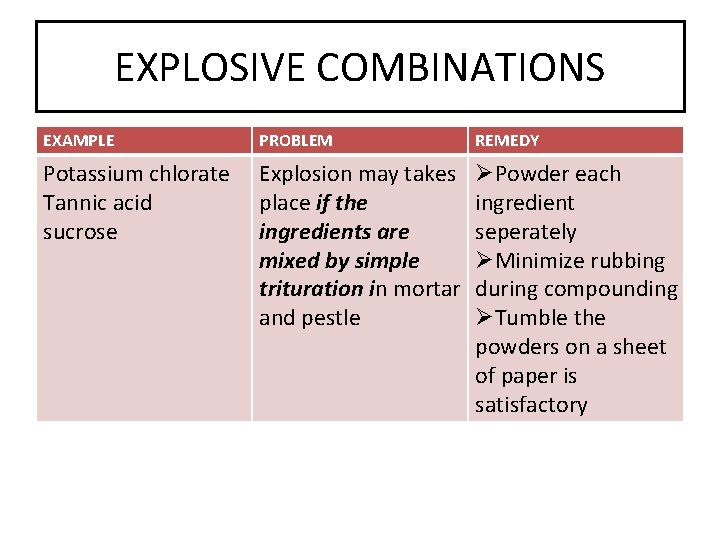
EXPLOSIVE COMBINATIONS EXAMPLE PROBLEM REMEDY Potassium chlorate Tannic acid sucrose Explosion may takes place if the ingredients are mixed by simple trituration in mortar and pestle ØPowder each ingredient seperately ØMinimize rubbing during compounding ØTumble the powders on a sheet of paper is satisfactory

Racemization • Conversion of optically active form of a drug substance to an optically inactive form without a change in chemical constitution associated with reduction in pharmacological activity Examples • Adrenaline • Ephedrine • Norephedrine • HYOSCYMINE – racemizes to atropine

Complexation • The medicaments or excipients get bound to or trapped inside the macromolecules • The complexes are too large to penetrate cell membranes • Reversible complexation is useful • POVIDONE – IODINE Complex : useful to reduce the irritancy and stability of the drug

Other changes 1. 2. 3. 4. 5. 6. 7. Cementation Polymerization Development of heat Lowering of temperature Double decomposition Substitution Addition

PHYSICAL INCOMPATIBILITIES • They are easier to PREDICT, DETECT AND CORRECT by applying proper technique , mixing, selection of additives etc • They are manifested by 1. Non-uniform 2. Unsighty 3. Unpalatable products

CLASSIFICATION • • • INSOLUBILITY PRECIPITATION SEPERATION OF IMMISCIBLE LIQUIDS LIQUEFACTION OF SOLID PHYSICAL COMPLEXATION MISCELLANEOUS

INSOLUBILITY • When a drug is insoluble in a solvent , it may create in proper administration of the dose • Ex: • Solubility enhancers • Alteration in the volume • Settling - suspending agents • Poor wettability – levigating agents

Precipitation of the drug • A Solubilized substance may precipitate from its solution if a non – solvent for the substance is added to the solution • Poor solvent – order of mixing • Precipitation – suspension • Cap locking – co-solvents • Grainy semisolids – non uniform cooling EX: 1. HYDROCOLLOIDS +ALC addition --> PPTS 2. Aromatic waters + electrolytes addition -> VOLATILE OILS precipitates 3. ELECTROLYTES + in SOAP EMULSIONS -> cracking

Liquefaction Eutectic substance (LOW MELTING SOLIDS )such as camphor, menthol, thymol if mixed together undergo liquefaction ex: • Eutectic powders • Insufflations • Siedliz powder TO OVERCOME: 1. Mixtures dispensed separately 2. Triturate separately with adsorbent to give free flowing product 3. If already liquefaction occurred -> adsorbed on powder -> filled in capsules -> dispensed

Example – Liquefaction Ø Ø Menthol – 2 g Camphor -2 g Ammonium chloride – 20 g Make a powder 1. If the ingredients are triturated together , they shall liquefy due to the formation of eutectic mixture 2. Powder form is obtained by mixing them separately in suitable adsorbent like Mg Co 3 3. 3 mixtures dispensed separately to obtain a powder

Adsorption and complexation Drug + excipient Physical complexes 1. Alter appearance 2. Alter medicament activity ex : Adsorption of cyanocobalamin by talc as tablet lubricant decrease the adsorption of this vitamin in GIT Adsorption of preservatives like parabens onto drugs lead to loss of antimicrobial activity

INORGANIC INCOMPATIBILITIES 1. Incompatibilities of metals and their salts 2. Incompatibilities of acids 3. Incompatibilities of alkalies ORGANIC INCOMPATIBILITIES BY 1. Purine bases 2. Alkaloids 3. Pyrazolone derivatives 4. Amino acids 5. Quaternary ammonium compounds 6. Glycosides 7. Anesthetics

INCOMPATIBILITIES OF THE METALS AND THEIR SALTS GROUP 1 A ( Li, Na, K, Rb, Cs, Fr) 1. sodium salicylate + potassium bicarbonate – sodium bicarbonate ppt , darkening due to salicylates 2. K+ Tartaric acids- ppt , Sulfurated potash incompatible with acids and liberate H 2 S 3. Lithium carbonate – in alkaline or neutral solutions GROUP 1 B (CU, Ag, Au) 1. 2. 3. 4. Copper salts ppts tannic acid , arsenates , alkalis etc Cupric salts – iodides, acetates Silver salts decomposed by light and form metallic silver Ago – powerful oxidizing agent and causes explosion when triturated with reducing agents 5. Gold salts +weak reducing agents - gold metal

INCOMPATIBILITIES OF THE METALS AND THEIR SALTS • Group II- A ( Be, Mg, Ca, Ba, Ra) Mg 1. 2. 3. 4. Mg. trisilicate, adsorbs alkaloids , dyes, toxins Mg. O – adsorbs moisture, co 2 when exposed to air Mg. sulfate – water soluble salt , but ppts on addition on soluble phosphates, carbonates , salicylates Mg. salts react with bromide reacts with ammonia –( ANTACIDS X Bromide preparations) Ca. Incompatible with hydroxides , citrates , carbonates , oxalates , tartrates, phosphates , forms precipitates except in acids

INCOMPATIBILITIES OF THE METALS AND THEIR SALTS Group II – B: ( Zn, Cd , Hg) Zinc : 1. Soluble zinc salts incompatible with alkali hydroxides, carbonates, phosphates and converts into insoluble zinc compounds 2. They also precipitates fatty acids , acacia , proteins , tannins Mercury : Mercury salts reduced to free metals by light , moisture , trituration Mercurous salts 1. 2. 3. 4. oxidizing agents Mercuric salts Hgcl 2 + 4 KI Hg + K 2 Hg. I 4 +2 KCl Mercurous salts ppts alkaloids proteins tannins organic acids Yellow Hg. O gets pptd from alkali hydroxide solutions Combined use of ammoniated mercury + salicylic acid in ointments causes severe irritation due to the formation of mercuric salicylate Calomel decomposes into mercuric chloride and free mercury when exposed to sunlight or triturated excessively

INCOMPATIBILITIES OF THE METALS AND THEIR SALTS Group III –A ( B, Al , Ga, In Ti ) 1. Aluminium solutions are acidic 2. Incompatible to weak acids , borax 3. Produce effervescence with carbonates 4. Complexes the penicillin , sulphonamides when mixed with organic salts 5. Alum incompatible with soluble sulfates

INCOMPATIBILITIES OF THE METALS AND THEIR SALTS Group VII – B( Mn , Tc , Re) 1. Mn hypophosphite , Mn salts ppt out as Mn. hydroxide 2. These ppts darkens on standing due to absorption of oxygen 3. Mn. O 2 is a strong oxidizing agent and causes explosion if triturated with reducing agents like phosphides , hypophosphites and organic substances

INCOMPATIBILITIES OF THE METALS AND THEIR SALTS Group VIII ( Fe, Ru , Os , Co , Rh, Ir , Ni , Pd, Pt ) Ferrous salts in aqueous solutions are unstable They gets oxidized to ferric state by precipitation of a basic ferric salt 4 Fe. SO 4 + 2 H 2 O+ O 2 4 Fe(OH)SO 4 Oxidation reaction can be prevented by adding iodides and hypophosphites Incompatible with alkali hydroxides – white ppt of Fe(OH)2 – green – brown due to oxidation Ferric salts gets reduced to ferric state by iodides and hypophosphites 2 Fe 3+ + 2 I 2 Fe 2+ + I 2 Precipitation can be prevented by addition of organic hydroxy acids or their salts such as tartrates, citrates etc or polyhydric alcohols such as glycerin and sugars

INCOMPATIBILITIES OF NON METALS • Most of the non-metallic elements form acids and exhibit acidic incompatibilities • Other non-metal incompatibilities include – ü Carbon ü Sulphur ü Iodine

INCOMPATIBILITIES OF NON METALS Carbon – • Charcoal gets easily oxidized , - so don't triturate with oxidizing agents • High adsorptive characteristics – so dispensed separately if potent drugs are present since they may get inactivated SULPHUR – Sulphur gets readily oxidized in presence of water On heating with fixed hydroxides – solubilizes forming metal thiosulphates 14 S + 12 KOH K 2 S 3+2 K 2 S 3 O 3+2 K 2 S+K 2 S 5+6 H 2 O Iodine 1. Iodine acts as an oxidizing agents in alkaline solution 2. Incompatible with reducing agents like hypophosphite's , arsenites I 2+H 3 As. O 3+H 2 O 3 HI+H 3 As. O 4 3. Iodine + turpentine oil - gives explosive combination 4. Iodine + potassium iodide – precipitates alkaloids 5. Iodine + oxidizing agents – gives iodates

INCOMPATIBILITIES OF ACIDS • ALL ACIDS EXHIBIT INCOMPATIBILITIES ü Strong acids ü Weak acids ü Oxidizing acids ü Reducing acids ü Acidic preparations

INCOMPATIBILITIES OF ACIDS • Strong acids highly ionize in aq. solutions • Sulphuric acid chars many organic substances , forming carbon , charring of sugar C 6 H 1206 6 C + 6 H 20 • Hot, concentrated sulphuric acid acts as a strong oxidizing agent for metals, non-metals Hg+ 2 H 2 SO 4 Hg. SO 4 + SO 2 + 2 H 20 • Orthophosphoric acid reacts with salts of lead and silver to give precipitates

INCOMPATIBILITIES OF ACIDS • Weak acids slightly ionize in aq. solutions • All weak acids reacts with alkali carbonates to give CO 2 Oxidising acids -incompatible with org. sub and give explosive combinations • Reducing acids - incompatible with oxidising agents and give explosive combinations ü Ergot fluid extracts ü Nux vomica fluid extracts ü Compound pepsin elixer ü Ammonium acetate solution ü Ferric chloride solution ü Citric acid syrup D O N T C O M B I N E Carbonates Bicarbonates Chorates Hypochlorites nitrates

INCOMPATIBILITIES OF ALKALIES • Metallic alkaloids precipitate due to solubility changes and salt formation 1. Strong alkali hydroxides – soluble in water 2. Weak alkali hydroxides – insoluble in water 3. Amphoteric alkali hydroxides-soluble in water by addition with acid ü Fluid extract of senega ü Ammonium valerate elixer ü Calcium hydroxide solution ü Aromatic spirit of ammonia INCOMPATIBL E WITH ALKALIES 1. Neutralizes acids 2. Precipitates alkaloids 3. Precipitates heavy metals from salt solutions

ORGANIC INCOMPATIBILITIES Some specific incompatibilities include : ü Purine bases (xanthines) ü Alkaloids ü Pyrazolone derivatives ü Amino acids ü Quaternary ammonium compounds ü Glycosides ü Anaesthetics

Xanthines • Weak basic properties • Scarcely available in the form of salts with acids • Salts so formed are weak mixtures

Alkaloids ALKALOIDAL COMPONENT INCOMPATIBILITY Alkalies and alkaline reactants like borax, potassium citrate Free alkaloids precipitates Tannins and its preparations Insoluble tannates Picric acid Free alkaloid precipitates cocaine , colchicine, atropine Hydrolysed by acids and alkalies
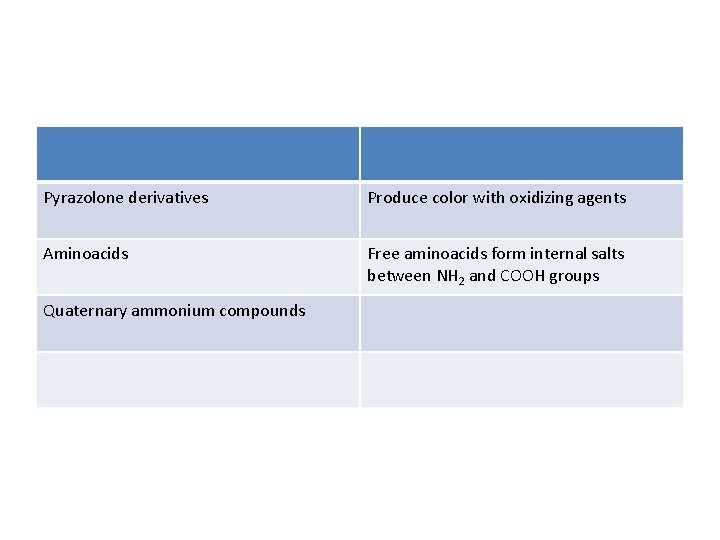
Pyrazolone derivatives Produce color with oxidizing agents Aminoacids Free aminoacids form internal salts between NH 2 and COOH groups Quaternary ammonium compounds
- Slides: 49