Presentation of Capabilities V 050917 WOODFIELD PHARMACEUTICAL LLC

Presentation of Capabilities V 050917 WOODFIELD PHARMACEUTICAL, LLC Crafting Contract Manufacturing Solutions

Topics • Overview And History • Pharmaceutical Development • Commercial Manufacturing Services • Regulatory Support • Primary And Secondary Packaging


Overview and History • Thirty (30) Years As A Respected CMO • Founded In 1988 As A Privately Owned Company G&S Enterprises Doing Business As Great Southern Laboratories (GSL) • GSL Assets Were Purchased In July 2012 By Pernix Manufacturing, LLC (PML), A Wholly Owned Subsidiary Of Pernix Therapeutics • Pernix Manufacturing, LLC Was Purchased By Woodfield Pharmaceutical, LLC In April 2014 • Customized Solutions With A Range Of Machinery And Equipment, Scientific Innovation And Pharmaceutical Expertise

Assets and Values • Crafting CMO Solutions • Experienced Technical Team • c. GMP Compliant Systems • Competitive Pricing • Expertise Interacting With U. S. Regulatory Agencies • DEA Licensed Facility (Schedules I-V) • Turn-Key Operations • Regulatory Support (IND, NDA, ANDA, CBE Submissions) • Drug Establishment License (Manufacturing and Packaging)

Capabilities • Customized Development, Manufacture and Packaging For Oral Solutions, Liquid Solutions, Suspensions And Semi-Solids • Research And Development, Materials Procurement, Qualification, Formulation, Process Development, Optimization, Analytical Method Development, Validation • Development And Support Of INDs, NDAs And ANDAs • Production And Packaging Of Over-The Counter, Prescription And Specialty Products Including Cosmetics, Nutritional Supplements And Medical Foods • Pre-Formulation, Formulation Development, Stability Studies, Method Development • Pre-Clinical, Phase I Clinical, Late-Stage Clinical Trial Materials • Registration Batches, Commercial Production, Stability Studies • Registered Medical Device Manufacturer (Topical)

Facilities Manufacturing, Packaging, Laboratory and Warehouse Facilities Located in Houston, TX • 48, 600 Square Feet Of Warehouse Space • 25, 000 Square Feet Of Manufacturing And Packaging Areas • 10, 000 Square Feet Of QC Analytical Chemistry And Microbiology Laboratory Areas • 5, 000 Square Feet Of Office Space • Vault, Cage And Storage Areas For Controlled Substances • Stability Chambers • Flexible Production Capacities

Snigdha Chennamaneni Research and Development Scientist

Pharmaceutical Development • Scope Of Services • Process Optimization • Pre-Formulation • Product Development • Formulation Development • Scale-Up • Validation • Site/Technology Transfer • Analytical Services • Product Application Support

Scope of Services End-to-End Solutions From Development To Commercialization • Oral Solutions, Liquid Solutions, Suspensions And Semi-Solids • Formulation Development • Packaging • Controlled Substances • Stability Studies (Long Term, Intermediate And Accelerated • Analytical And Microbiological Method Development And Validation Conditions) • Manufacturing • Regulatory Submissions
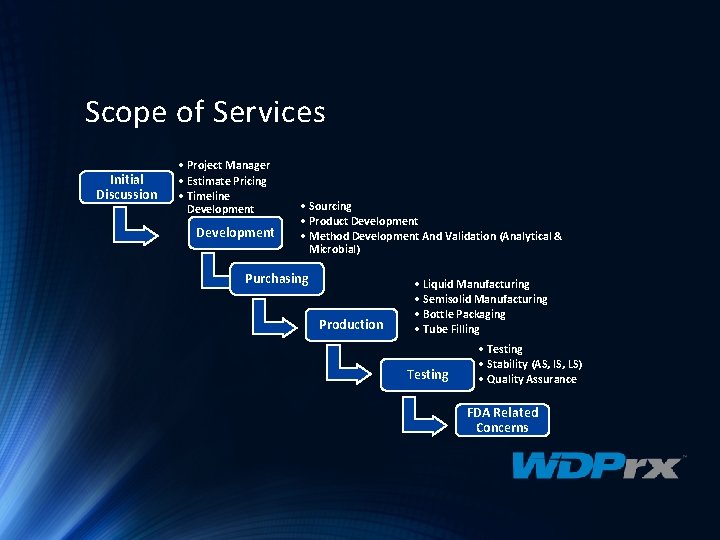
Scope of Services Initial Discussion • Project Manager • Estimate Pricing • Timeline Development • Sourcing • Product Development • Method Development And Validation (Analytical & Microbial) Purchasing Production • Liquid Manufacturing • Semisolid Manufacturing • Bottle Packaging • Tube Filling Testing • Testing • Stability (AS, IS, LS) • Quality Assurance FDA Related Concerns

Pre-Formulation Optimizing Physico-Chemical Properties Of APIs For Potency And Stability • Selection Of Excipients And Review Against IID • Deformulation Of Innovator/RLD • Stability Studies: Solution Stability, Solid State Stability, ICH Photostability, Thermal Cycle Stability • Inherent Properties: p. Ka, Log p/Log D, Intrinsic Dissolution • Excipient Compatiblity

Analytical Services Validate API Suitability For Product Requirements • Assess Prototype Formulations • Analytical Method Development And Validation • Determine Product Stability – Stability Studies • Discover Batch-To-Batch Variability • Verification of USP Methods • Transfer Of Analytical Procedures • Involvement From Quality Control Team and R&D Analytical Team

Process Optimization Reduce Costs By Creating Efficiencies In Development and Manufacture • Review Product Processes and Procedures • Inter-Department Coordination • Eliminate Process Steps • Improve Yields • Produce Quality Product • Confirm Product Meets Specifications • Cleaning Validation, Specifications And Batch Records

Product Development Key Benefits • Quality Control: The Highest Level Of Production Quality • c. GMP Compliant Systems • DEA Licensed Facility (Schedules I-V) • Cost Of Services: Delivering Highest Quality Within Budget • Production Capacity: Scalable Capacity Speeds Time-To-Market • Flexible Capabilities From Low-Volume To High-Volume Production
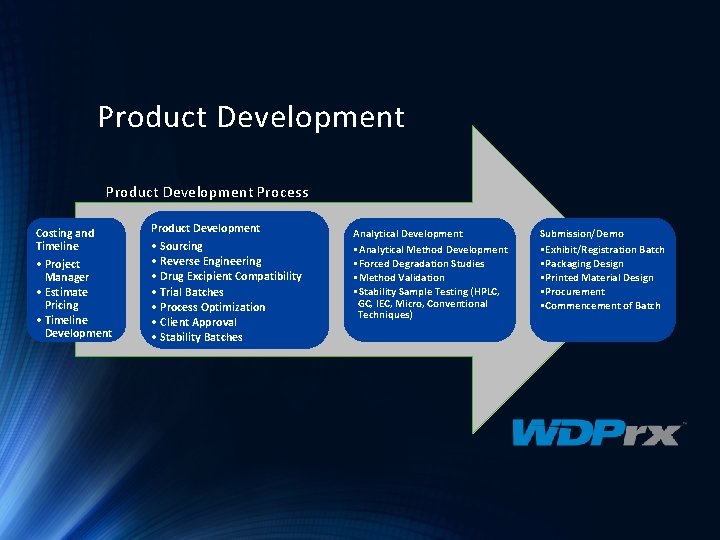
Product Development Process Costing and Timeline • Project Manager • Estimate Pricing • Timeline Development Product Development • Sourcing • Reverse Engineering • Drug Excipient Compatibility • Trial Batches • Process Optimization • Client Approval • Stability Batches Analytical Development • Analytical Method Development • Forced Degradation Studies • Method Validation • Stability Sample Testing (HPLC, GC, IEC, Micro, Conventional Techniques) Submission/Demo • Exhibit/Registration Batch • Packaging Design • Printed Material Design • Procurement • Commencement of Batch

Scale-Up Best Practices To Increase Production For Sales and Distribution Requirements • Assure Adequate Supply Of API And Raw Materials • Evaluate Process Step Changes From Small-Batch To Full Production • Compare Small-Scale Sample Data To Achieve Key Production Insights • Develop Process Ranges And Monitor With Pre-Approved Protocol • Conform Procedures To SUPAC (FDA) • Adhere To Regulatory Requirements

Site/Technology Transfer Efficient Transition To New Manufacturing Facility • Assess Original Manufacturer Capabilities From Raw Materials Through Production • Compliance With SUPAC Guidelines • Develop Transfer Protocols: Equipment Differences, Operating Ranges • Complete Testing And Validation • Regulatory Filing Support • Packaging Line Trials • Health And Safety Review

Product Application Support Streamlining Process For Regulatory Documentation Submission And Approval • Preparation And Submission (e. CTD Format) Or Submission-Ready Documentation • Authorized U. S. Agent • Lifecycle Management Of Recently Marketed Products • CMC (Chemistry, Manufacturing, Control) • Labeling • Adhere To NDA And ANDA And OTC Regulatory Requirements


Commercial Manufacturing A Wide Variety Of Non-Sterile Liquid Options • Oral Solutions, Liquid Solutions, Suspensions And Semi-Solids • Flexible Batch Sizes • Development, Manufacturing, Packaging And Testing • Manufacturing Suites With Segregation And Engineering Controls • Controlled Substances (CI-CV) • Clinical Trial Supplies • Packaging and Labeling

Cheryl Towns Regulatory Manager

Regulatory Support All Phases Of Clinical Development • Preparation And Packaging For INDs, NDAs, ANDAs, OTCs • Registration And Re-Registration Of Market Authorizations (MAs) • Comprehensive Risk Evaluation And Mitigation (REMS) Strategies Assessment • Clinical Trial Supplies
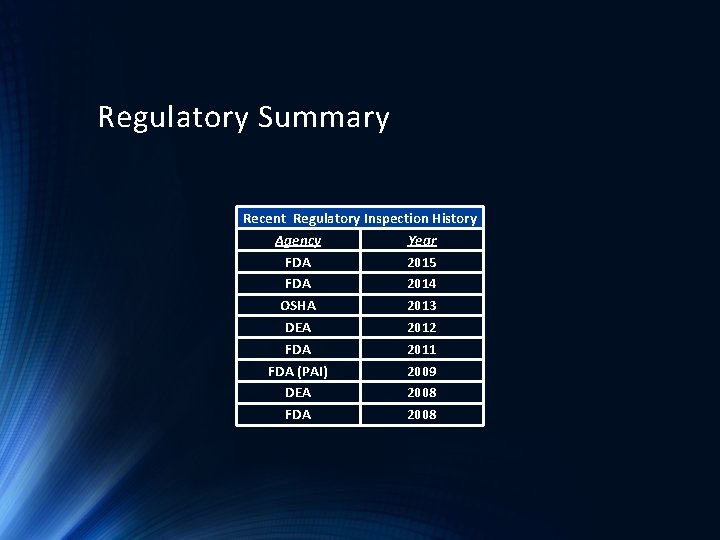
Regulatory Summary Recent Regulatory Inspection History Agency Year FDA 2015 FDA 2014 OSHA 2013 DEA 2012 FDA 2011 FDA (PAI) 2009 DEA 2008 FDA 2008
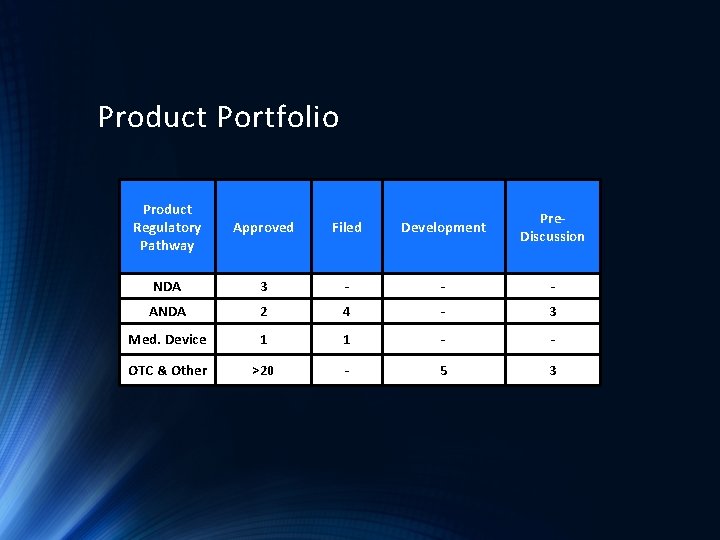
Product Portfolio Product Regulatory Pathway Approved Filed Development Pre. Discussion NDA 3 - - - ANDA 2 4 - 3 Med. Device 1 1 - - OTC & Other >20 - 5 3

Clinical Trial Supplies Custom Solutions For Unique Clinical Study Protocols • Investigational Materials Dispensed To Patients On Time And Correctly • Pre-Clinical, Phase I Clinical, Late-Stage Clinical Trial Materials • Additional Quality Requirements Unique To Clinical Trial Supply Environment: c. GMP, GCP, GLP, CFRs, ICH • Written Quality Agreement To Comply With Client Protocols • Stability Studies: Chemical And Physical Properties, Excipient Compatibility, Manufacturing Process, Interaction With Packaging • Multi-Product Kitting, Labeling And Fulfillment


Primary And Secondary Packaging Wide Range Of Filling Options Offer Dosage and Distribution Flexibility • Clinical Trial Customized • Bottle Filling: Plastic And Glass Bottles And Jars, Nasal Spray Solutions Devices, Metal And Laminate • Controlled Substances (CII-CV) Tubes • Gravity-Fed Lines With No Mechanical Intervention • Stainless Steel Piping • Manual, Semi-Automated And Fully Automated Fill • Tamper Evidence Applications • Secondary Packaging Options Include Carton, Tray, Shrink Wrapping

DSCSA Serialization Support Services • DSCSA Compliant Packaging and Labeling For Solids, Semi-Solids and Liquids • Services For Non-Serialized Clients To Become Compliant • From DSCSA Compliant ASN Generation To Primary and Secondary Packaging Serialization • Range of Options To Assist Manufacturers When: • Equipment Is Serialized With Delay In Level 4 Solution • Level 4 Solution Implemented With Delay In Equipment Validation • No Level 4 Or Equipment Serialization Solution • Serialized Product But Requires DSCSA-Compliant Advanced Ship Notice (ASN)


Location Houston, TX Site Map WDPrx 10863 Rockley Road Houston, TX 77099 (281) 530 -3077 www. WDPrx. com info@wdprx. com
- Slides: 31