CHRONIC COMPLICATIONS OF DIABETES MELLITUS By AKPAREVA EWOMAZINO

CHRONIC COMPLICATIONS OF DIABETES MELLITUS By AKPAREVA EWOMAZINO

OUTLINE OVERVIEW Types Risk Factors CHRONIC COMPLICATIONS

WHAT IS DIABETES MELLITUS? Diabetes mellitus is a metabolic disease that is characterized by chronic hyperglycemia due to insulin deficiency, tissue resistance to insulin or both. Polyuria, Polydipsia, Polyphagia and Weight Loss.

Types Type 1 DM- previously known as IDDM, autoimmune destruction of beta cells of islet of Langerhans. Type 2 DM- tissue resistance to insulin Gestational DM – first discovered in pregnancy. Human placental lactogen, A. k. a human chorionic somatomammotropin. MODY - is a rare autosomal dominant inherited form of diabetes, due to one of several single-gene mutations causing defects in insulin production Others



Risk factors

complications Could be Acute or Chronic Acute Diabetic ketoacidosis Hyperglycemia hyperosmolar state Hypoglycemia Lactic Acidosis Diabetic coma

Chronic complications Vascular Non-vascular
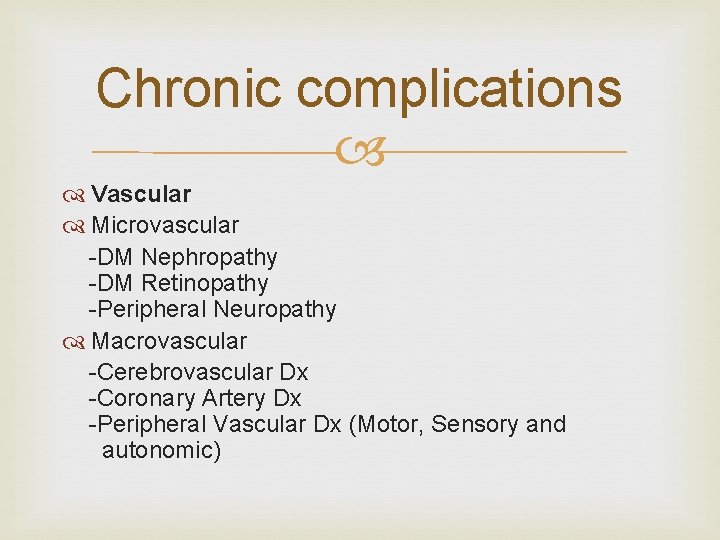
Chronic complications Vascular Microvascular -DM Nephropathy -DM Retinopathy -Peripheral Neuropathy Macrovascular -Cerebrovascular Dx -Coronary Artery Dx -Peripheral Vascular Dx (Motor, Sensory and autonomic)

Chronic Complication Non Vascular -Infection -cataract -Glaucoma -Periodontal disease -Dermatological

Pathophysiology • Non-enzymatic glycosylation : of a wide variety of proteins, such as haemoglobin, collagen, low-density lipoprotein (LDL) and tubulin in peripheral nerves. This leads to an accumulation of advanced glycosylated end-products, causing injury and inflammation via stimulation of pro-inflammatory factors, such as complement and cytokines. Advanced glycation end products (AGEs)
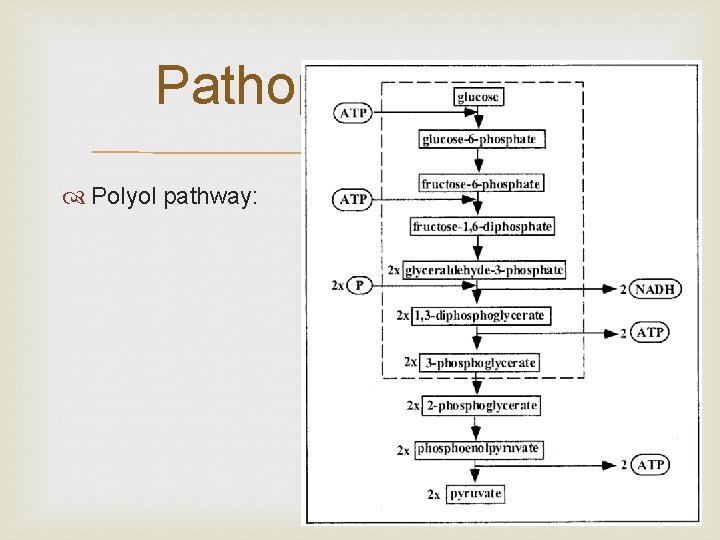
Pathophysiology Polyol pathway:
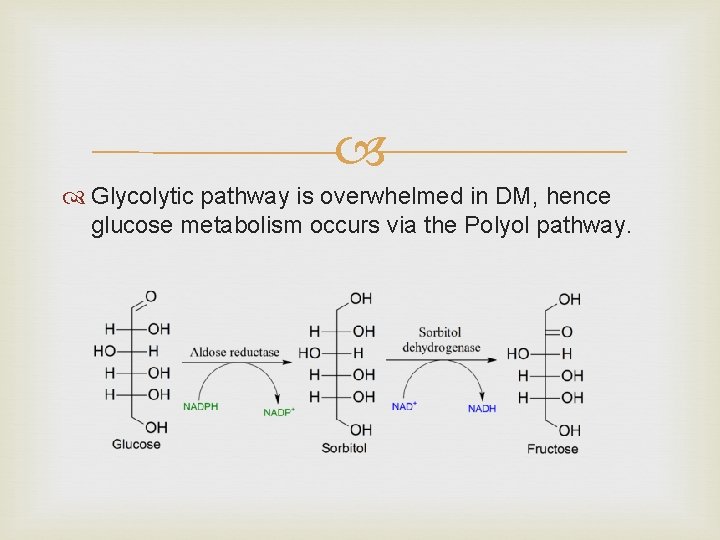
Glycolytic pathway is overwhelmed in DM, hence glucose metabolism occurs via the Polyol pathway.
Polyol pathway. The metabolism of glucose by increased intracellular aldose reductase leads to accumulation of sorbitol and fructose. As sorbitol does not easily dissolve across cell membranes this increases cellular osmolarity, ultimately leading to cell damage. Also causes changes in vascular permeability, cellproliferation and capillary structure via stimulation of protein kinase C and transforming growth factor beta (TGF-β).
• Abnormal microvascular blood flow. This impairs the supply of nutrients and oxygen. In insulin resistance the high levels of blood insulin results in increased production and activity of ET-1, which promotes vasoconstriction and elevates blood pressure Microvascular occlusion is due to vasoconstrictors, such as endothelins and thrombogenesis, and leads to endothelial damage.

• Other factors. These include the formation of reactive oxygen species and stimulation of growth factors TGF-β and vascular endothelial growth factor (VEGF). These growth factors are released by ischaemic tissues and cause endothelial cells to proliferate.

DM Nephropathy The kidney may be damaged by diabetes in three main ways: • glomerular damage • ischaemia resulting from hypertrophy of afferent and efferent arterioles • ascending infection. - common in women but occurs in men due to bladder stasis from autonomic neuropathy

Clinical nephropathy secondary to glomerular disease usually manifests 15– 25 years after diagnosis of diabetes and affects 25– 35% of patients diagnosed under the age of 30 years. It is the leading cause of premature death in young diabetic patients.

Pathophysiology of DM Nephropathy The earliest functional abnormality in the diabetic kidney is renal hypertrophy associated with a raised glomerular filtration rate. This appears soon after diagnosis and is related to poor glycaemic control. As the kidney becomes damaged by diabetes, the afferent arteriole (leading to the glomerulus) becomes vasodilated to a greater extent than the efferent glomerular arteriole. This increases the intraglomerular filtration pressure, further damaging the glomerular capillaries. This process eventually leads to glomerular sclerosis.

DM Retinopathy Most commonly diagnosed diabetes-related complication. Its prevalence increases with the duration of diabetes Some 20% of people with type 1 diabetes will have retinal changes after 10 years, rising to >95% after 20 years 20– 30% of people with type 2 diabetes have retinopathy at diagnosis.

Pathophysiology of DM Retinopathy The metabolic consequences of poorly controlled diabetes cause intramural pericyte death, and thickening of the basement membrane in the small blood vessels of the retina. Eventually leads to occlusion of the vessels.
Non-proliferative: dot and blot haemorrhage(micro aneurysm) hard exudates Preproliferative: multiple cotton wool spots Proliferative: features of neovascularization Maculopathy: hard exudate seen in the macula


Peripheral Neuropathy Diabetes can damage peripheral nervous tissue in a number of ways. The vascular hypothesis postulates occlusion of the vasa nervorum as the prime cause. Since hyperglycaemia leads to increased formation of sorbitol and fructose in Schwann cells, accumulation of these sugars may disrupt function and structure. Tingling, Numbness, Burning or pain

NEUROPATHY The following varieties of neuropathy occur symmetrical, mainly sensory polyneuropathy (distal) • acute painful neuropathy • mononeuropathy and mononeuritis multiplex – cranial nerve lesions – isolated peripheral nerve lesions • diabetic amyotrophy (asymmetrical motor diabetic neuropathy) • autonomic neuropathy.


Macrovascular • Cerebrovascular Dx: 2 – 4 times more likely. • Coronary artery disease, leading to angina or myocardial infarction. MI is 2– 4 times as likely • Peripheral vascular disease, which contributes to intermittent claudication (exertion-related leg and foot pain) as well as diabetic foot. Amputation of a foot for gangrene is 50 times as likely.

PATHOPHYSIOLOGY Numerous studies have shown that patients with diabetes mellitus have accelerated atherosclerotic vascular disease, and major advances in understanding its pathogenesis have been made. Current suggestions are that endothelial injury may be the initial event in the genesis of atherosclerosis, followed by platelet adhesion and aggregation at the site of injury.
Advanced Glycation End-Products: protein and lipids become glycated on exposure to excess sugars. AGEs can induce crosslinking of collagen causing vascular stiffening and entrapment of LDL in the arterial walls AGEs protein cause LDL glycation which can promotes LDL oxydation which may initiate an inflammatory pathway vascular endothelial cells.

In uncontrolled diabetes, elevated plasma low density lipoprotein levels and decreased plasma high density lipoprotein levels favor lipid deposition in large vessels. There is evidence of a thrombotic state in certain diabetic patients. Collectively, these abnormalities of endothelial, platelet, smooth muscle, lipoprotein, and coagulation behavior may be viewed as contributing to the problem of accelerated atherosclerosis in diabetes.

Non-vascular Complications Infection The immune response is impaired in individuals with diabetes mellitus. Cellular studies have shown that hyperglycemia both reduces the function of immune cells and increases inflammation. chemotaxis and phagocytosis by polymorphonuclear leucocytes are impaired because, at high blood glucose concentrations, neutrophil superoxide generation is impaired.

Infections in DM Skin – staphylococcal infections (boils, abscesses, carbuncles) – mucocutaneous candidiasis • Gastrointestinal tract – periodontal disease – rectal and ischiorectal abscess formation (when control is very poor) • Urinary tract – urinary tract infections (in women) – pyelonephritis – perinephric abscess • Lungs – staphylococcal and pneumococcal pneumonia – Gram-negative bacterial pneumonia – tuberculosis • Bone – spontaneous staphylococcal spinal osteomyelitis.

Glaucoma After diabetic retinopathy, retinal vein occlusion is the second most common cause of vision loss. This results from a thrombus forming at the central retinal vein near the lamina cribrosa. It is characterized by diffuse retinal hemorrhages, venous dilation and tortuosity, optic disc swelling, cotton wool spots and macular edema. It is a common cause of iris neovascularization, which may lead to secondary glaucoma

Cataract develops earlier in people with diabetes than in the general population. Sustained very poor diabetes control with a degree of ketosis can cause an acute cataract (snowflake cataract), which comes on rapidly. Fluctuations in blood glucose concentration cause refractive variability, as a result of osmotic changes within the lens.

Conclusion Diabetes Mellitus is a disease with morbid complications and so all effort must be put in place to decrease its incidence while ensuring adequate blood glucose control in people with DM.
- Slides: 36