CH 3 Organic Compounds Alkanes and Their Stereochemistry


















- Slides: 58

CH 3: Organic Compounds: Alkanes and Their Stereochemistry Renee Y. Becker CHM 2210 Valencia Community College 1

Why this Chapter • Alkanes are unreactive, but provide useful vehicle to introduce important ideas about organic compounds • Alkanes will be used to discuss basic approaches to naming organic compounds • We will take an initial look at 3 -D aspects of molecules 2

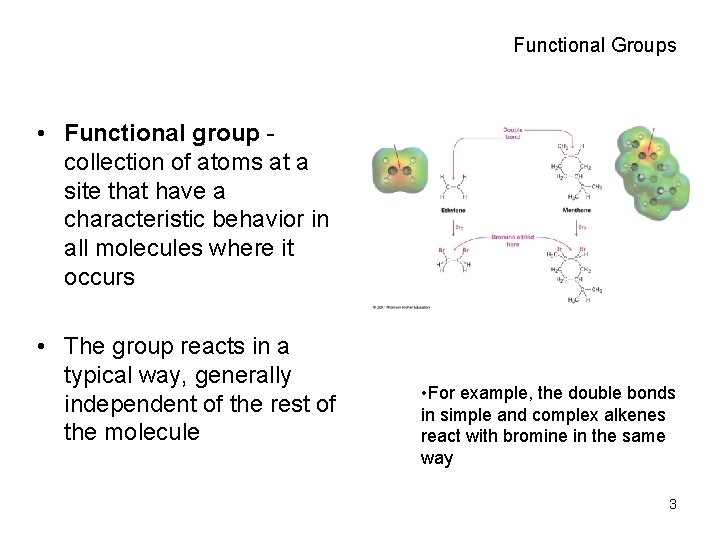
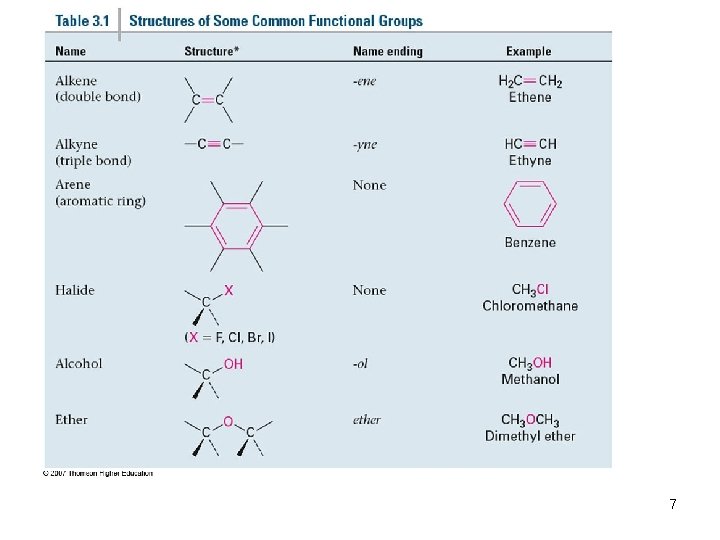
Functional Groups • Functional group collection of atoms at a site that have a characteristic behavior in all molecules where it occurs • The group reacts in a typical way, generally independent of the rest of the molecule • For example, the double bonds in simple and complex alkenes react with bromine in the same way 3

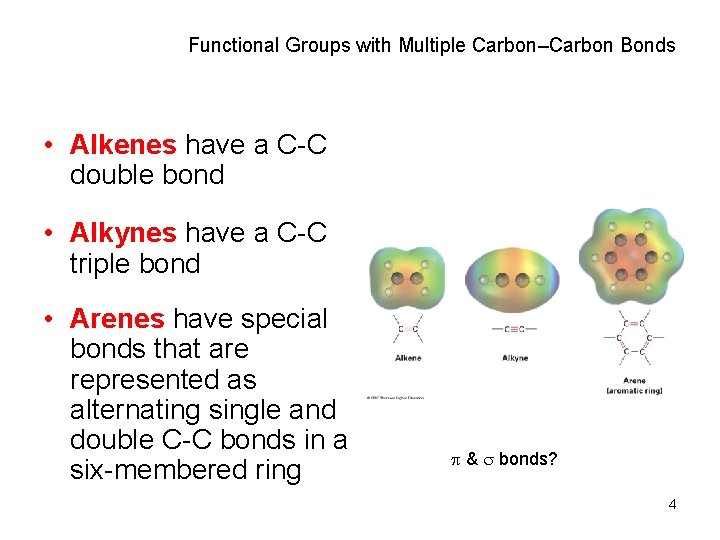
Functional Groups with Multiple Carbon–Carbon Bonds • Alkenes have a C-C double bond • Alkynes have a C-C triple bond • Arenes have special bonds that are represented as alternating single and double C-C bonds in a six-membered ring & bonds? 4

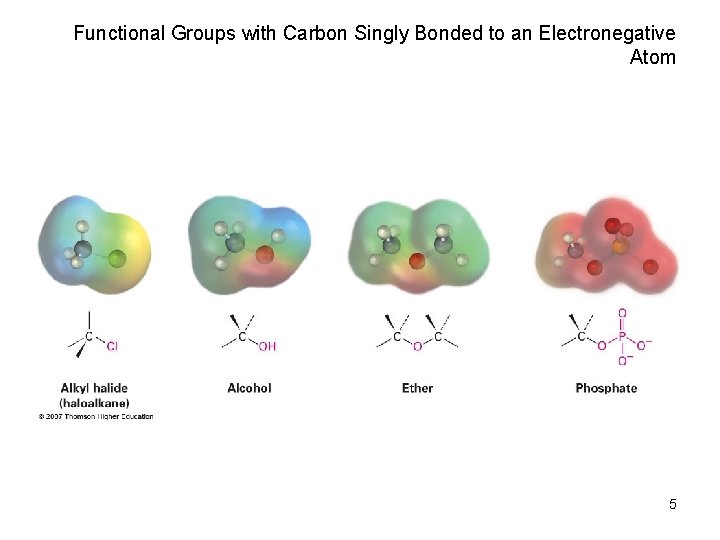
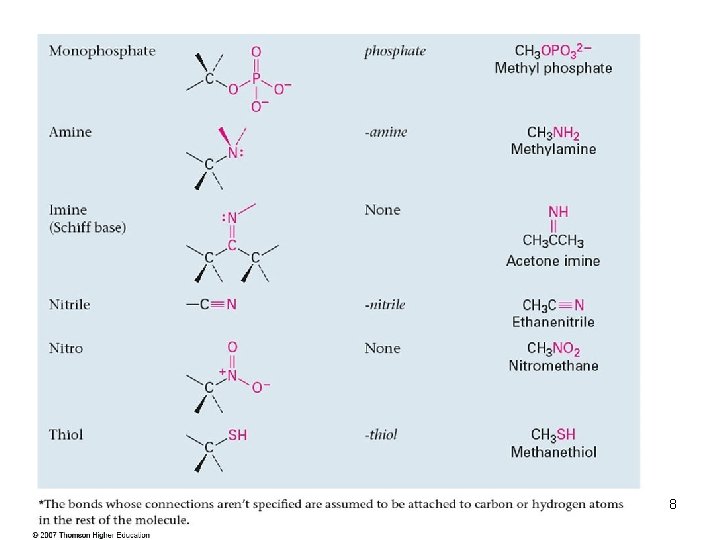
Functional Groups with Carbon Singly Bonded to an Electronegative Atom 5

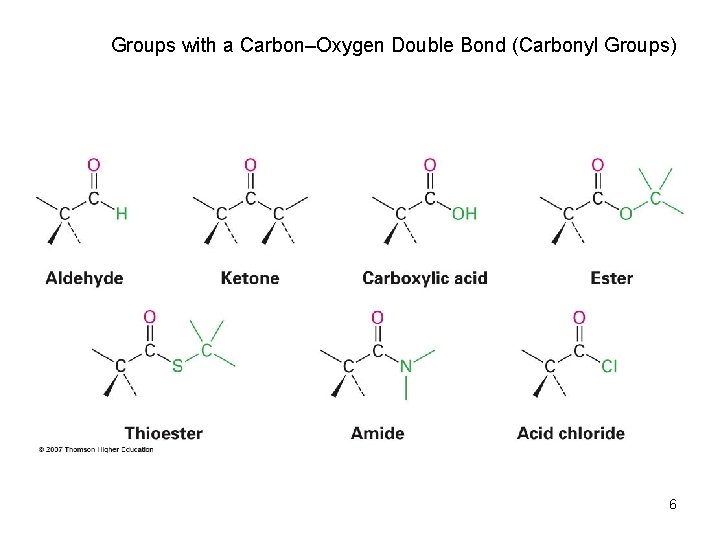
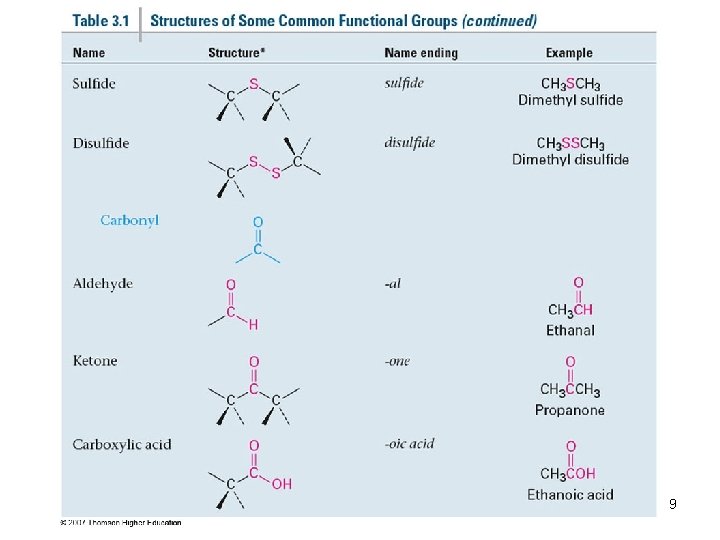
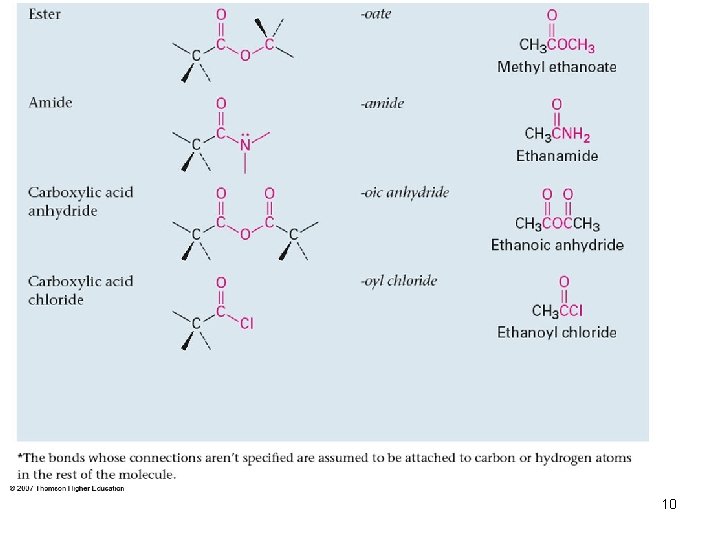
Groups with a Carbon–Oxygen Double Bond (Carbonyl Groups) 6

7

8

9

10

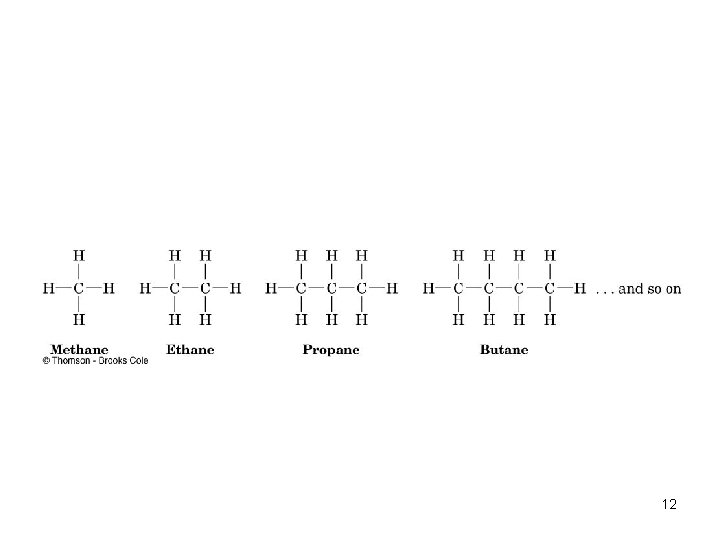
Alkanes • Alkanes: Compounds with C-C single bonds and C-H bonds only (no functional groups) • Connecting carbons can lead to large or small molecules • The formula for an alkane with no rings in it must be Cn. H 2 n+2 where the number of C’s is n • Alkanes are saturated with hydrogen (no more can be added • They are also called aliphatic compounds 11

12

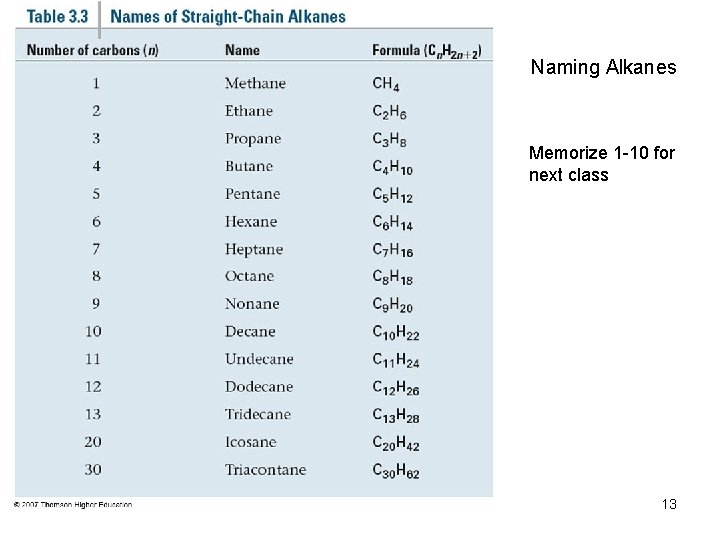
Naming Alkanes Memorize 1 -10 for next class 13

Alkane Isomers • The molecular formula of an alkane with more than three carbons can give more than one structure – C 4 (butane) = butane and isobutane – C 5 (pentane) = pentane, 2 -methylbutane, and 2, 2 dimethylpropane • Alkanes with C’s connected to no more than 2 other C’s are straight-chain or normal alkanes • Alkanes with one or more C’s connected to 3 or 4 C’s are branched-chain alkanes 14

Constitutional Isomers • Isomers that differ in how their atoms are arranged in chains are called constitutional isomers • Compounds other than alkanes can be constitutional isomers of one another • They must have the same molecular formula to be isomers 15

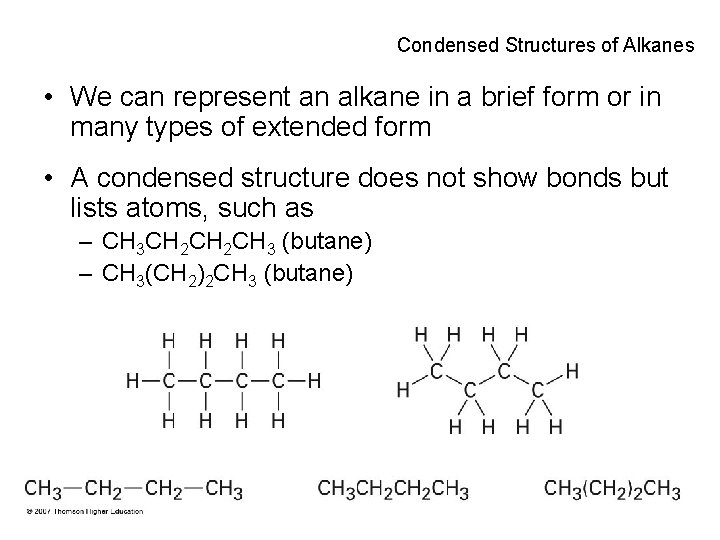
Condensed Structures of Alkanes • We can represent an alkane in a brief form or in many types of extended form • A condensed structure does not show bonds but lists atoms, such as – CH 3 CH 2 CH 3 (butane) – CH 3(CH 2)2 CH 3 (butane) 16
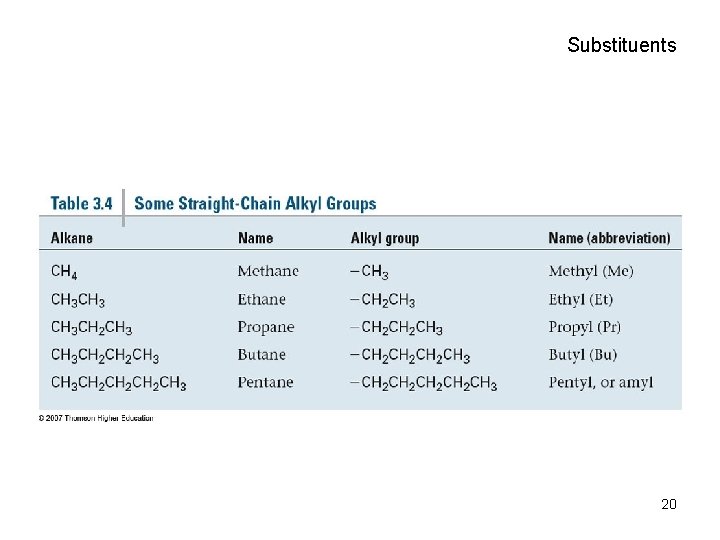
Alkyl Groups • Alkyl group – remove one H from an alkane (a part of a structure) • General abbreviation “R” (for Radical, an incomplete species or the “rest” of the molecule) • Name: replace -ane ending of alkane with -yl ending – CH 3 is “methyl” (from methane) – CH 2 CH 3 is “ethyl” from ethane 17

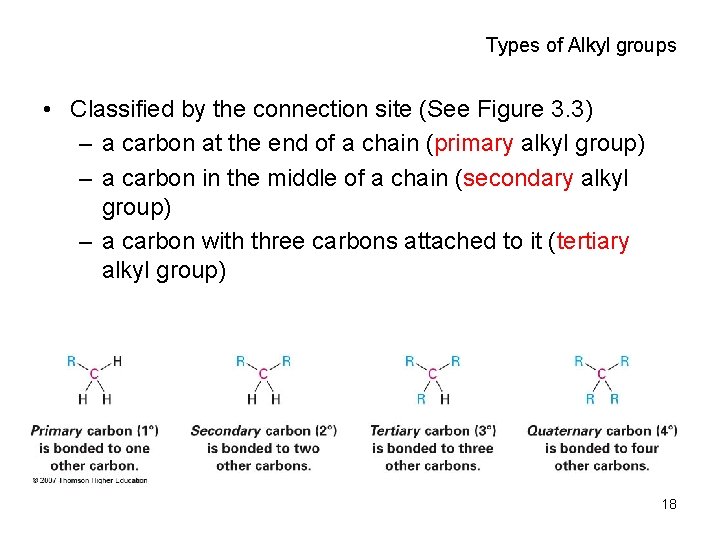
Types of Alkyl groups • Classified by the connection site (See Figure 3. 3) – a carbon at the end of a chain (primary alkyl group) – a carbon in the middle of a chain (secondary alkyl group) – a carbon with three carbons attached to it (tertiary alkyl group) 18

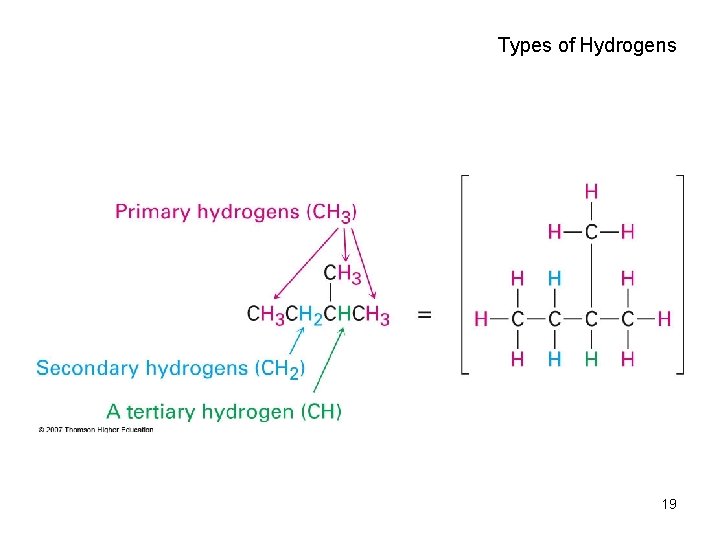
Types of Hydrogens 19

Substituents 20

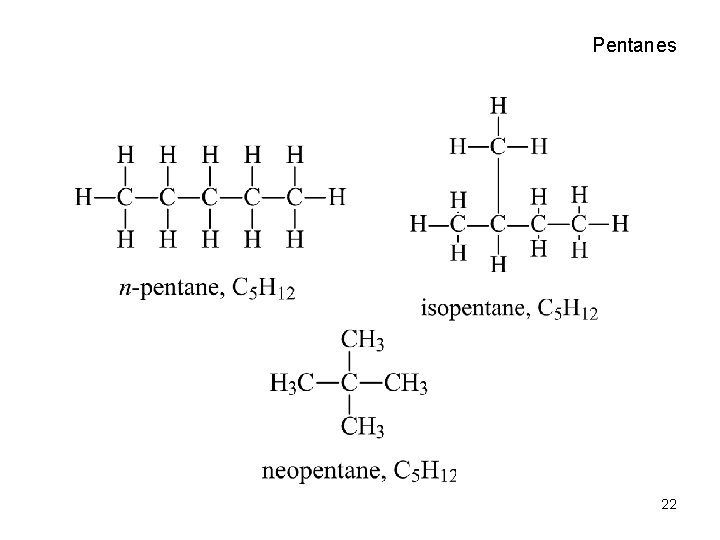
Common Names • Isobutane, “isomer of butane” • Isopentane, isohexane, etc. , methyl branch on next-to-last carbon in chain. • Neopentane, most highly branched • Five possible isomers of hexane, 18 isomers of octane and 75 for decane! 21

Pentanes 22

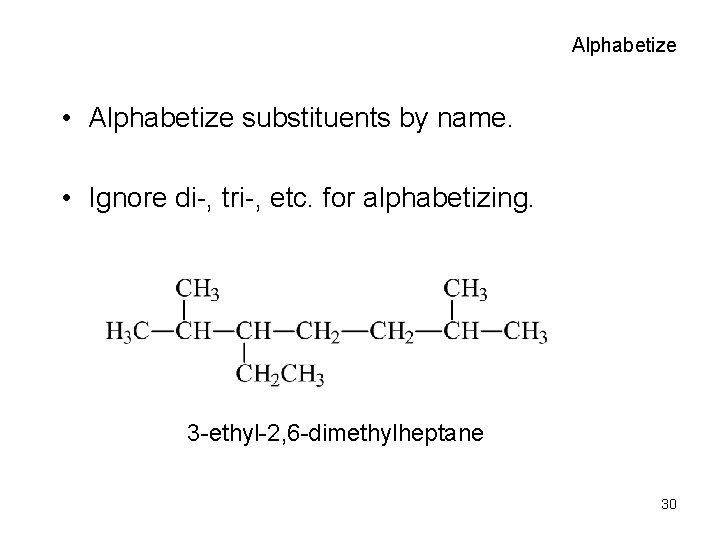
IUPAC Names • Find the longest continuous carbon chain. • Number the carbons, starting closest to the first branch. • Name the groups attached to the chain, using the carbon number as the locator. • Alphabetize substituents. • Use di-, tri-, etc. , for multiples of same substituent. 23

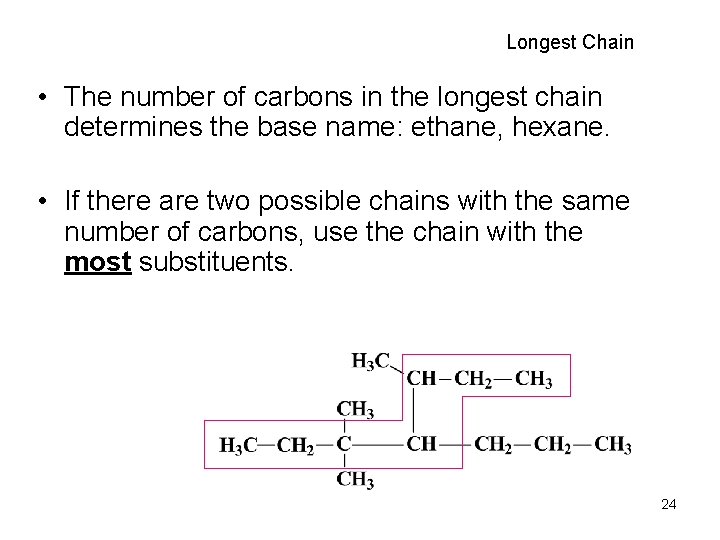
Longest Chain • The number of carbons in the longest chain determines the base name: ethane, hexane. • If there are two possible chains with the same number of carbons, use the chain with the most substituents. 24

Number the Carbons • Start at the end closest to the first attached group. • If two substituents are equidistant, look for the next closest group. 1 3 2 4 5 6 7 25

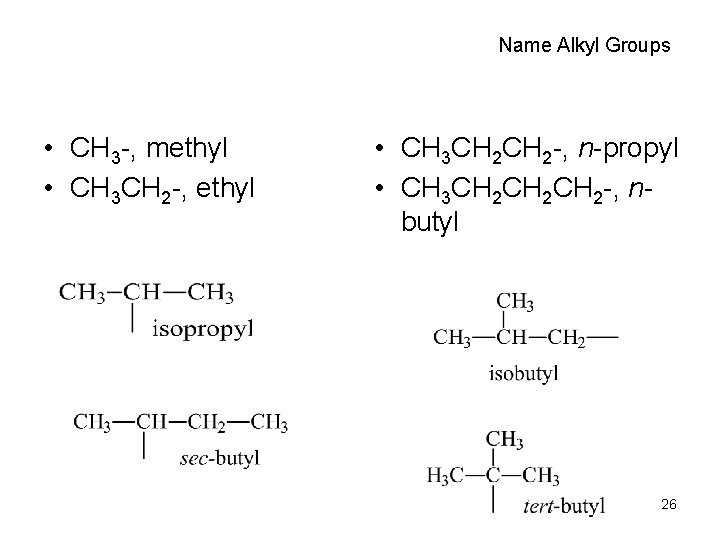
Name Alkyl Groups • CH 3 -, methyl • CH 3 CH 2 -, ethyl • CH 3 CH 2 -, n-propyl • CH 3 CH 2 CH 2 -, nbutyl 26

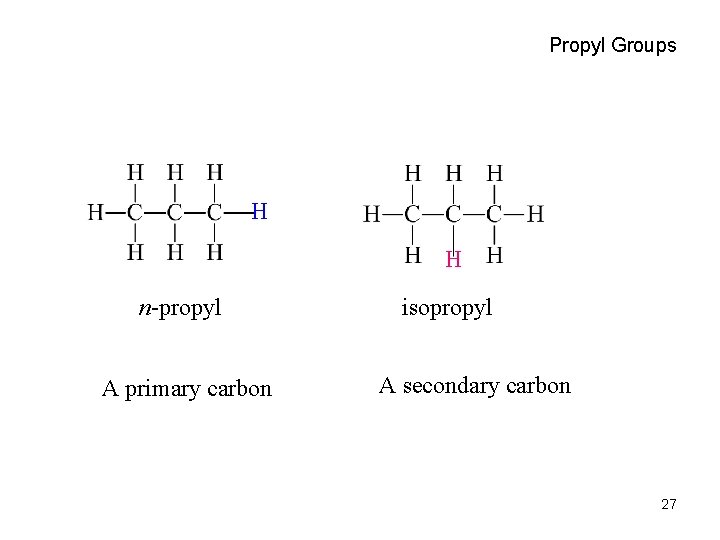
Propyl Groups H H n-propyl A primary carbon isopropyl A secondary carbon 27

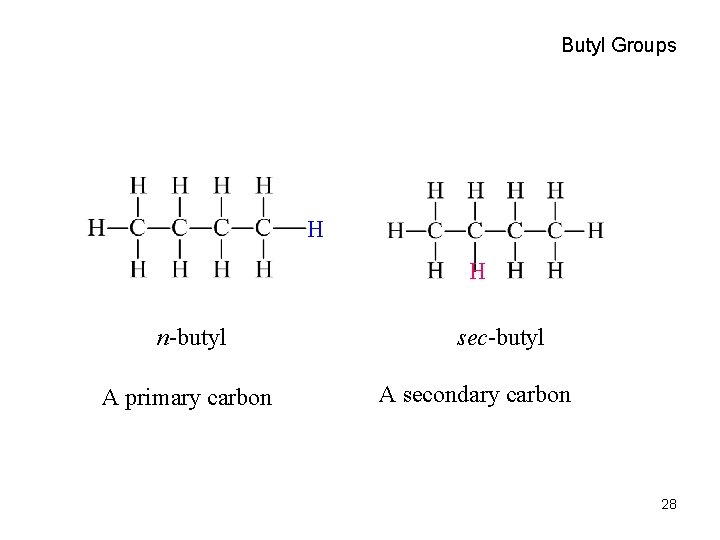
Butyl Groups H H n-butyl A primary carbon sec-butyl A secondary carbon 28

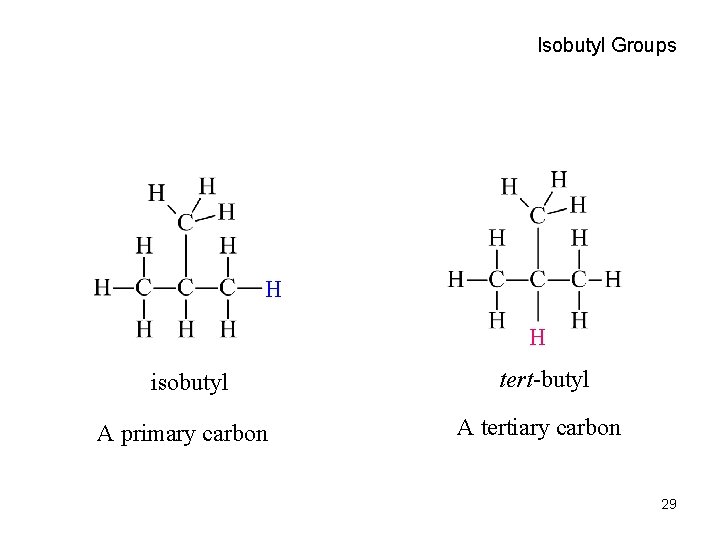
Isobutyl Groups H H isobutyl A primary carbon tert-butyl A tertiary carbon 29

Alphabetize • Alphabetize substituents by name. • Ignore di-, tri-, etc. for alphabetizing. 3 -ethyl-2, 6 -dimethylheptane 30

Example 1 Write structures for the following: a) 3 -ethyl-3 -methylpentane b) 4 -t-butyl-2 -methylheptane c) 5 -isopropyl-3, 3, 4 -trimethyloctane d) 3 -ethyl-2, 4, 5 -trimethylheptane 31

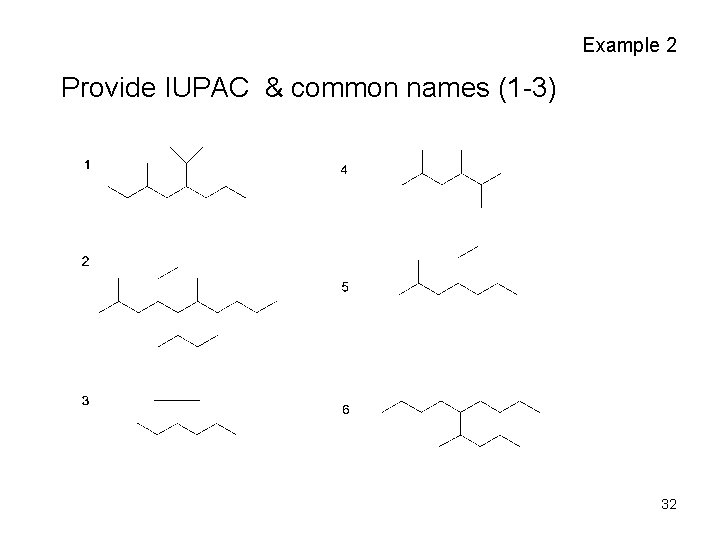
Example 2 Provide IUPAC & common names (1 -3) 32

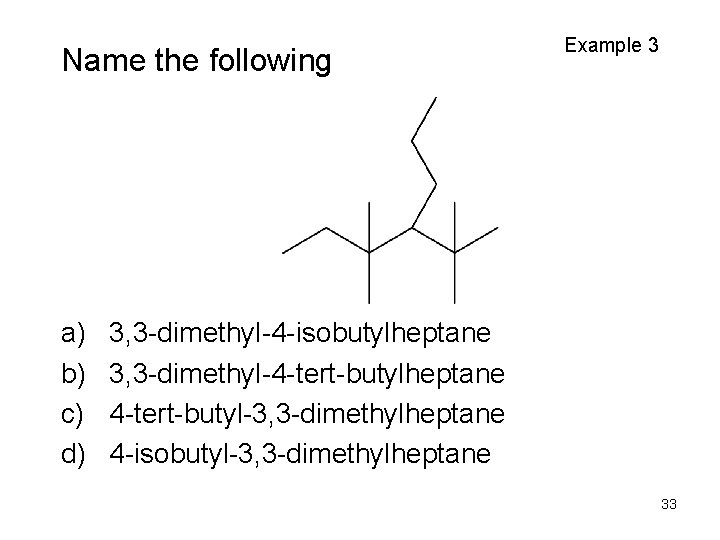
Name the following a) b) c) d) Example 3 3, 3 -dimethyl-4 -isobutylheptane 3, 3 -dimethyl-4 -tert-butylheptane 4 -tert-butyl-3, 3 -dimethylheptane 4 -isobutyl-3, 3 -dimethylheptane 33

Complex Substituents • If the branch has a branch, number the carbons from the point of attachment. • Name the branch off the branch using a locator number. • Parentheses are used around the complex branch name. • For alphabetizing use the first letter of the complex sub. Even if it is a numerical (di, tri, etc) 34

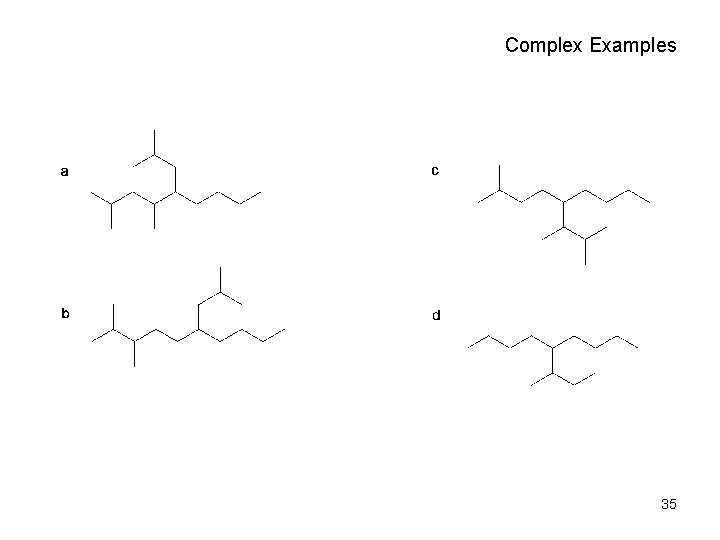
Complex Examples 35

Example 4 Draw the structures and give their more common names a) (1 -methyl) group b) (2 -methylpropyl) group c) (1 -methylpropyl) group d) (1, 1 -dimethyl) group 36

Example 5 Draw the structures a) 4 -(1 -methyl)heptane b) 5 -(1, 2, 2 -trimethylpropyl)nonane 37

Assignment • For next class draw the 9 isomers of heptane and name them 38

Properties of Alkanes • Called paraffins (low affinity compounds) because they do not react as most chemicals • They will burn in a flame, producing carbon dioxide, water, and heat • They react with Cl 2 in the presence of light to replace H’s with Cl’s (not controlled) 39

Physical Properties: Alkanes • Solubility: hydrophobic • Density: less than 1 g/m. L • Boiling points increase with increasing carbons (little less for branched chains). • Melting points increase with increasing carbons (less for odd-number of carbons). 40

Boiling Points of Alkanes Branched alkanes have less surface area contact, so weaker intermolecular forces. 41

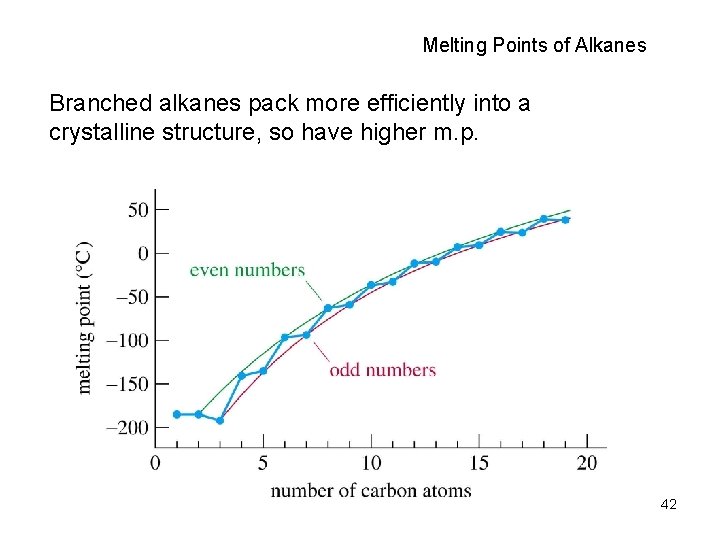
Melting Points of Alkanes Branched alkanes pack more efficiently into a crystalline structure, so have higher m. p. 42

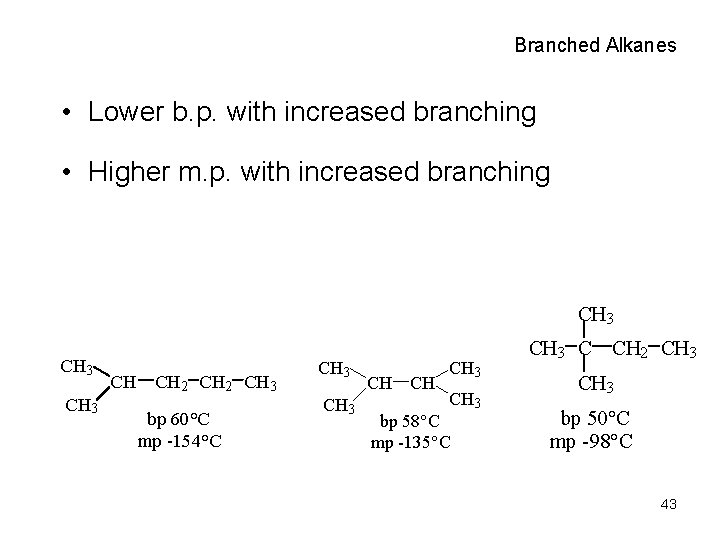
Branched Alkanes • Lower b. p. with increased branching • Higher m. p. with increased branching CH 3 CH CH 2 CH 3 bp 60°C mp -154°C CH 3 CH CH CH 3 bp 58°C mp -135°C CH 3 C CH 2 CH 3 bp 50°C mp -98°C 43

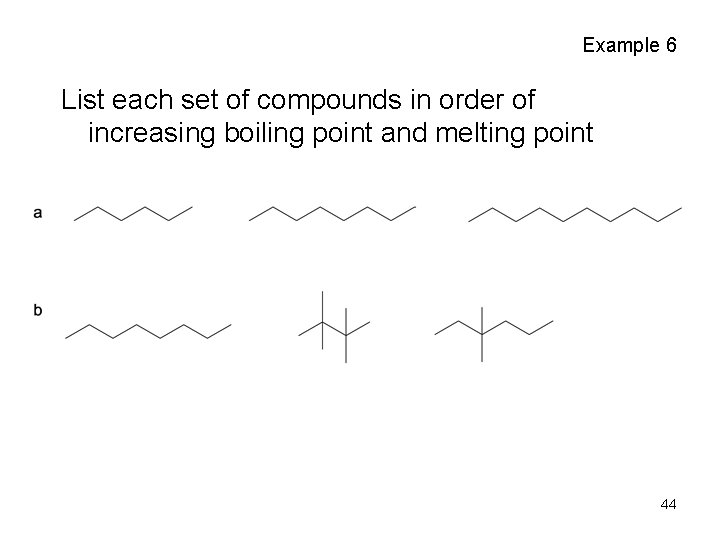
Example 6 List each set of compounds in order of increasing boiling point and melting point 44

Example 7 Which of the following has the highest boiling point? a) 3 -methylpentane b) 2, 2 -dimethylbutane c) Hexane d) Methane 45

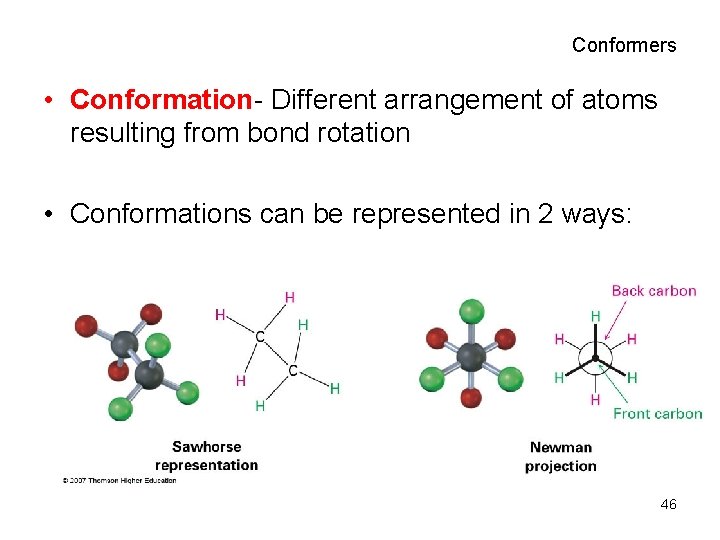
Conformers • Conformation- Different arrangement of atoms resulting from bond rotation • Conformations can be represented in 2 ways: 46

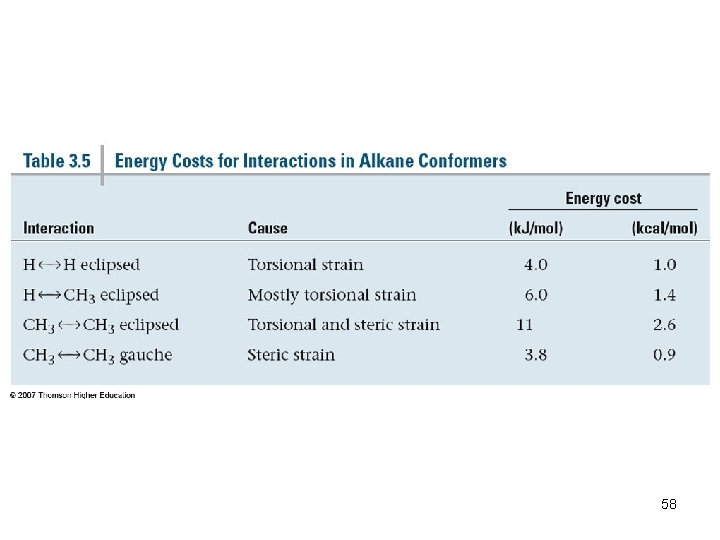
Torsional Strain • We do not observe perfectly free rotation • There is a barrier to rotation, and some conformers are more stable than others • Staggered- most stable: all 6 C-H bonds are as far away as possible • Eclipsed- least stable: all 6 C-H bonds are as close as possible to each other 47

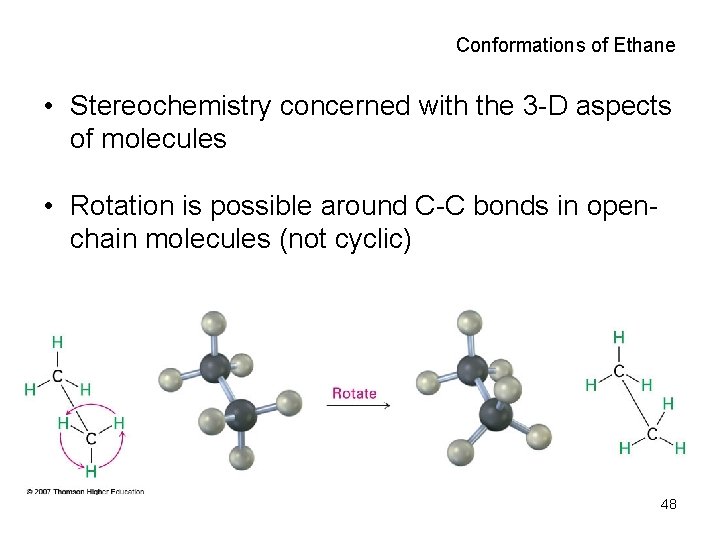
Conformations of Ethane • Stereochemistry concerned with the 3 -D aspects of molecules • Rotation is possible around C-C bonds in openchain molecules (not cyclic) 48

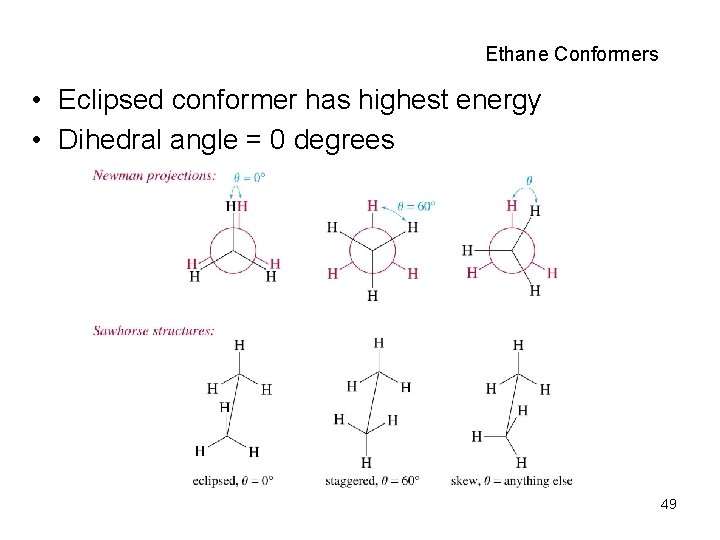
Ethane Conformers • Eclipsed conformer has highest energy • Dihedral angle = 0 degrees 49

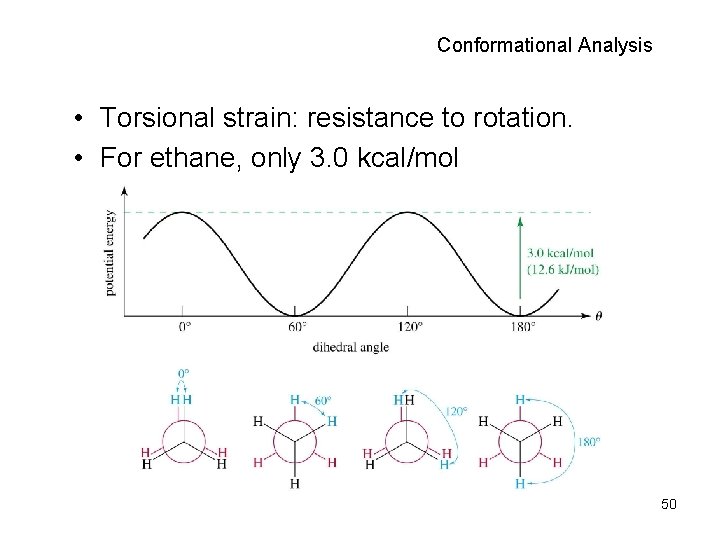
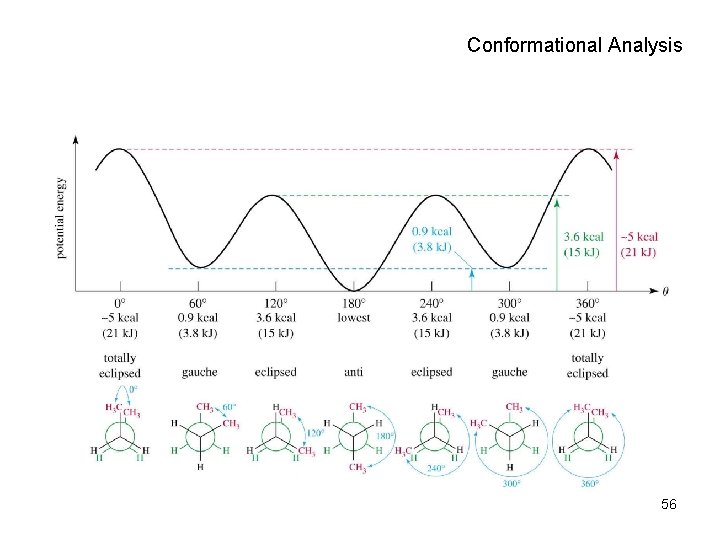
Conformational Analysis • Torsional strain: resistance to rotation. • For ethane, only 3. 0 kcal/mol 50

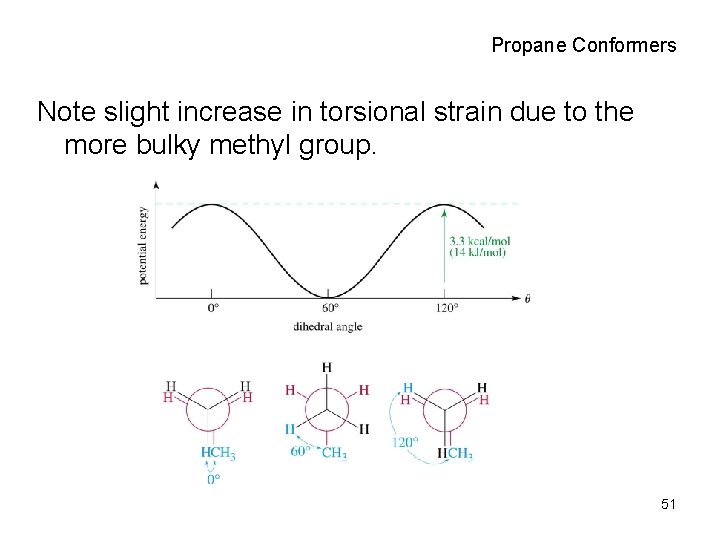
Propane Conformers Note slight increase in torsional strain due to the more bulky methyl group. 51

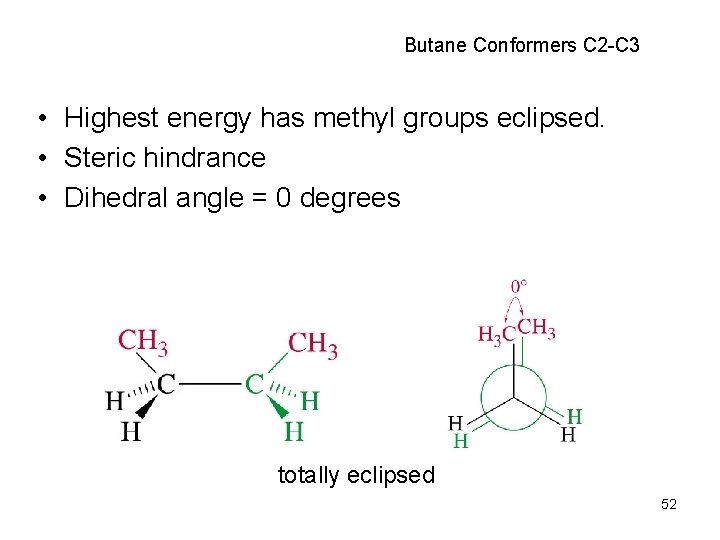
Butane Conformers C 2 -C 3 • Highest energy has methyl groups eclipsed. • Steric hindrance • Dihedral angle = 0 degrees totally eclipsed 52

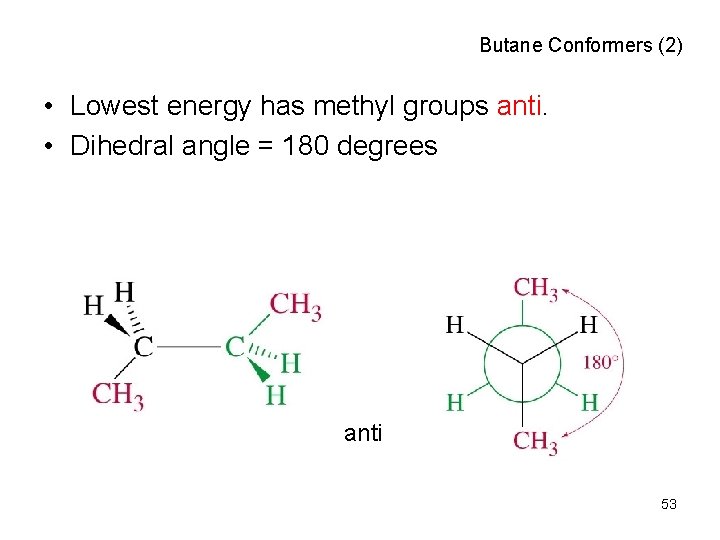
Butane Conformers (2) • Lowest energy has methyl groups anti. • Dihedral angle = 180 degrees anti 53

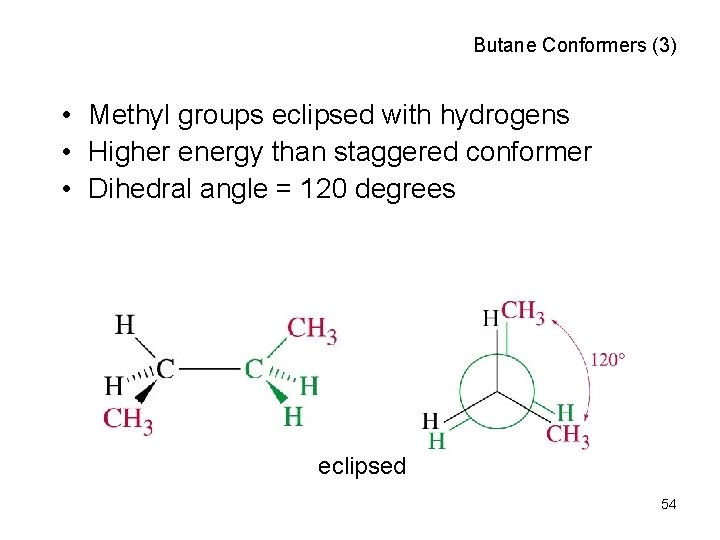
Butane Conformers (3) • Methyl groups eclipsed with hydrogens • Higher energy than staggered conformer • Dihedral angle = 120 degrees eclipsed 54

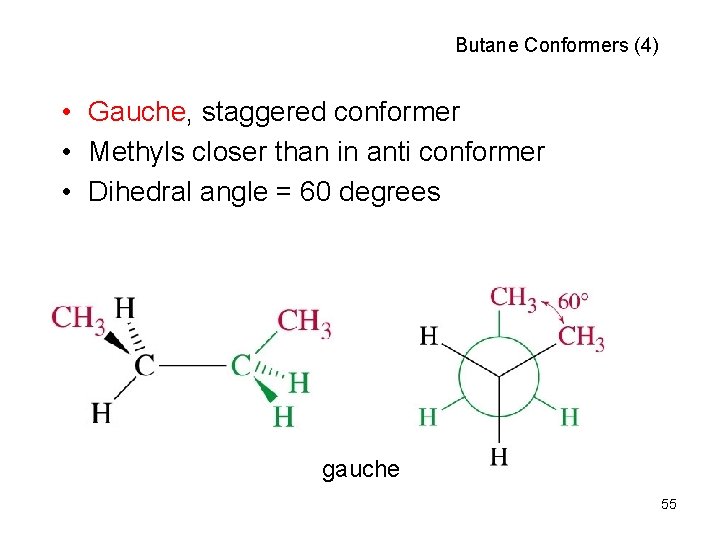
Butane Conformers (4) • Gauche, staggered conformer • Methyls closer than in anti conformer • Dihedral angle = 60 degrees gauche 55

Conformational Analysis 56

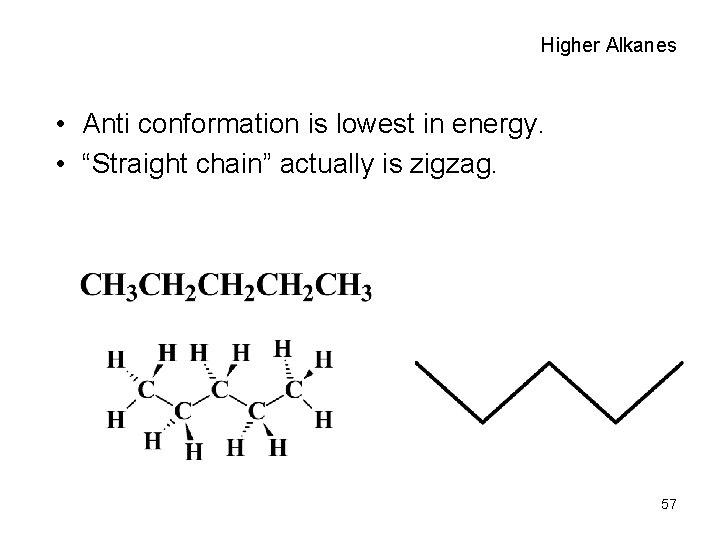
Higher Alkanes • Anti conformation is lowest in energy. • “Straight chain” actually is zigzag. 57

58