Properties of Organic and Inorganic Compounds Experiment 1











- Slides: 16

Properties of Organic and Inorganic Compounds Experiment 1 Chem 121 Organic Chemistry Laboratory 1

What is ORGANIC CHEMISTRY? 2

Introduction Vital force – ‘vitalism’ Friedrich Wohler overthrew vitalism ◦ Synthesis of urea from ammonium cyanate Distinguishing feature: organic compounds all contain the CARBON 3

What is ORGANIC chemistry? the study of carbon containing compounds ◦ Other elements in organic compounds: H, O, N, S, P, Cl, Br, I and other transition metals Why Carbon? ◦ Can share four valence electrons ◦ Form strong covalent bonds ◦ Form rings and long chains, e. g. benzene and DNA 4

Definition of terms Ionic compounds: compounds made up of a metal cation and a nonmetal anion ◦ e. g. Na. Cl, KNO 3 Intramolecular forces of attraction: forces existing within molecules that holds the atoms together ◦ e. g. Ionic bond, covalent bond, metallic bond Intermolecular forces of attraction: 5

Intermolecular Forces of Attraction Ion-dipole ◦ Between an ionic compound a polar compound e. g. Na. Cl dissolved in water Dipole-dipole ◦ Between two polar compounds e. g. HCl dissolved in water 6

Hydrogen Bonding ◦ Requirement: H atoms bonded to F, O, N ◦ Strongest intermolecular force ◦ e. g. NH 3 in H 2 O London dispersion forces/van der Waals forces ◦ Between two NONPOLAR compounds ◦ Weakest intermolecular force; present in all organic molecules 7

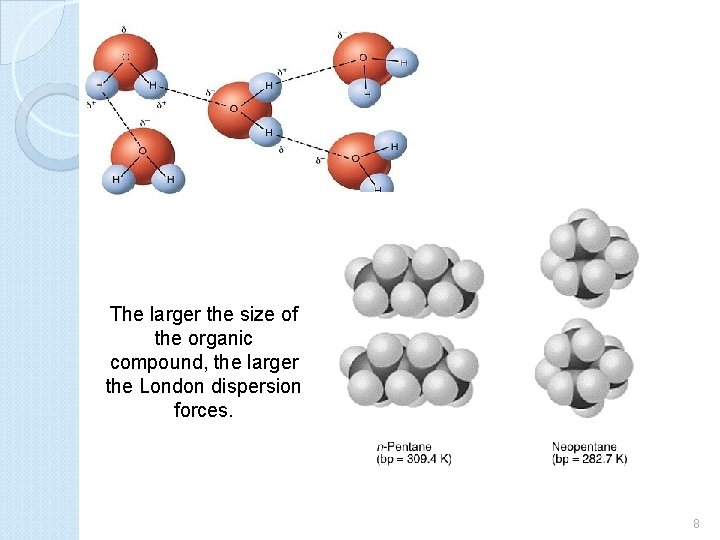
The larger the size of the organic compound, the larger the London dispersion forces. 8

Properties of Organic Compounds Flammable ◦ Due to the C-C bond energies in organic compounds ◦ Energy released is in the form of heat Ethanol vs. Water ◦ Ethanol – produces the distinct blue flame ◦ Water – smothers flame instead of generating one 9

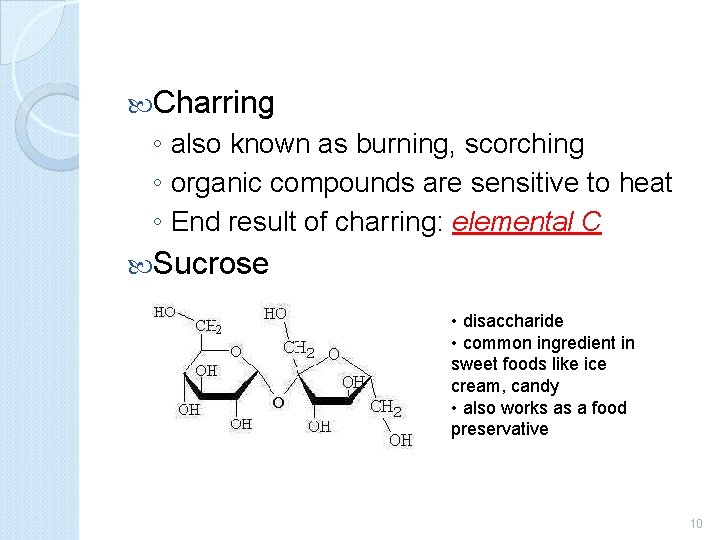
Charring ◦ also known as burning, scorching ◦ organic compounds are sensitive to heat ◦ End result of charring: elemental C Sucrose • disaccharide • common ingredient in sweet foods like ice cream, candy • also works as a food preservative 10

IMF of sucrose: London dispersion force IMF of Na. Cl and Ca. CO 3: no intermolecular force, but intramolecular (IONIC BOND) Remember always: Intramolecular forces are way STRONGER than intermolecular forces. 11

Solubility ◦ relies on the intermolecular forces of organic compounds ◦ ‘like dissolves like’ ◦ Polar solvents dissolve in polar solutes. ◦ Nonpolar solvents dissolve in nonpolar solutes. ◦ Organic compounds = mostly nonpolar It only follows that most organic compounds are soluble in organic solvents. 12

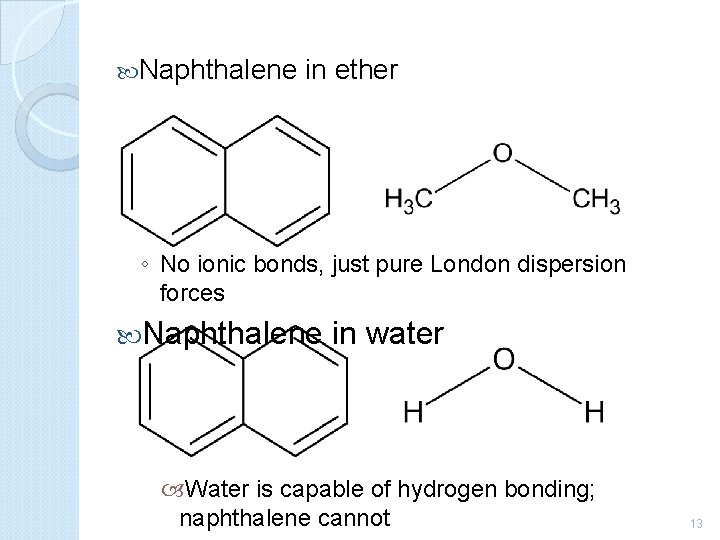
Naphthalene in ether ◦ No ionic bonds, just pure London dispersion forces Naphthalene in water Water is capable of hydrogen bonding; naphthalene cannot 13

Electrical Conductivity Electrical conductivity is only possible when a compound contains charged particles (i. e. an electrolyte) ◦ e. g. Na. Cl, Na. NO 3 are electrolytes Since most organic compounds are molecular, not ionic, it does not conduct electricity. 14

1 M sucrose 1 M Na. Cl 1 M ethanol hexane 15

Summary Organic chemistry is the study of carbon compounds. Organic compounds have the ff properties: ◦ Flammable ◦ Combustible ◦ Immiscble in polar solvents like water ◦ Non-electrolytes; do not conduct electricity 16