ORGANIC CHEMISTRY Organic Chemistry Study of carbon and
ORGANIC CHEMISTRY
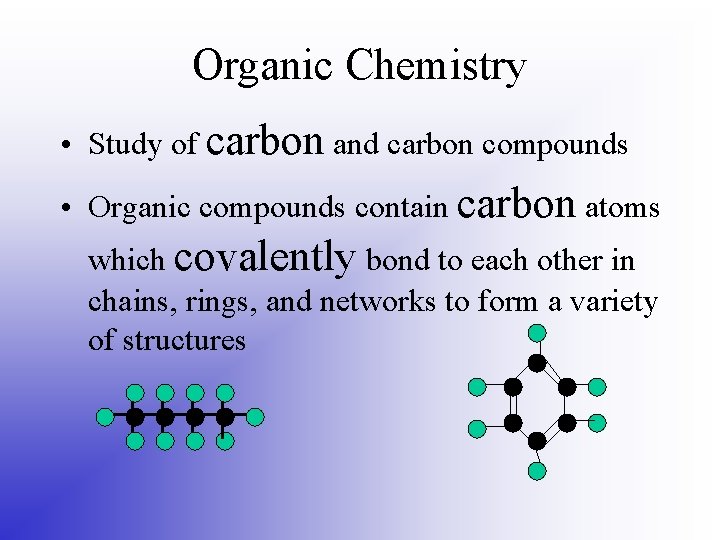
Organic Chemistry • Study of carbon and carbon compounds • Organic compounds contain carbon atoms which covalently bond to each other in chains, rings, and networks to form a variety of structures

General Characteristics of Organic Compounds • Nonpolar • Soluble in nonpolar solvents / insoluble in polar solvents (water) “Like dissolves Like” • Poor conductors of heat and electricity – Exception: aqueous organic acids conduct • Low melting and boiling points • React slower than inorganic compounds
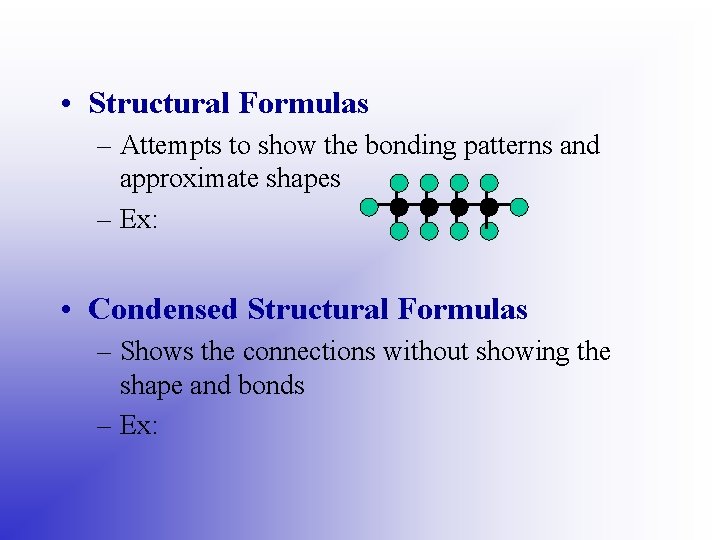
• Structural Formulas – Attempts to show the bonding patterns and approximate shapes – Ex: • Condensed Structural Formulas – Shows the connections without showing the shape and bonds – Ex:

Bonding • Carbon- has 4 valence electrons, needs 4 more, forms 4 bonds • Hydrogen – has 1 valence electron, needs 1 more, forms 1 bond • Oxygen – has 6 valence electrons, needs 2 more, forms 2 bonds • Nitrogen – has 5 valence electrons, needs 3 more, forms 3 bonds • Halogens (group 17) – has 7 valence electrons, needs 1 more, forms 1 bond

Homologous Series (Families) • Group of related compound in which each member has one more group – Ex: CH 3 CH 2 CH 3 and CH 3 CH 2 CH 3 • Members of a group will have similar structures and properties • As the members of a series increase in mass, their boiling (and melting) points increase – due to increased intermolecular forces

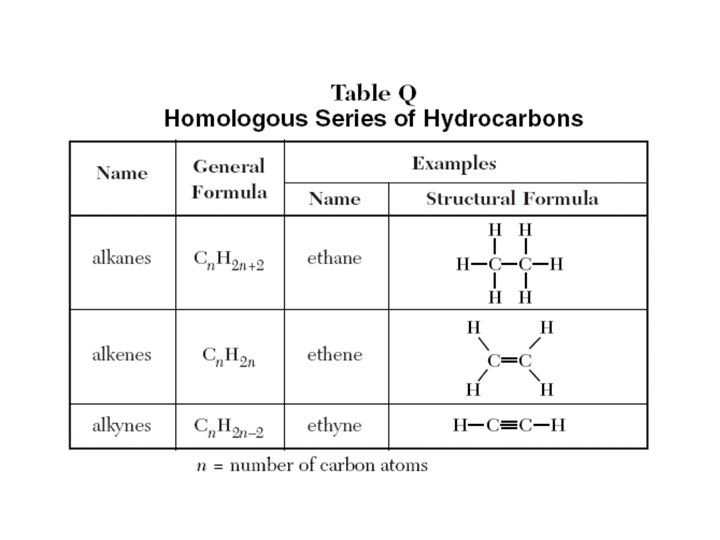
Hydrocarbons • Organic compounds that contain only CARBON and HYDROGEN ALKANES. . . SINGLE bonds between Carbons Saturated hydrocarbon Cn. H 2 n+2 Family Name ALKENES. . . ONE DOUBLE bond between two Carbons ALKYNES. . . ONE TRIPLE bond between two Carbons Unsaturated hydrocarbon Unsaturated Hydrocarbon Cn. H 2 n-2
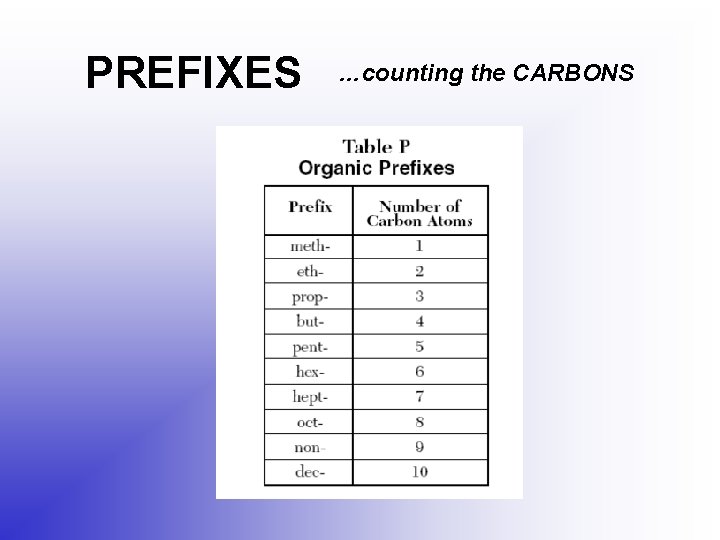
PREFIXES …counting the CARBONS

ALKANES • Contain all single bonds • Naming: carbon prefix + -ane Examples: Butane Octane

ALKENES • Contain 1 double bond • Naming: carbon prefix + -ene – A number is used in front to indicate the location of the double bond – If a number is not indicated the double bond comes after the 1 st carbon Example: 2 -Pentene

Diene • Contains 2 double bonds • Naming: location-carbon prefix-diene • Example: 1, 3 -pentadiene

ALKYNES • Contains 1 triple bond • Naming: location - carbon prefix + -yne Examples: 3 – hexyne Propyne

• Saturated Compounds – contains all single carbon-carbon bonds • Unsaturated Compounds – contains at least one multiple carbon-carbon bond
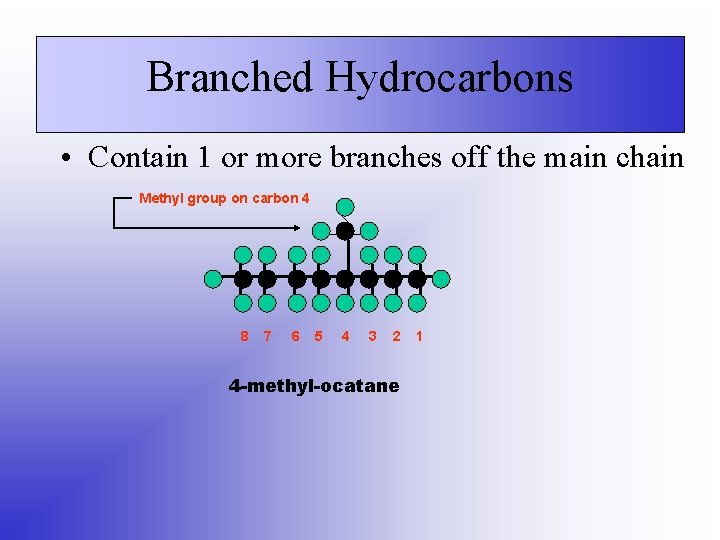
Branched Hydrocarbons • Contain 1 or more branches off the main chain Methyl group on carbon 4 8 7 6 5 4 3 2 4 -methyl-ocatane 1

Naming Branched Hydrocarbons • Find the longest continuous chain, name it • To name the branches: number of carbons + “-yl” – Examples: • CH 3 = methyl • CH 2 CH 3 = ethyl • The location of the alkyl (branched group) is indicated – If there is more than one of the same branch, use prefixes (di, tri, tetra, …) – The carbon chain must be numbered from the end that will give the lowest numbers for the branches Example: 2, 2, 3 – trimethyl pentane
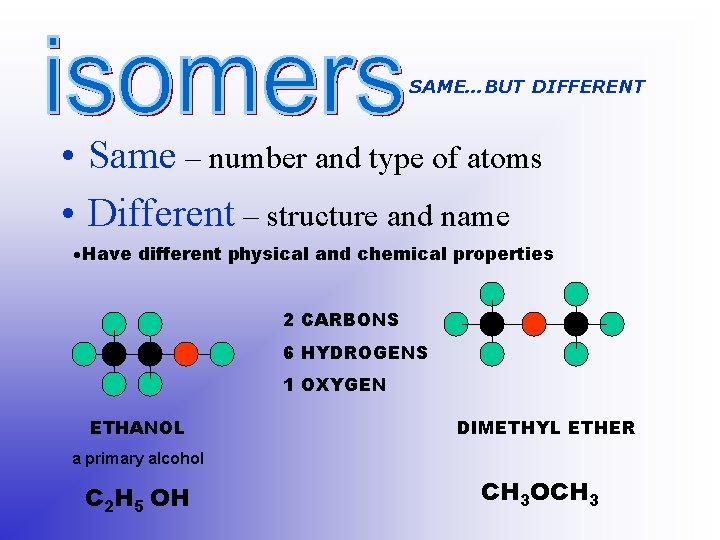
SAME…BUT DIFFERENT • Same – number and type of atoms • Different – structure and name Have different physical and chemical properties 2 CARBONS 6 HYDROGENS 1 OXYGEN ETHANOL DIMETHYL ETHER a primary alcohol C 2 H 5 OH CH 3 OCH 3

GROUPS and COMPOUNDS

• Functional Group: halogen (group 17 element) • Name: location-halide-carbon chain • Examples: 1. 2 -bromobutane 2. 1, 3 - dichloropentane

ALCOHOLS • • • Functional Group: -OH No more than one –OH group can be attached to any one carbon The carbon to which the –OH group is attached must have all single bonds Alcohols are not bases (do not ionize in water) Name: hydrocarbon name, replace the final –e with –ol Examples: 1. Methanol 2. 2 -Propanol

Types of Alcohols • Monohydroxy – contain 1 –OH group – Primary – Secondary – Tertiary • Dihydroxy (Diol) – contain 2 –OH groups • Trihydroxy (Triol) – contain 3 –OH groups

Primary Alcohols • • • Contain 1 –OH group -OH group is attached to the end of the chain Examples: 1. Ethanol 2. Propanol

H SECONDARY ALCOHOL • Contain 1 –OH group R-C-R O H • Carbon that is attached to the –OH group is attached to 2 other carbon atoms 2 -Butanol

R TERTIARY ALCOHOL • Contain 1 –OH group R-C-R O H • Carbon attached to the –OH group is attached to three other carbon atoms 2 METHYL, 2 -BUTANOL

2 -OH groups are present Example: 1, 2 ethanediol

3 -OH groups are present Example: 1, 2, 3 propanetriol

• Functional Group = -COOH • Name: Hydrocarbon name, drop the final “e” and replace it with “–oic” followed by the word “acid” • Examples: 1. Ethanoic Acid 2. Butanoic Acid
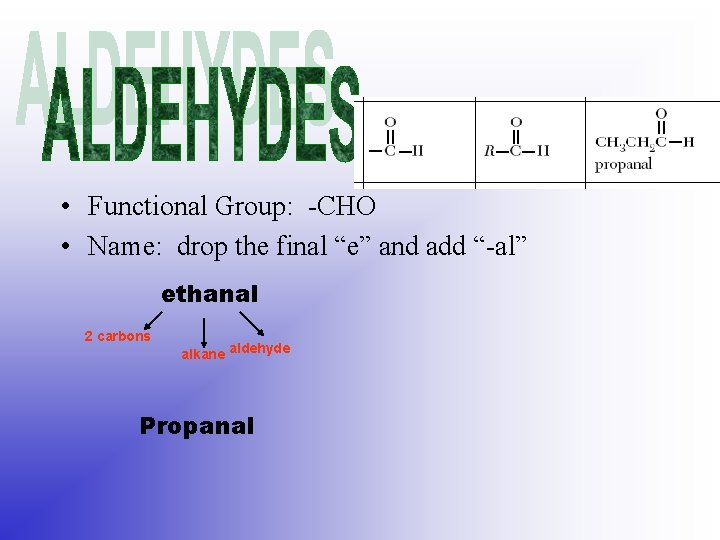
• Functional Group: -CHO • Name: drop the final “e” and add “-al” ethanal 2 carbons alkane aldehyde Propanal

• Functional Group: -CO • Name: Hydrocarbon name, drop the final “e”, add “one” Propanone 2 -Butanone

• Functional group: -O- (chain-O-chain) • Name: 1. Name each hydrocarbon chain – drop the ending add “yl” 2. Add “ether” to the end diethyl ether Ethyl methyl ether

• Functional Group: -COOR • Formed from a dehydration reaction of an alcohol and an acid • Fruity odor • Name: • Name each hydrocarbon chain – The part with the –CO gets the “-oate” ending – The other part gets the “-yl” ending

Ester Examples • Ethyl Methanoate • Ethyl Ethanoate

• Functional Group: NH 2 • Name the chain, add “amine” to the end ethanamine C 2 H 5 NH 2

• Functional Group: R-CONH 2 • Name: Name the chain, add “amide” to the end Butanamide Propanamide

• Functional Groups: -NH 2 and –COOH

• Just like an alcohol, but –SH instead of –OH Example: Methyl Thiol (CH 3 SH)

SUBSTITUTION ADDITION ESTERIFICATION SAPONIFICATION POLYMERIZATION COMBUSTION FERMENTATION

• Something takes the place of something else • Starts with a saturated hydrocarbon • 2 reactants, 2 products Ethane Chlorine

• • Start with an unsaturated hydrocarbon Break the double (or triple) bond Add in an atom (or group) 2 reactants, 1 product Ehtene Cl 2 1, 2 Dichloroethane
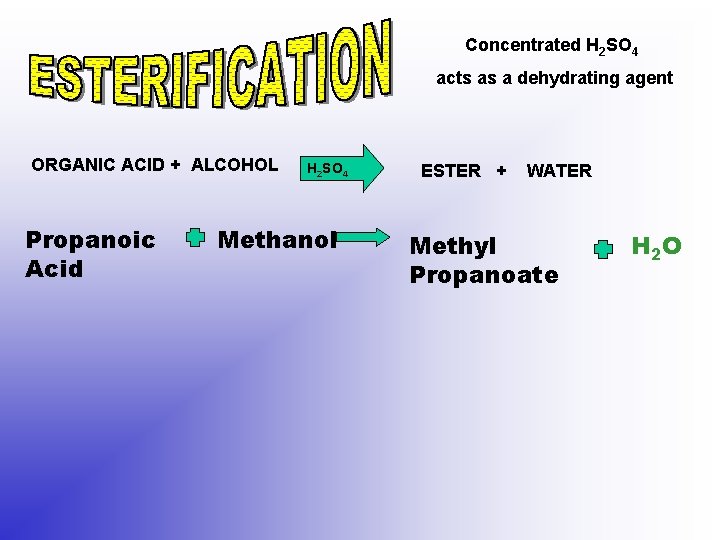
Concentrated H 2 SO 4 acts as a dehydrating agent ORGANIC ACID + ALCOHOL Propanoic Acid H 2 SO 4 Methanol ESTER + WATER Methyl Propanoate H 2 O

• Reverse of esterification, carried out in the presence of a base FAT (glycerol ester) Strong base (Na. OH or KOH) SOAP glycerol (trihydroxy alcohol) 1, 2, 3 propane triol
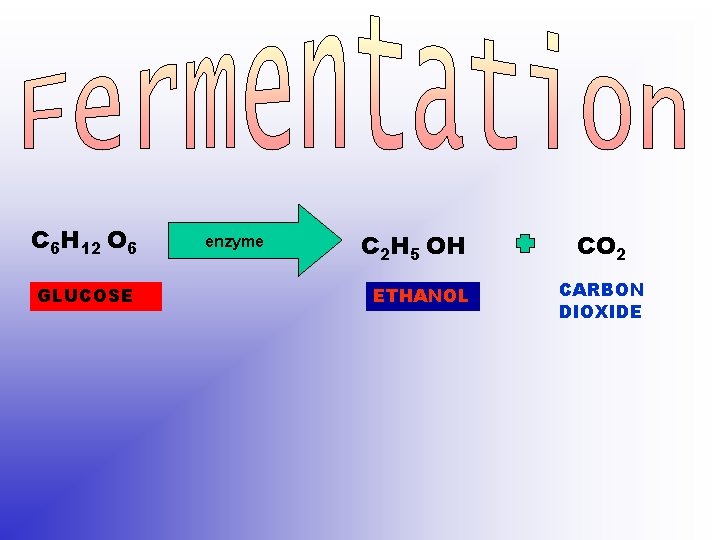
C 6 H 12 O 6 GLUCOSE enzyme C 2 H 5 OH ETHANOL CO 2 CARBON DIOXIDE
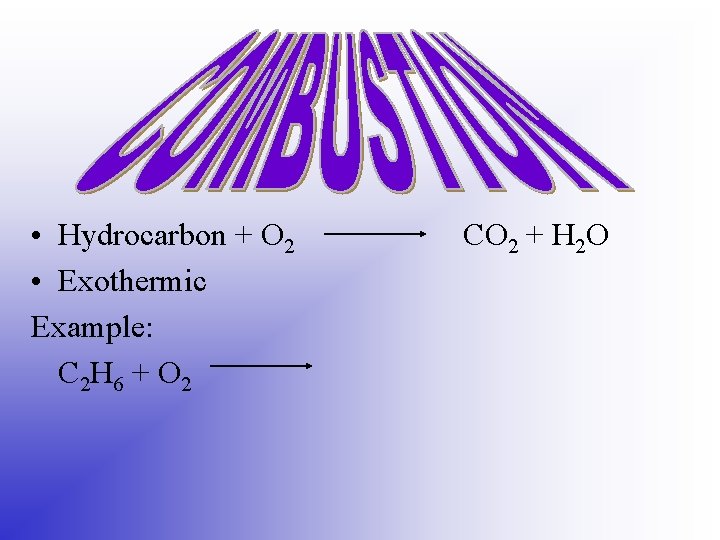
• Hydrocarbon + O 2 • Exothermic Example: C 2 H 6 + O 2 CO 2 + H 2 O

Poly. . . poly…poly. . . polymerization • Chains of small units make up a MACROMOLECULE • Examples: Plastics, proteins

Condensation Polymerization • Bonding of monomers by a dehydration reaction – OH groups of adjacent alcohols react, water is removed, the 2 molecules are connected by the remaining O • Examples: Nylon, Protein, Polyester

Addition Polymerization • Bonding of monomers of unsaturated compounds by “opening” a multiple bond in the carbon chain • Example: polyethylene and many plastics n C 2 H 4 (C 2 H 4)n
- Slides: 46