Organic Chemistry Classification of Hydrocarbons 24 1 Hydrocarbons
Organic Chemistry
Classification of Hydrocarbons 24. 1

Hydrocarbons • • Hydrocarbons are organic compounds that contain only hydrogen and carbon Types of hydrocarbons include § § Alkanes Alkenes Alkynes Aromatic 3

Alkanes • • Alkanes have only carbon to carbon single bonds Every carbon has four chemical bonds either to hydrogen or another carbon atom 4
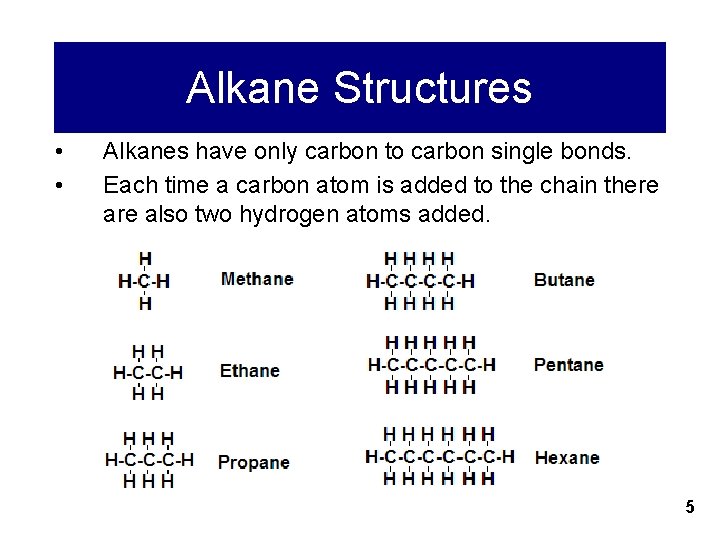
Alkane Structures • • Alkanes have only carbon to carbon single bonds. Each time a carbon atom is added to the chain there also two hydrogen atoms added. 5
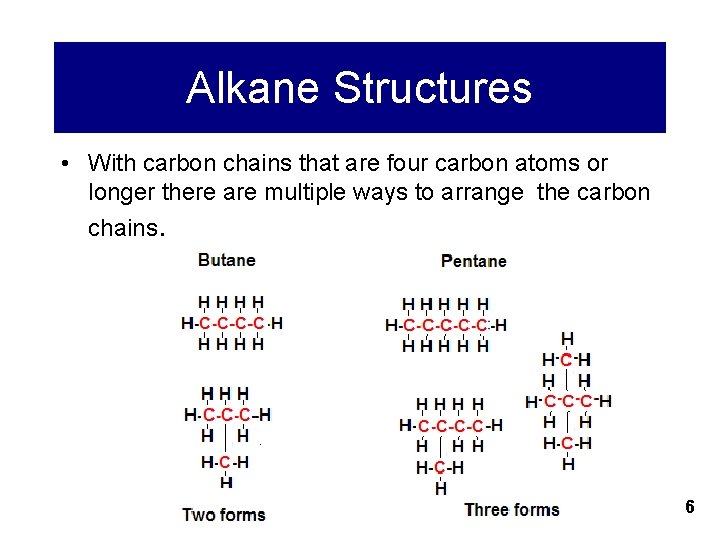
Alkane Structures • With carbon chains that are four carbon atoms or longer there are multiple ways to arrange the carbon chains. 6

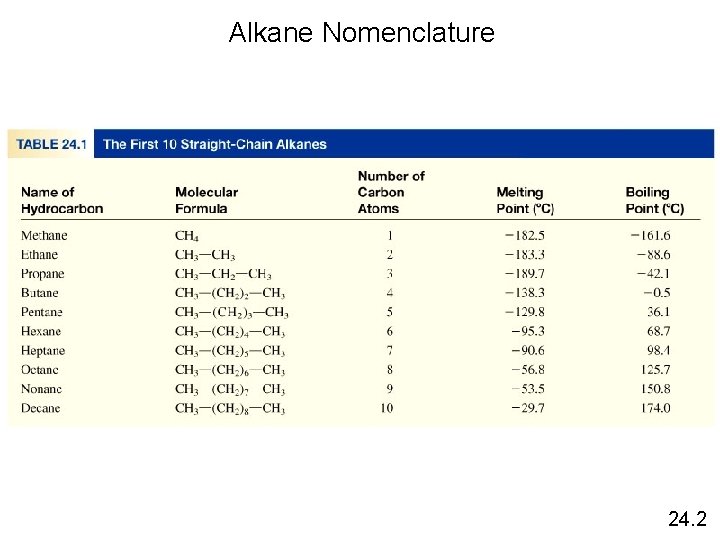
Alkanes have the general formula Cn. H 2 n+2 where n = 1, 2, 3, … • only single covalent bonds • saturated hydrocarbons because they contain the maximum number of hydrogen atoms that can bond with the number of carbon atoms in the molecule • a homologous series (Compounds that have the same general formula but differing lengths of carbon chains form a homologous series) CH 4 C 2 H 6 C 3 H 8 methane propane 24. 2

Naming Organic Compounds • • Originally compounds were named based on their source or use Many organic compounds were given common names which are still in use However many ambiguities resulted With the large number of organic compounds, a method for systematically naming them is very important 8

IUPAC Names • • • The International Union of Pure and Applied Chemists (IUPAC) developed a system for naming organic compounds. This system eliminated many of the ambiguities that plagued earlier naming systems Common names for many substances are still widely used 9

Naming Hydrocarbons using the IUPAC System A series of prefixes are used to designate the number of carbon atoms in a carbon chain meth 1 C hex 6 C eth 2 C hept 7 C prop 3 C oct 8 C but 4 C non 9 C pent 5 C dec 10 C 10

Naming Alkanes • • For straight chain hydrocarbons. The prefix indicates the number of carbon atoms. The suffix ane is added to designate that the compound is an alkane 11
Alkane Nomenclature 24. 2
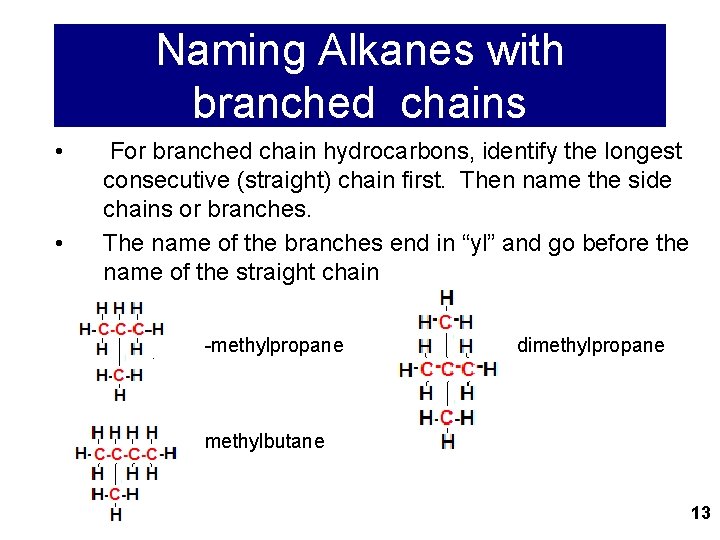
Naming Alkanes with branched chains • • For branched chain hydrocarbons, identify the longest consecutive (straight) chain first. Then name the side chains or branches. The name of the branches end in “yl” and go before the name of the straight chain -methylpropane dimethylpropane methylbutane 13

Alkane Nomenclature 1. The parent name of the hydrocarbon is that given to the longest continuous chain of carbon atoms in the molecule. CH 3 1 CH 2 2 CH 2 3 CH 4 CH 2 5 2. An alkane less one hydrogen atom is an alkyl group. 24. 2 CH 4 methane CH 3 methyl CH 2 6 CH 3 4 -methylheptane 7
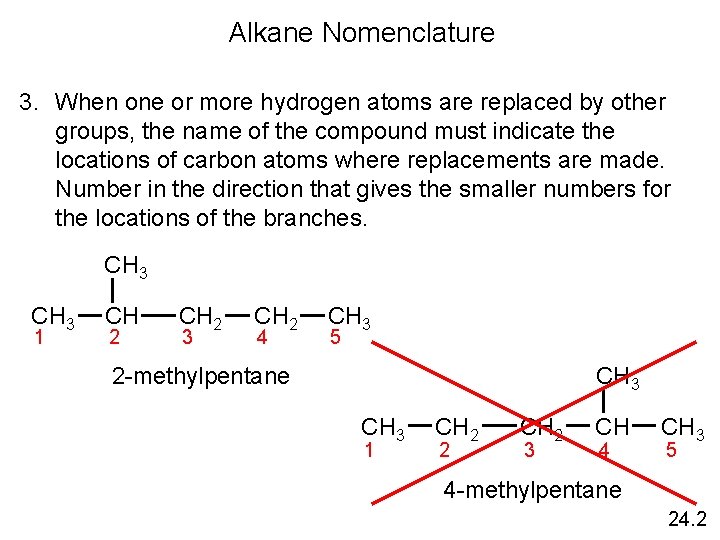
Alkane Nomenclature 3. When one or more hydrogen atoms are replaced by other groups, the name of the compound must indicate the locations of carbon atoms where replacements are made. Number in the direction that gives the smaller numbers for the locations of the branches. CH 3 1 CH 2 3 CH 2 4 CH 3 5 CH 3 2 -methylpentane CH 3 1 CH 2 2 CH 2 3 CH 4 CH 3 5 4 -methylpentane 24. 2
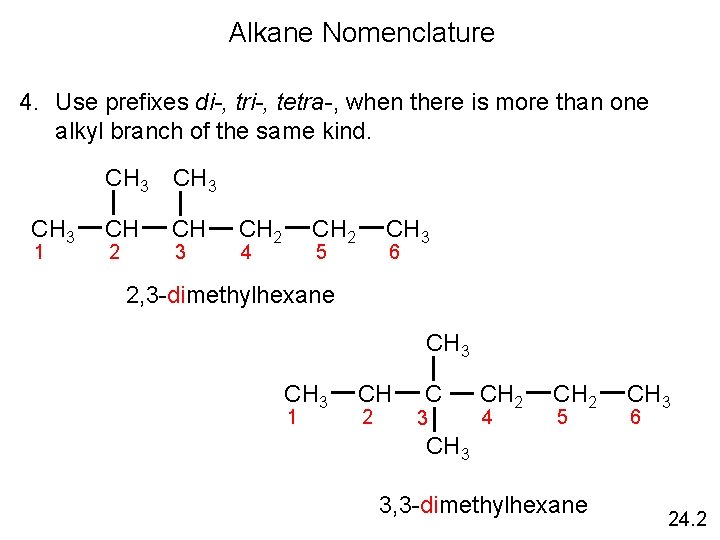
Alkane Nomenclature 4. Use prefixes di-, tri-, tetra-, when there is more than one alkyl branch of the same kind. CH 3 1 CH 3 CH CH 2 3 CH 2 4 CH 3 5 6 2, 3 -dimethylhexane CH 3 1 CH 2 C 3 CH 2 4 CH 2 5 CH 3 6 CH 3 3, 3 -dimethylhexane 24. 2

Alkane Nomenclature 5. Use previous rules for other types of substituents. Branches are placed alphabetically in the compound name. CH 3 1 Br NO 2 CH CH 2 3 CH 3 4 2 -bromo-3 -nitrobutane Br CH 2 1 NO 2 CH 2 2 CH 3 4 1 -bromo-3 -nitrobutane 24. 2
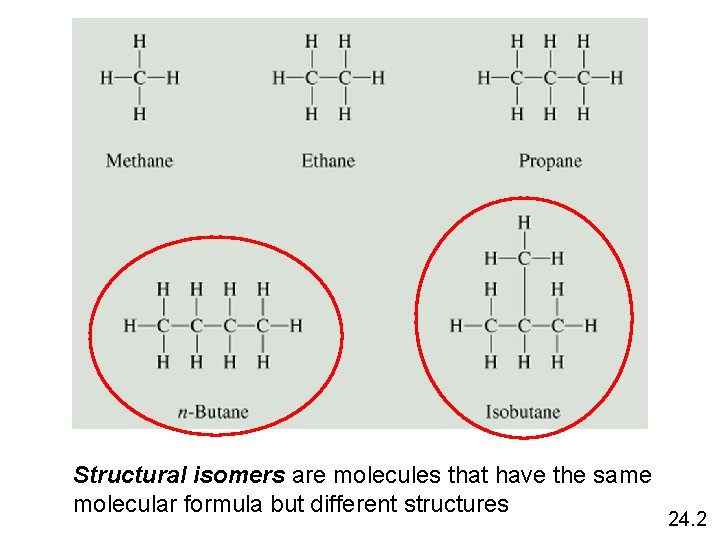
Structural isomers are molecules that have the same molecular formula but different structures 24. 2
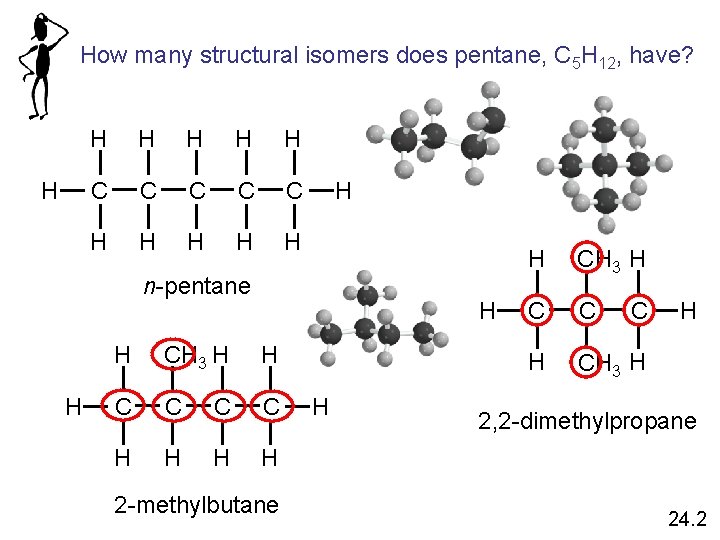
How many structural isomers does pentane, C 5 H 12, have? H H H C C C H H H n-pentane H H H CH 3 H H C C H H 2 -methylbutane H H CH 3 H C C H CH 3 H C H 2, 2 -dimethylpropane 24. 2

Alkenes • • • Alkenes have one (or more) carbon to carbon double bonds Since there are fewer hydrogen atoms in alkenes as a result of the double bond, alkenes are referred to as unsaturated. Alkanes on the other hand have the maximum number of hydrogen atoms. They are referred to as saturated. 20
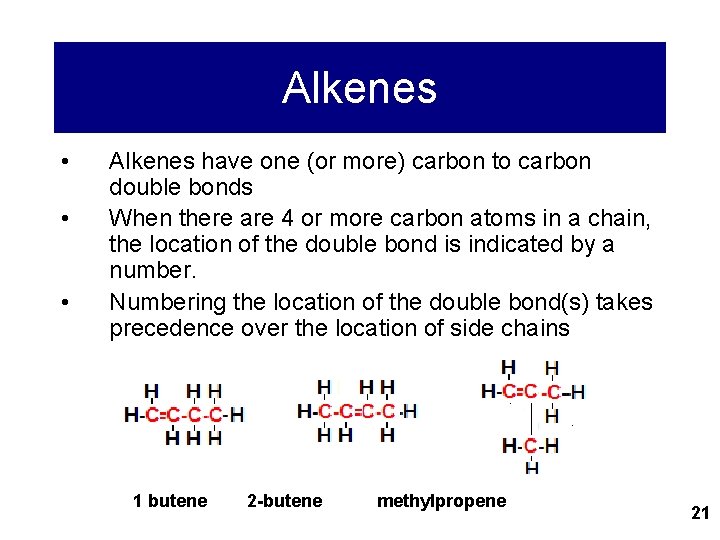
Alkenes • • • Alkenes have one (or more) carbon to carbon double bonds When there are 4 or more carbon atoms in a chain, the location of the double bond is indicated by a number. Numbering the location of the double bond(s) takes precedence over the location of side chains 1 butene 2 -butene methylpropene 21
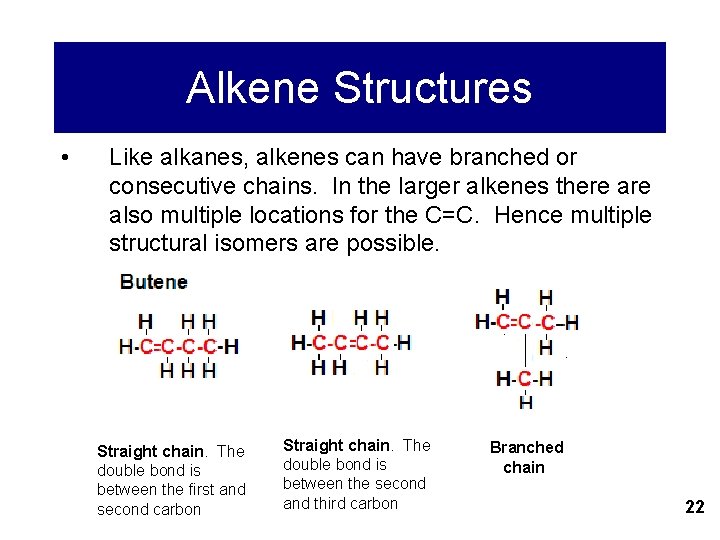
Alkene Structures • Like alkanes, alkenes can have branched or consecutive chains. In the larger alkenes there also multiple locations for the C=C. Hence multiple structural isomers are possible. Straight chain. The double bond is between the first and second carbon Straight chain. The double bond is between the second and third carbon Branched chain 22

Alkynes • • Alkynes have one (or more) carbon to carbon triple bonds Since there are fewer hydrogen atoms in alkynes as a result of the triple bond, alkynes like alkenes are referred to as unsaturated. 23

Alkynes • • When there are 4 or more carbon atoms in a chain, the location of the triple bond is indicated by a number. Numbering the location of the triple bond(s) takes precedence over the location of side chains 24
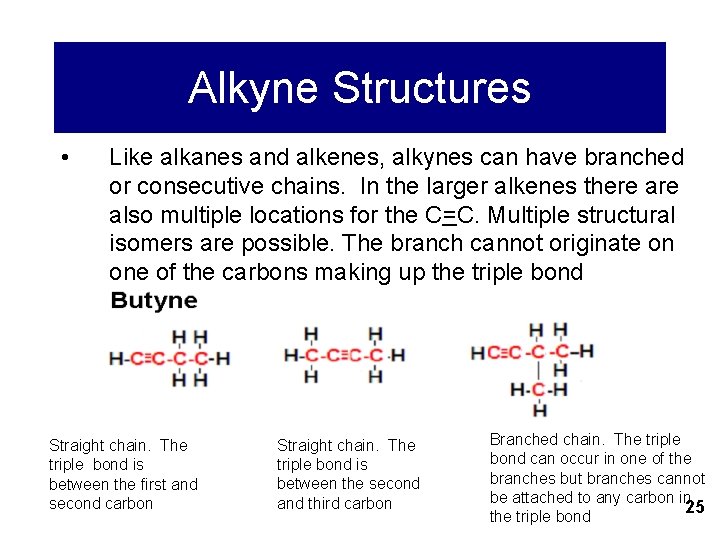
Alkyne Structures • Like alkanes and alkenes, alkynes can have branched or consecutive chains. In the larger alkenes there also multiple locations for the C=C. Multiple structural isomers are possible. The branch cannot originate on one of the carbons making up the triple bond Straight chain. The triple bond is between the first and second carbon Straight chain. The triple bond is between the second and third carbon Branched chain. The triple bond can occur in one of the branches but branches cannot be attached to any carbon in 25 the triple bond

Ring Structures • Hydrocarbons that exist in chains are known as aliphatic hydrocarbons • The ends of a chain may be joined to form a ring structure. • These compounds are known as cyclic structures 26

Saturated Cyclic Hydrocarbons • A number of the smaller alkanes exist as cyclic structures including – Cyclopropane – Cyclobutane – Cyclopentane – Cyclohexane 27

Saturated Cyclic Hydrocarbons Cyclic alkanes have the general formula Cn. H 2 n. The additional C-C bond results in the loss of two hydrogen atoms 28

Unsaturated Cyclic Hydrocarbons – • A few cyclic hydrocarbons may contain C=C double bonds. Two of the most common are cyclopentene and cyclohexene shown in the diagram at the left 29

Cyclic Hydrocarbons – Condensed structures • • Writing structural formulas for cyclic hydrocarbons can be tedious. These short form structures are commonly used. Each vertex represents a carbon atom and it is implied that there are enough H atoms on each vertex to make four bonds 30
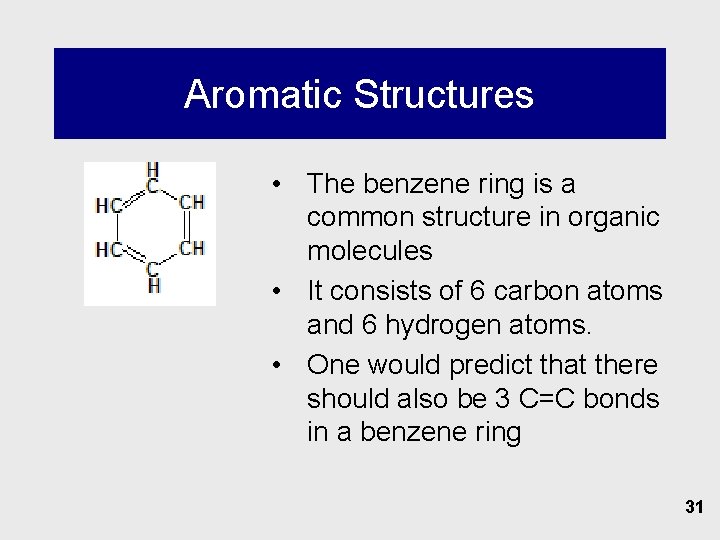
Aromatic Structures • The benzene ring is a common structure in organic molecules • It consists of 6 carbon atoms and 6 hydrogen atoms. • One would predict that there should also be 3 C=C bonds in a benzene ring 31
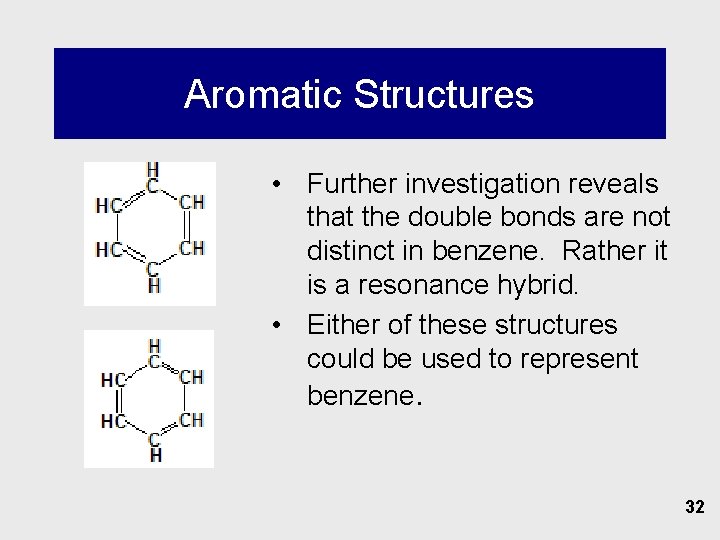
Aromatic Structures • Further investigation reveals that the double bonds are not distinct in benzene. Rather it is a resonance hybrid. • Either of these structures could be used to represent benzene. 32
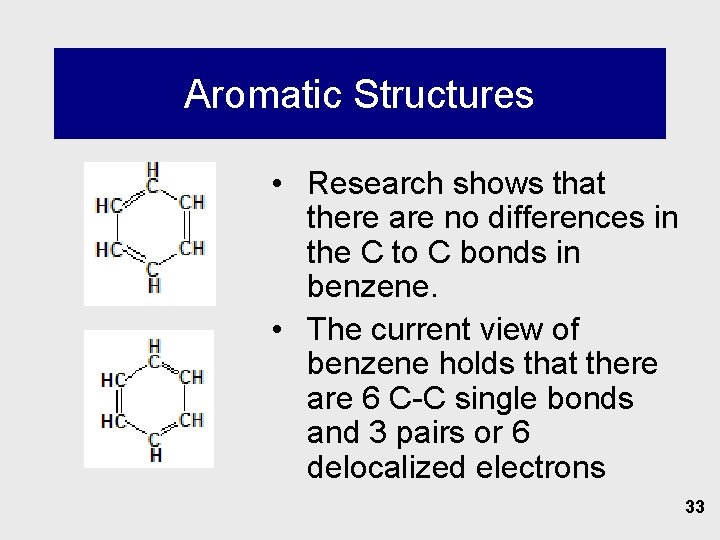
Aromatic Structures • Research shows that there are no differences in the C to C bonds in benzene. • The current view of benzene holds that there are 6 C-C single bonds and 3 pairs or 6 delocalized electrons 33
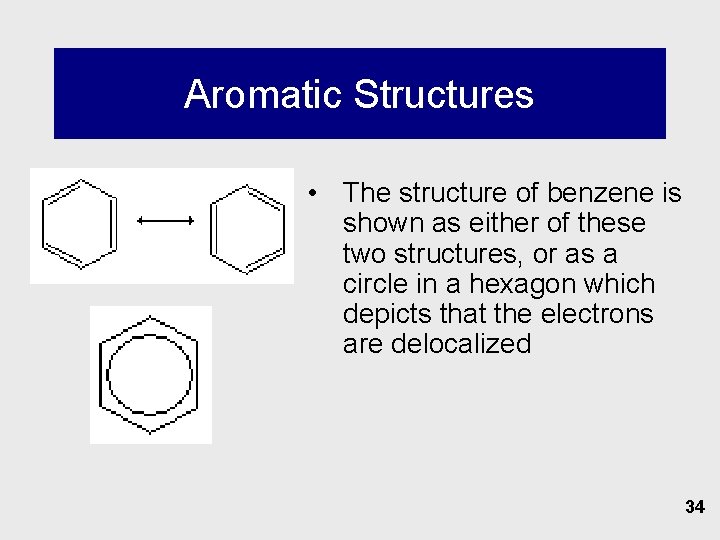
Aromatic Structures • The structure of benzene is shown as either of these two structures, or as a circle in a hexagon which depicts that the electrons are delocalized 34
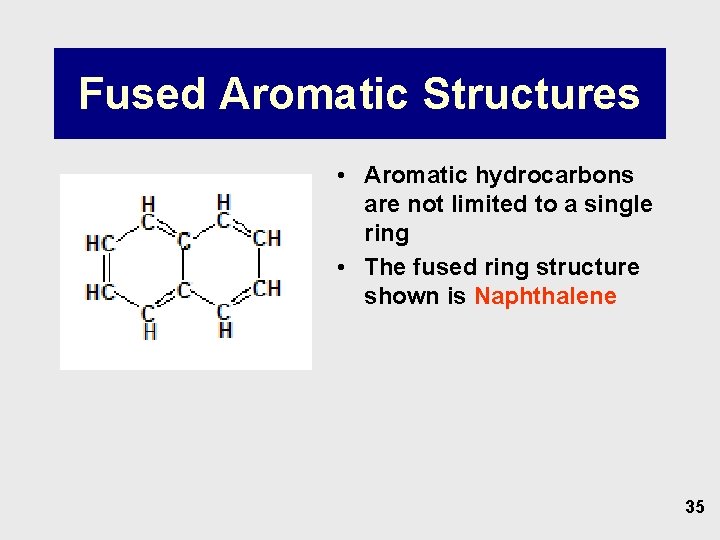
Fused Aromatic Structures • Aromatic hydrocarbons are not limited to a single ring • The fused ring structure shown is Naphthalene 35

Naming Compounds With Functional Groups • Various functional groups have unique suffixes that designate the functional group. • The functional group takes precedence in numbering the carbon chain. • Branches to the carbon chain are named in the usual manner. alcohols “ol” Amides “amide” Aldehydes “al” Amines “amine” or amino as a prefix Ketones “one” Ethers Ethoxy as prefix Acids “oic” halohydrocarbo Fluoro, bromo, chloro ns or iodo Esters “oate” 36
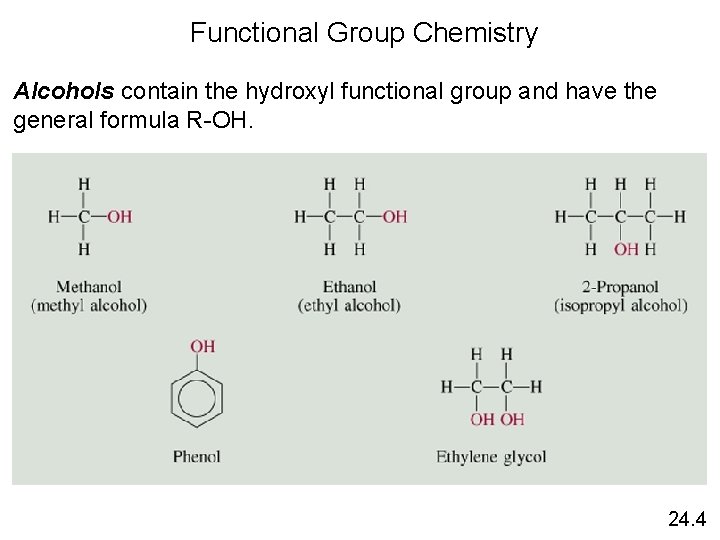
Functional Group Chemistry Alcohols contain the hydroxyl functional group and have the general formula R-OH. 24. 4

Alcohol Classification Alcohols may be classified as - Primary - Secondary - Tertiary Depending on whether the carbon atom that is attached to the –OH group is surrounded by one, two or three other carbon atoms
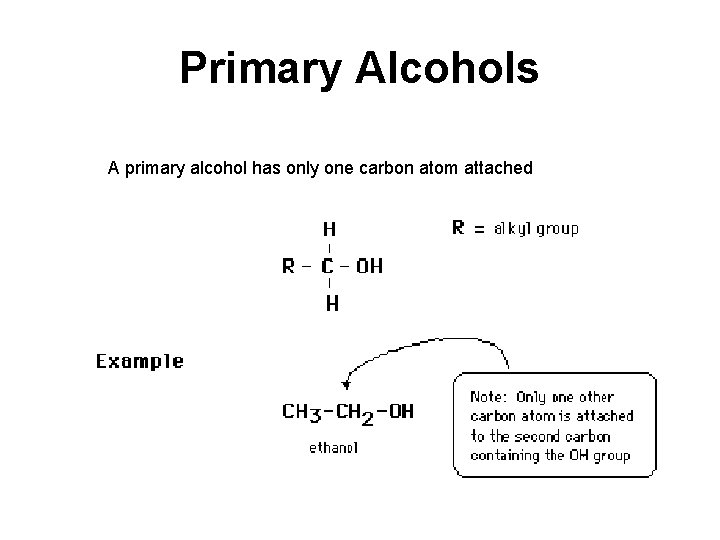
Primary Alcohols A primary alcohol has only one carbon atom attached

Secondary Alcohols A secondary alcohol has 2 carbon chains attached to the group on which the –OH resides
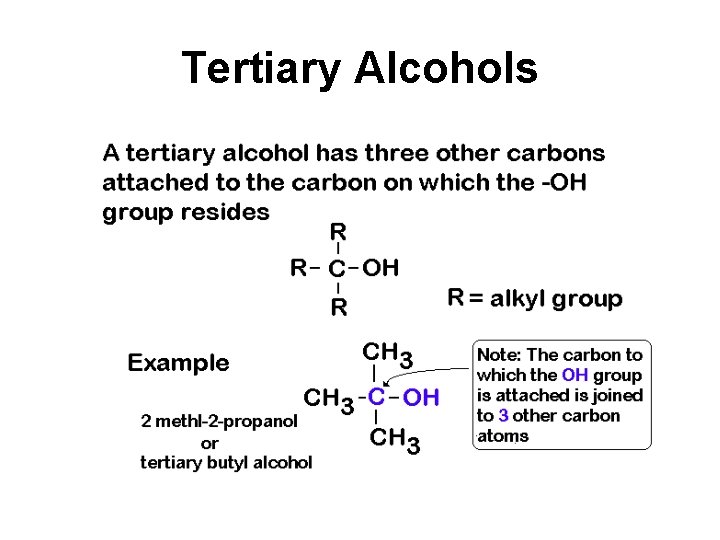
Tertiary Alcohols
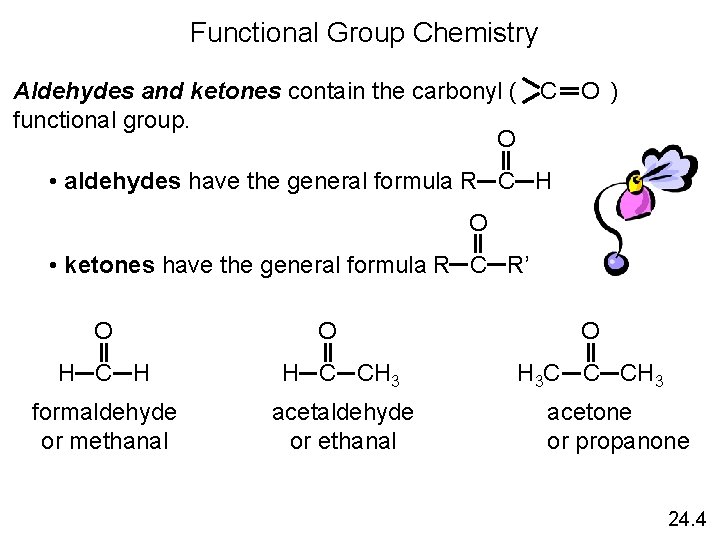
Functional Group Chemistry O Aldehydes and ketones contain the carbonyl ( ) C functional group. O • aldehydes have the general formula R C H O • ketones have the general formula R C R’ O O H C H H C CH 3 formaldehyde or methanal acetaldehyde or ethanal O H 3 C C CH 3 acetone or propanone 24. 4

Functional Group Chemistry Carboxylic acids contain the carboxyl ( -COOH ) functional group. methanoic acid butanoic acid benzoic acid 24. 4
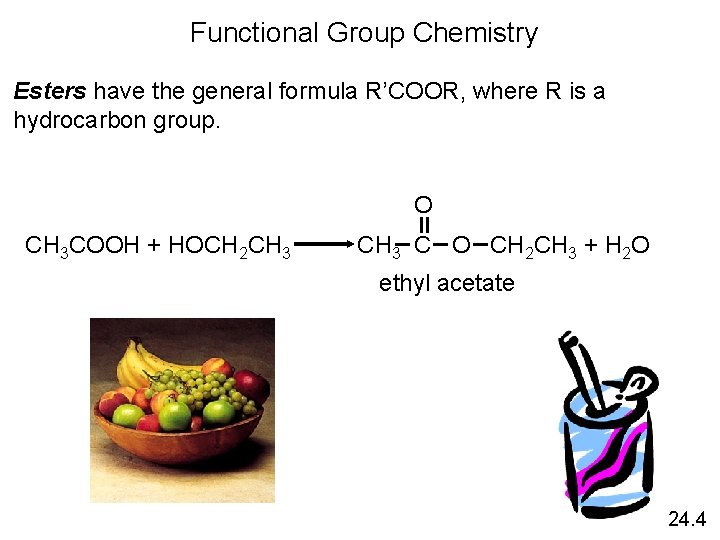
Functional Group Chemistry Esters have the general formula R’COOR, where R is a hydrocarbon group. O CH 3 COOH + HOCH 2 CH 3 C O CH 2 CH 3 + H 2 O ethyl acetate 24. 4

Alkane Reactions Combustion CH 4 (g) + 2 O 2 (g) CO 2 (g) + 2 H 2 O (l) DH 0 = -890. 4 k. J Halogenation - Also known as free radical substitution. • Many organic molecules undergo substitution reactions. • In a substitution reaction one atom or group of atoms is removed from a molecule and replaced with a different atom or group. Example: Cl 2 + CH 4 CH 3 Cl + HCl 24. 2

Three Basic Steps in a Free Radical Mechanism • Chain initiation The chain is initiated (started) by UV light breaking a chlorine molecule into free radicals. Cl 2 2 Cl. • Chain propagation reactions These are the reactions which keep the chain going. CH 4 + Cl. CH 3. + HCl CH 3. + Cl 2 CH 3 Cl + Cl. • Chain termination reactions These are reactions which remove free radicals from the system without replacing them by new ones. 2 Cl. Cl 2 CH 3. + Cl. CH 3 Cl CH 3. + CH 3 46
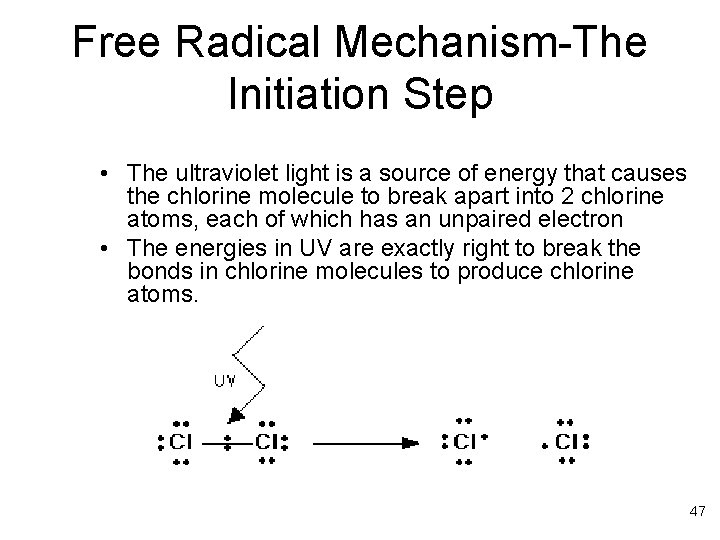
Free Radical Mechanism-The Initiation Step • The ultraviolet light is a source of energy that causes the chlorine molecule to break apart into 2 chlorine atoms, each of which has an unpaired electron • The energies in UV are exactly right to break the bonds in chlorine molecules to produce chlorine atoms. 47
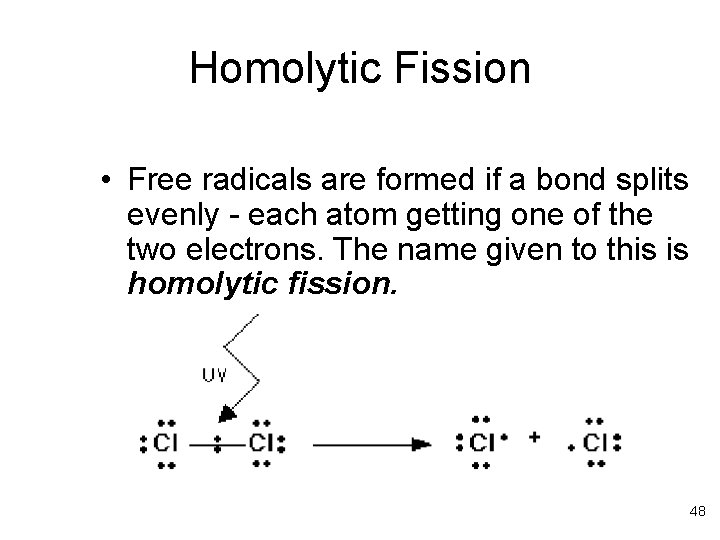
Homolytic Fission • Free radicals are formed if a bond splits evenly - each atom getting one of the two electrons. The name given to this is homolytic fission. 48

Free Radical Propagation • The productive collision happens if a chlorine radical hits a methane molecule. • The chlorine radical removes a hydrogen atom from the methane. That hydrogen atom only needs to bring one electron with it to form a new bond to the chlorine, and so one electron is left behind on the carbon atom. A new free radical is formed - this time a methyl radical, CH 3. 49

Free Radical Propagation II • If a methyl radical collides with a chlorine molecule the following occurs: CH. + Cl CH Cl + Cl. • • • 3 2 3 The methyl radical takes one of the chlorine atoms to form chloromethane In the process generates another chlorine free radical. This new chlorine radical can now go through the whole sequence again, It will produce yet another chlorine radical - and so on. 50

Termination Steps • The free radical propagation does not go on for ever. • If two free radicals collide the reaction is terminated. 2 Cl. Cl 2 CH 3. + Cl. CH 3 Cl CH 3. + CH 3 51

Exercise • Write the steps in the free radical mechanism for the reaction of chlorine with methyl benzene. The overall reaction is shown below. The methyl group is the part of methyl benzene that undergoes attack. 52
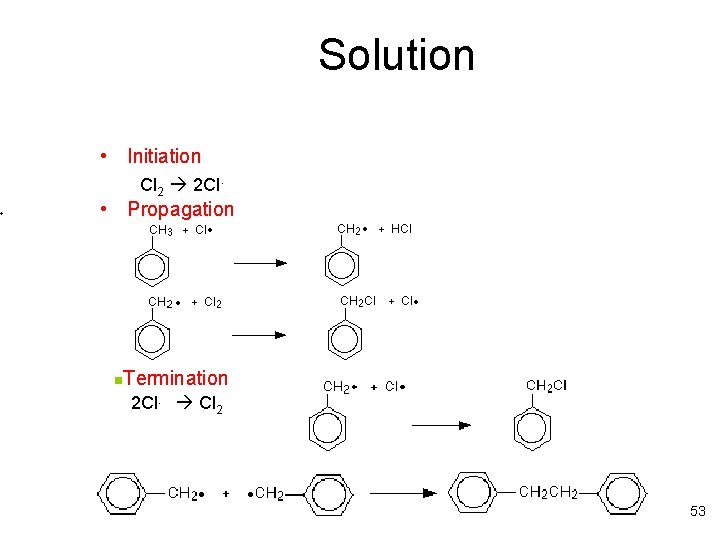
Solution • Initiation Cl 2 2 Cl. • Propagation Termination 2 Cl. Cl 2 n 53
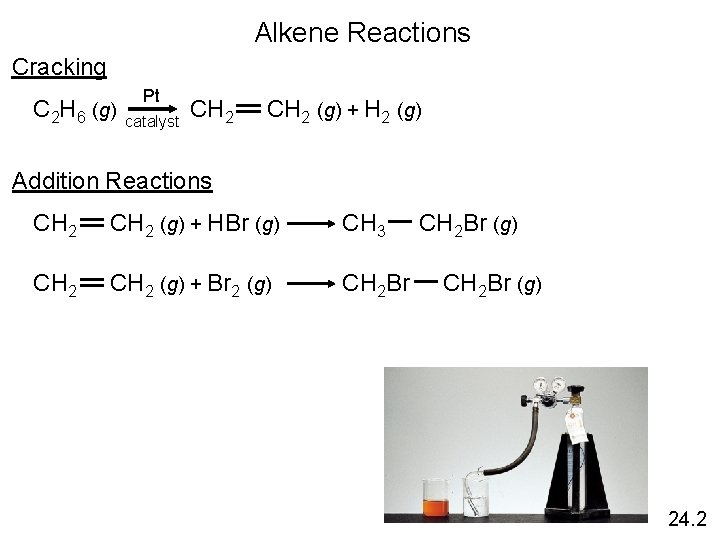
Alkene Reactions Cracking Pt C 2 H 6 (g) CH 2 (g) + H 2 (g) catalyst Addition Reactions CH 2 (g) + HBr (g) CH 3 CH 2 Br (g) CH 2 (g) + Br 2 (g) CH 2 Br (g) 24. 2
- Slides: 54