Macromolecules Organic Compounds Organic compounds compounds that contain






















- Slides: 41

Macromolecules = Organic Compounds • Organic compounds – compounds that contain carbon – Most Common Elements in living things: C, H, O, N, P, S • We will study: – – Carbohydrates Lipids Proteins Nucleic Acids

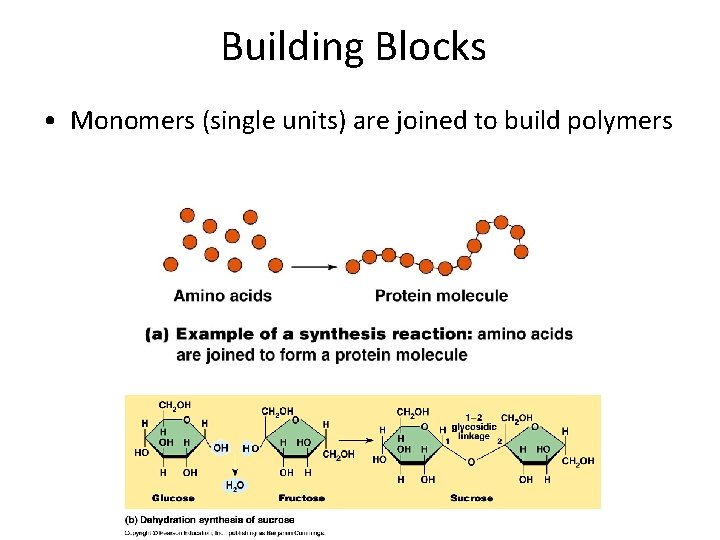
Building Blocks • Monomers (single units) are joined to build polymers

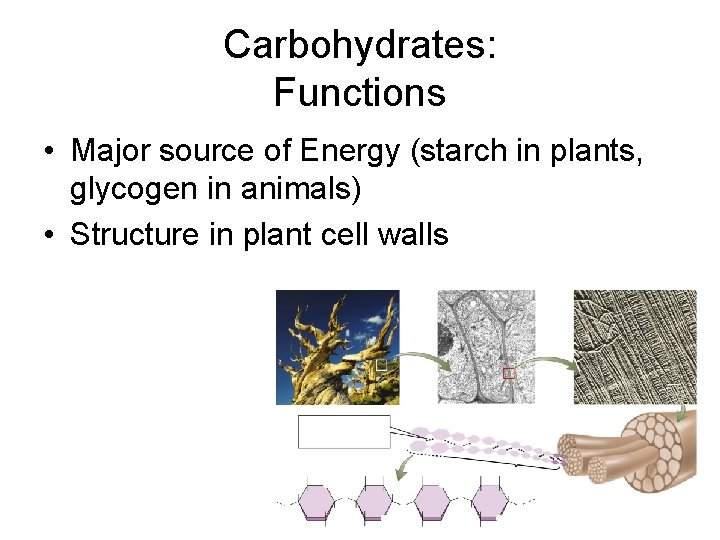
Carbohydrates: Functions • Major source of Energy (starch in plants, glycogen in animals) • Structure in plant cell walls

Carbohydrates: Where are they found? • • Wheat Breads Pasta Sugars (candy, cakes, etc. ) Fruits Vegetables Most are plant-based

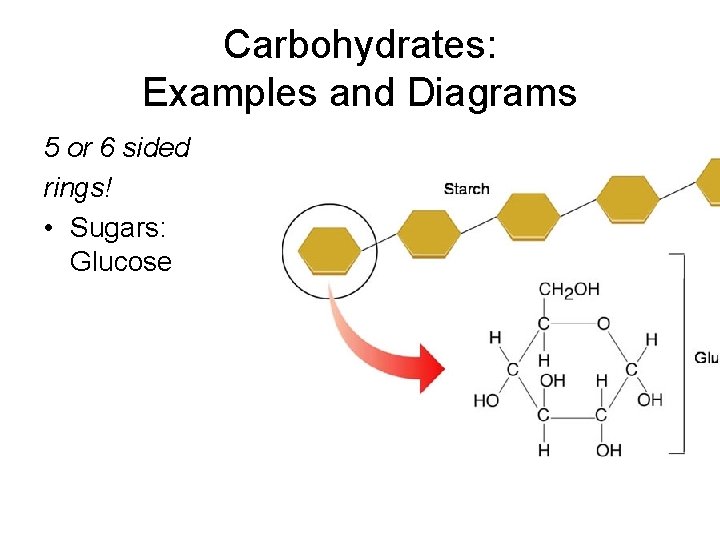
Carbohydrates: Elements and Building Blocks • Structure: 5 or 6 carbon ring • Elements: C, H, O • Monomers – monosaccharide (1 ring sugar) ex: glucose C 6 H 12 O 6

Carbohydrates: Examples and Diagrams 5 or 6 sided rings! • Sugars: Glucose

Polymer=monomers bonded together • Polymers = polysaccharide (multi-ring sugar) - glycogen (animal-stored energy in the liver) - starch (plant-stored energy) - cellulose (plant cell walls) - chitin (in exoskeleton or shell or insects)

Do now 1) Name the function of carbohydrates 2) Which foods provide us with carbs? 3) What is the monomer of carbs? What is an example? 4) Name 3 polymers of carbs and draw what they look like

Lipids (fats) Function: 1) 2) 3) 4) 5) Stores energy for later use Insulate the body to keep you warm Build cell membrane structure Wax on leaves: protective coating Steroids: hormones/cholesterol

Location: Where do I find Lipids? • Foods: butter, oils, salad dressing • Plants: in the seeds • Animals: in the connective tissues • Cells: Cell membrane

Structure of Lipids • Elements: C, H, O Large non-polar molecules that DO NOT dissolve in water. Why?

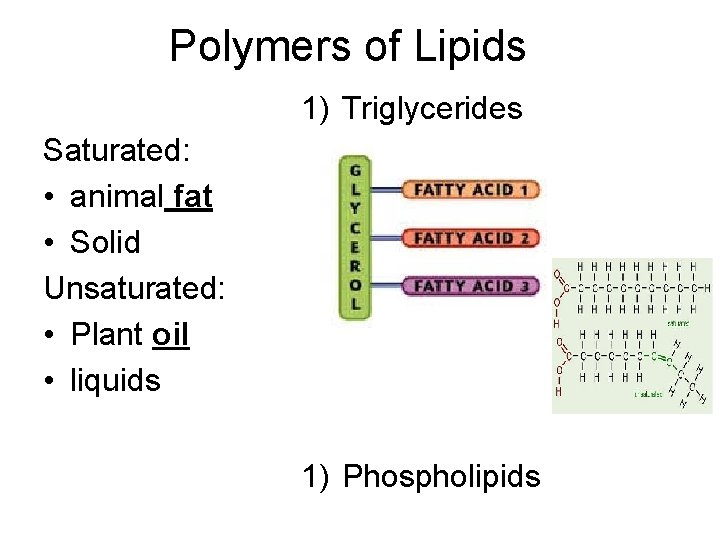
Building Blocks • Monomers – fatty acids and glycerol • Polymers – triglycerides (commonly fats, oils, waxes), phospholipids, steroids

Polymers of Lipids 1) Triglycerides Saturated: • animal fat • Solid Unsaturated: • Plant oil • liquids 1) Phospholipids

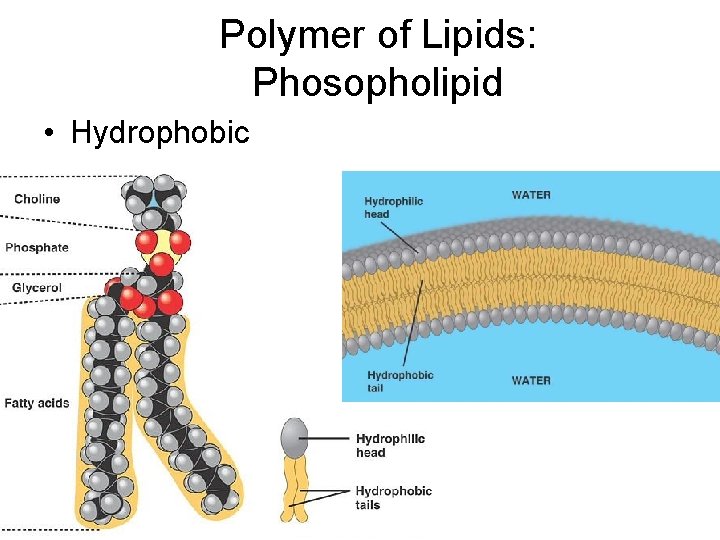
Polymer of Lipids: Phosopholipid • Hydrophobic

Do Now 1) Name some functions of lipids 2) Which foods provide us with lipids? 3) What are two monomers of lipids 4) Name 2 polymers of lipids. 5) What are phospholipids for? Is the tail hydrophobic or hydrophilic? Is the head hydrophobic or hydrophilic?

Proteins Functions: – Builds strong muscles – Collagen builds strong bones and connective tissue – Keratin builds strong skin, hair, and nails – Antibodies fight disease – Transport oxygen in blood in hemoglobin – ENZYMES reduce the time needed for reaction to start

Proteins • Location: Where do I get proteins? Animal products: meat, fish, dairy, eggs

Structural Proteins and Functional Proteins • Some proteins are structural and provide support in hair, horns, spider webs, etc. Fig. 2 -21

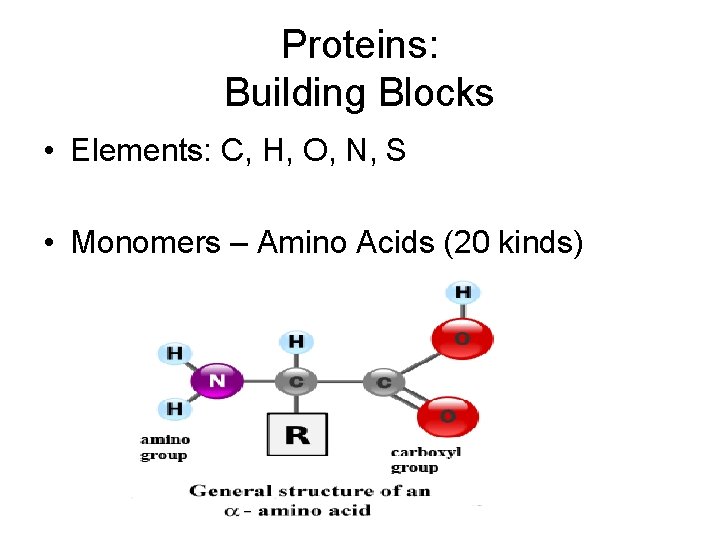
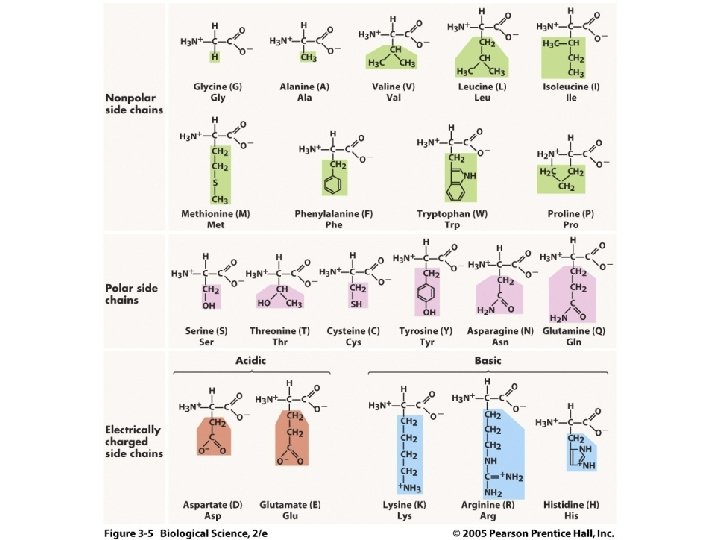
Proteins: Building Blocks • Elements: C, H, O, N, S • Monomers – Amino Acids (20 kinds)


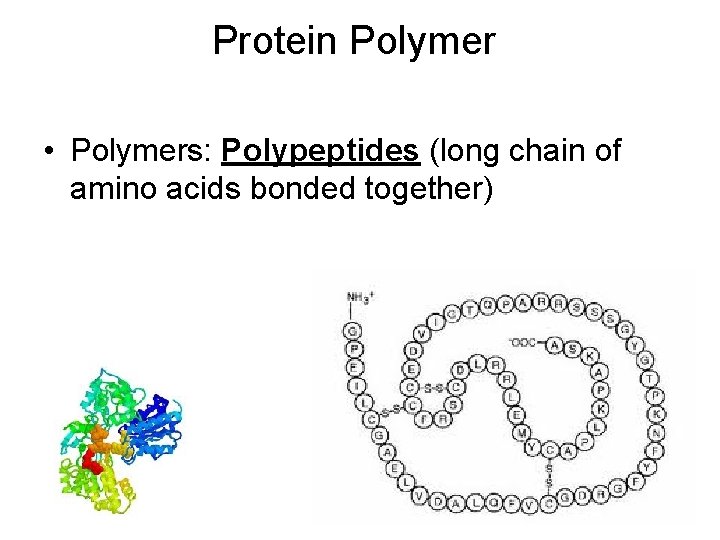
Protein Polymer • Polymers: Polypeptides (long chain of amino acids bonded together)

Do Now Review • • What are some functions of proteins? Which foods are high in protein? What is the polymer of proteins? What is the monomer of proteins? – How many different ones are there?

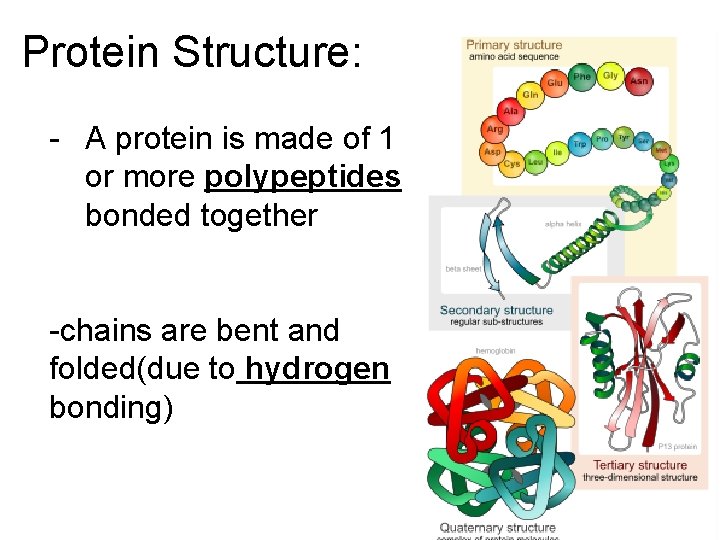
Protein Structure: - A protein is made of 1 or more polypeptides bonded together -chains are bent and folded(due to hydrogen bonding)

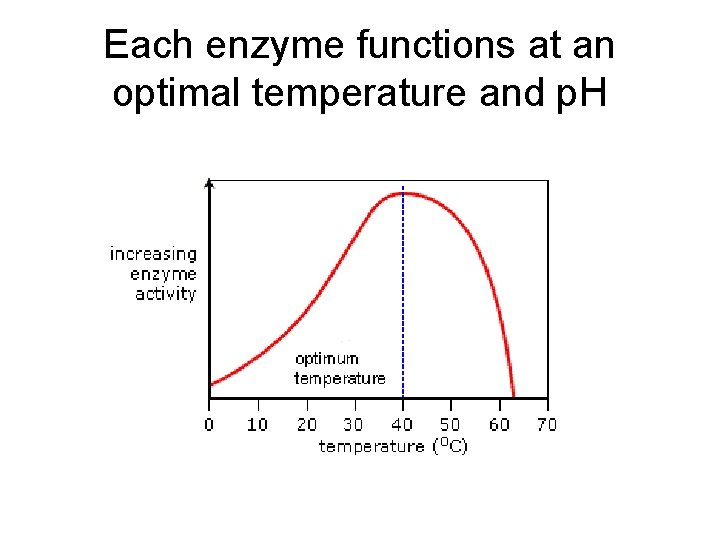
Protein Structure • Each protein has a special shape which determines its function • Temperature and p. H changes the protein’s shape so it doesn’t work anymore(denatured)

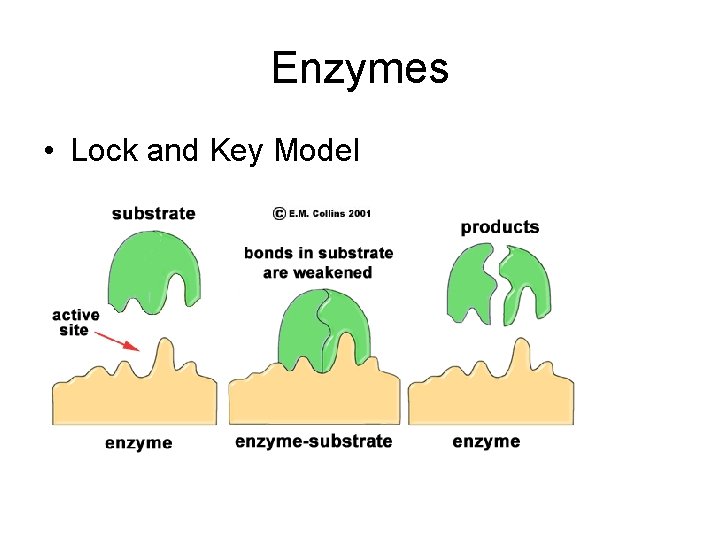
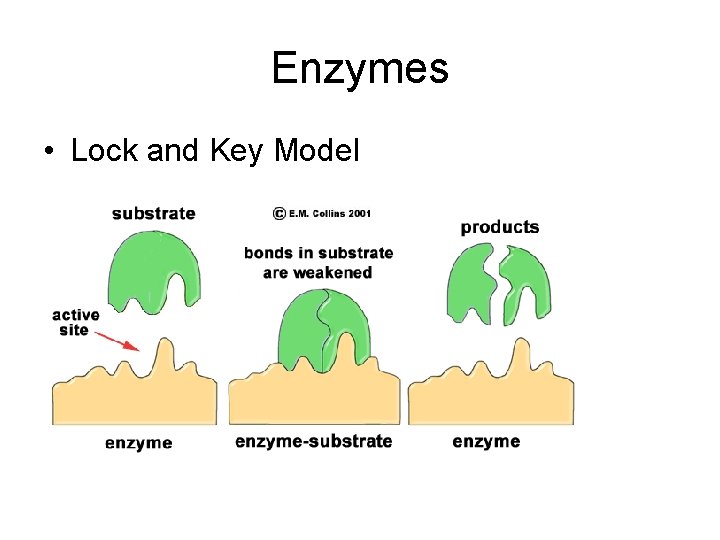
Enzymes • Lock and Key Model

Nucleic acids Function: –Store and transmit genetic information –Tells cells when and what proteins to make

Nucleic Acids Elements: CHONP

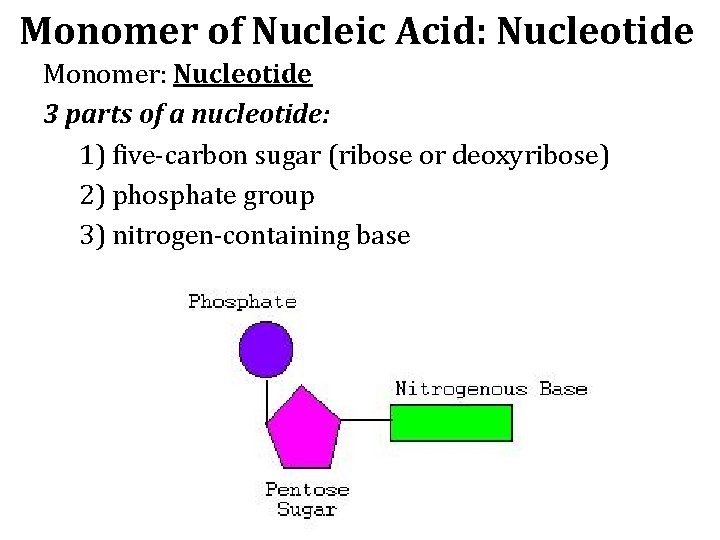
Monomer of Nucleic Acid: Nucleotide Monomer: Nucleotide 3 parts of a nucleotide: 1) five-carbon sugar (ribose or deoxyribose) 2) phosphate group 3) nitrogen-containing base

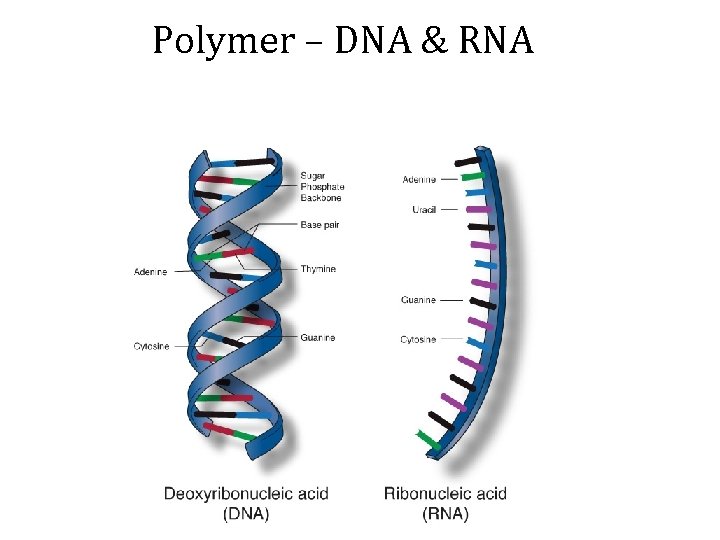
Polymer – DNA & RNA

DNA - Deoxyribonucleic Acid - Double strand of nucleotides - Contains the genetic code of cell - Found in nucleus Bases: Adenine (A) <==> Thymine (T) (apple tree) Cytosine (C) <==> Guanine (G)

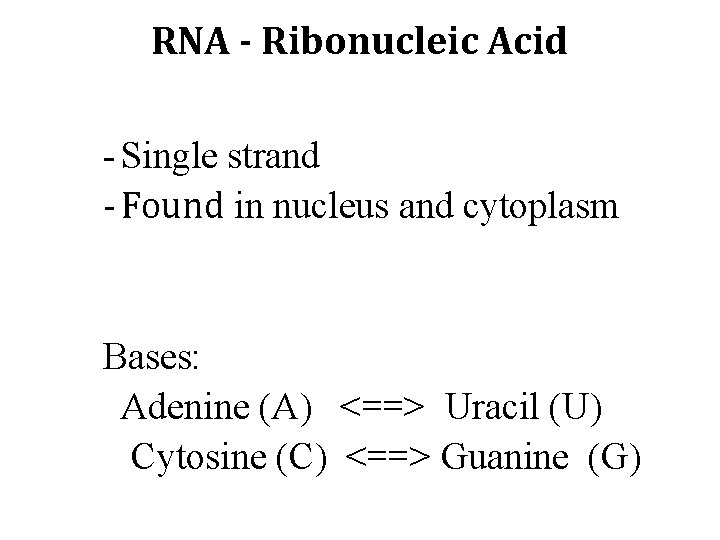
RNA - Ribonucleic Acid - Single strand - Found in nucleus and cytoplasm Bases: Adenine (A) <==> Uracil (U) Cytosine (C) <==> Guanine (G)

Nucleic Acids: Other Facts • In ALL cells

Done! 4 Macromolecules: 1) Carbohydrates 1) Lipids 1) Proteins 1) Nucleic Acids

Enzymes • Lock and Key Model

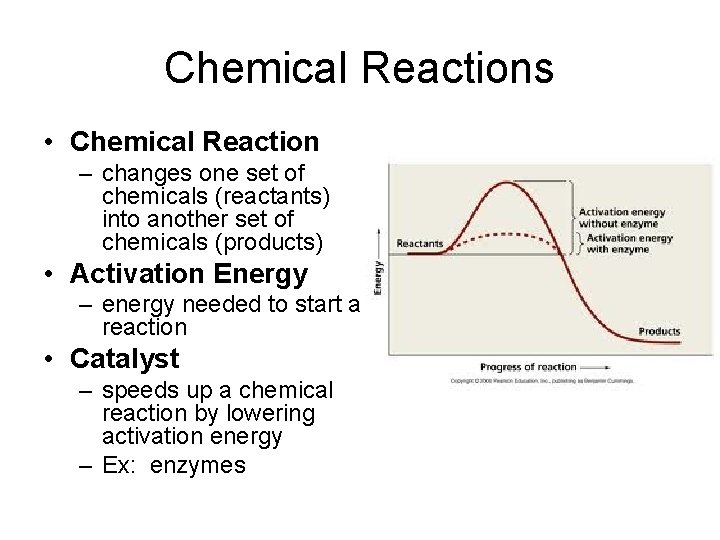
Chemical Reactions • Chemical Reaction – changes one set of chemicals (reactants) into another set of chemicals (products) • Activation Energy – energy needed to start a reaction • Catalyst – speeds up a chemical reaction by lowering activation energy – Ex: enzymes

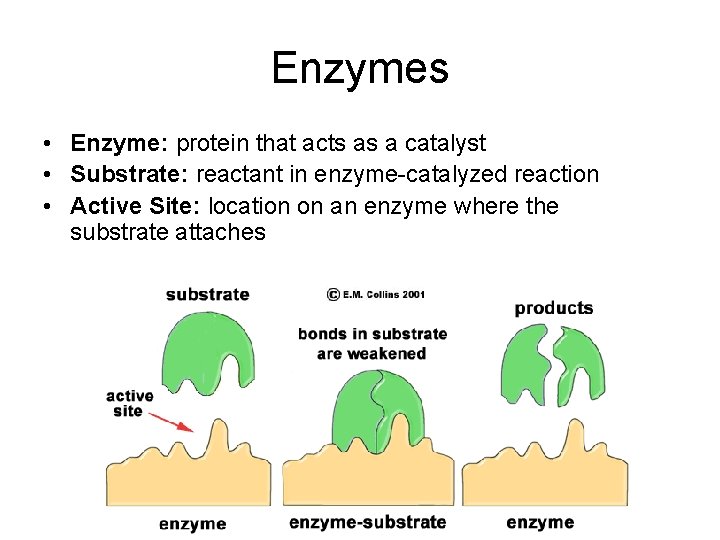
Enzymes • Enzyme: protein that acts as a catalyst • Substrate: reactant in enzyme-catalyzed reaction • Active Site: location on an enzyme where the substrate attaches


Each enzyme functions at an optimal temperature and p. H

Sample MCAS Question In the nucleus of a human cell, RNA polymerase travels along a DNA strand constructs a new strand of m. RNA. The new m. RNA strand leaves the nucleus through a pore in the nuclear membrane and enters the cytoplasm. The m. RNA associates with a ribosome and a new polypeptide is produced. Several types of organic molecules are mentioned in the paragraph above. a. Select two different organic molecules mentioned in the paragraph above and classify each as one of the four major types of organic molecules. You may use a table like the one below in your response. b. Briefly describe the structure and function of each organic molecule you identified in part (a. ). You may use a table like the one below in your response.

Answer Molecule Classification Structure Function

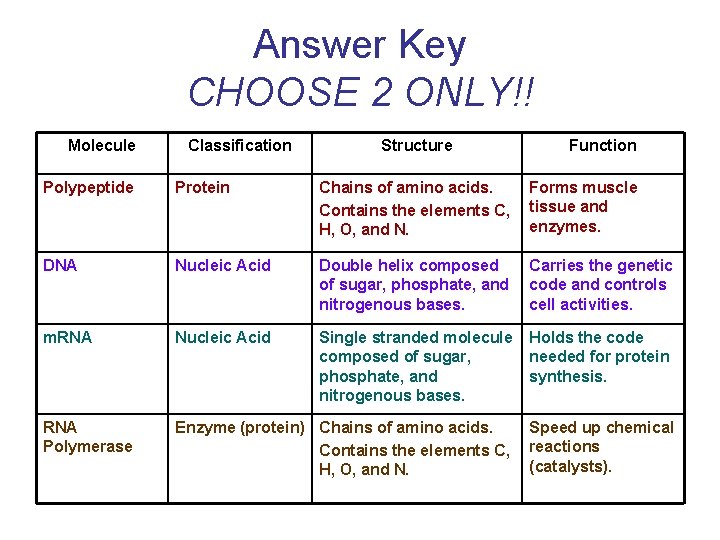
Answer Key CHOOSE 2 ONLY!! Molecule Classification Structure Function Polypeptide Protein Chains of amino acids. Contains the elements C, H, O, and N. Forms muscle tissue and enzymes. DNA Nucleic Acid Double helix composed of sugar, phosphate, and nitrogenous bases. Carries the genetic code and controls cell activities. m. RNA Nucleic Acid Single stranded molecule composed of sugar, phosphate, and nitrogenous bases. Holds the code needed for protein synthesis. RNA Polymerase Enzyme (protein) Chains of amino acids. Contains the elements C, H, O, and N. Speed up chemical reactions (catalysts).