Metabolic Changes of Drugs and Related Organic Compounds

Metabolic Changes of Drugs and Related Organic Compounds 3 rd stage/ 1 st semester Lecture 8 Shokhan J. Hamid

Acetylation • Acetylation constitutes an important metabolic route for drugs containing primary amino groups. • This encompasses primary aromatic amines (Ar. NH 2), sulfonamides (H 2 NC 6 H 4 SO 2 NHR), hydrazines (—NHNH 2), hydrazides (—CONHNH 2), and primary aliphatic amines. • The amide derivatives formed from acetylation of these amino functionalities are generally inactive and nontoxic. • The primary function of acetylation is to terminate pharmacological activity and detoxification. 2

• The acetyl group used in N-acetylation of xenobiotics is supplied by acetyl-Co. A. • Transfer of the acetyl group from this cofactor to the accepting amino substrate is carried out by soluble N-acetyltransferases present in hepatic reticuloendothelial cells. • Aromatic amine metabolites resulting from the reduction of aryl nitro compounds also are N-acetylated. • For example, the anticonvulsant clonazepam undergoes nitro reduction to its 7 -amino metabolite, which in turn is Nacetylated. 3
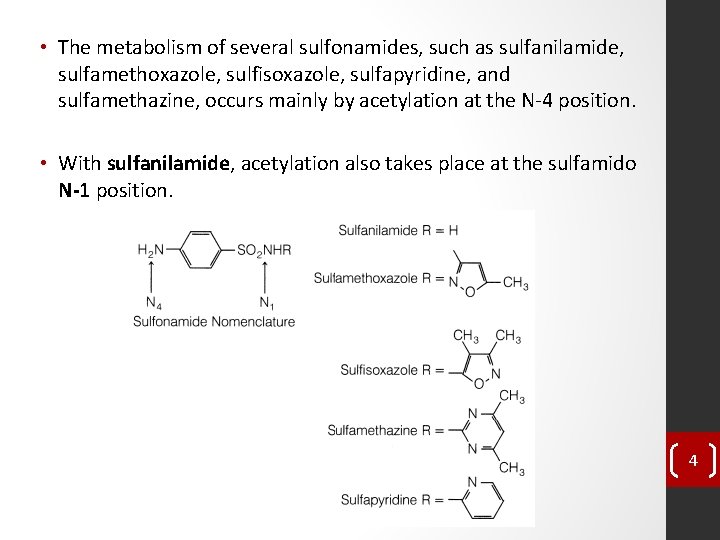
• The metabolism of several sulfonamides, such as sulfanilamide, sulfamethoxazole, sulfisoxazole, sulfapyridine, and sulfamethazine, occurs mainly by acetylation at the N-4 position. • With sulfanilamide, acetylation also takes place at the sulfamido N-1 position. 4
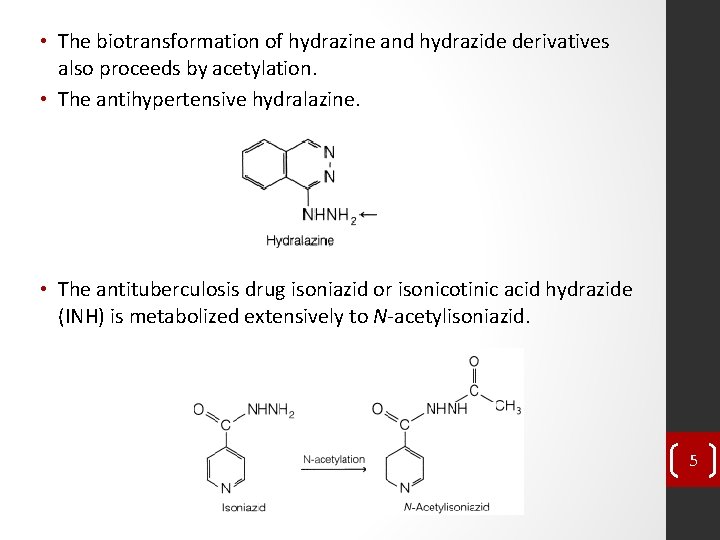
• The biotransformation of hydrazine and hydrazide derivatives also proceeds by acetylation. • The antihypertensive hydralazine. • The antituberculosis drug isoniazid or isonicotinic acid hydrazide (INH) is metabolized extensively to N-acetylisoniazid. 5
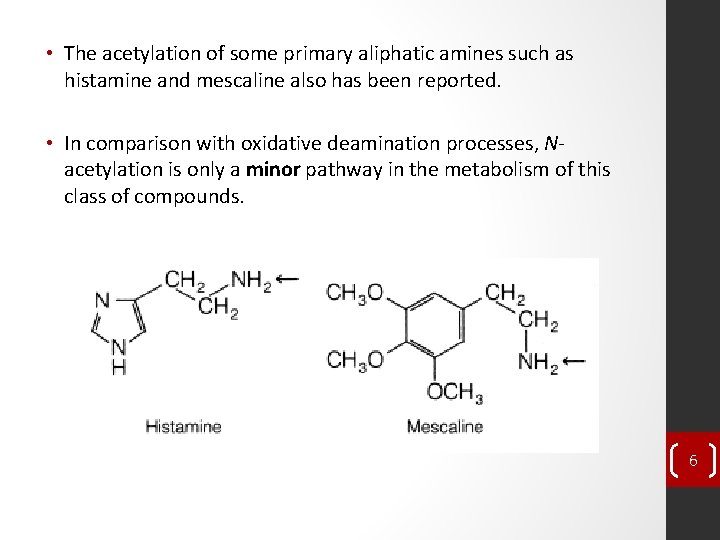
• The acetylation of some primary aliphatic amines such as histamine and mescaline also has been reported. • In comparison with oxidative deamination processes, Nacetylation is only a minor pathway in the metabolism of this class of compounds. 6

• The acetylation pattern of several drugs (e. g. , isoniazid, hydralazine) in the human population displays a bimodal character in which the drug is conjugated either rapidly or slowly with acetyl-Co. A. • This phenomenon is termed acetylation polymorphism. • Individuals are classified as having either slow or rapid acetylator phenotypes. • This variation in acetylating ability is genetic and is caused mainly by differences in N-acetyltransferase activity. • The proportion of rapid and slow acetylators varies widely among different ethnic groups throughout the world. • a high proportion of Eskimos and Asians are rapid acetylators, whereas Egyptians and some Western European groups are mainly slow acetylators. • Other populations are intermediate between these two extremes. 7

• Because of the bimodal distribution of the human population into rapid and slow acetylators, there appears to be significant individual variation in therapeutic and toxicological responses to drugs displaying acetylation polymorphism. • Slow acetylators seem more likely to develop adverse reactions, whereas rapid acetylators are more likely to show an inadequate therapeutic response to standard drug doses. • The antituberculosis drug isoniazid illustrates many of these points. • The plasma half-life of isoniazid in rapid acetylators ranges from 45 to 80 minutes; in slow acetylators, the half-life is about 140 to 200 minutes. 8

Methylation • Methylation reactions play an important role in the inactivation of numerous physiologically active biogenic amines (e. g. , norepinephrine, dopamine, serotonin, and histamine). • Methylation, however, constitutes only a minor pathway for conjugating drugs and xenobiotics. • Most methylated products tend to be pharmacologically inactive. • The coenzyme involved in methylation reactions is Sadenosylmethionine (SAM). • The transfer of the activated methyl group from this coenzyme to the acceptor substrate is catalyzed by various cytoplasmic and microsomal methyltransferases. 9
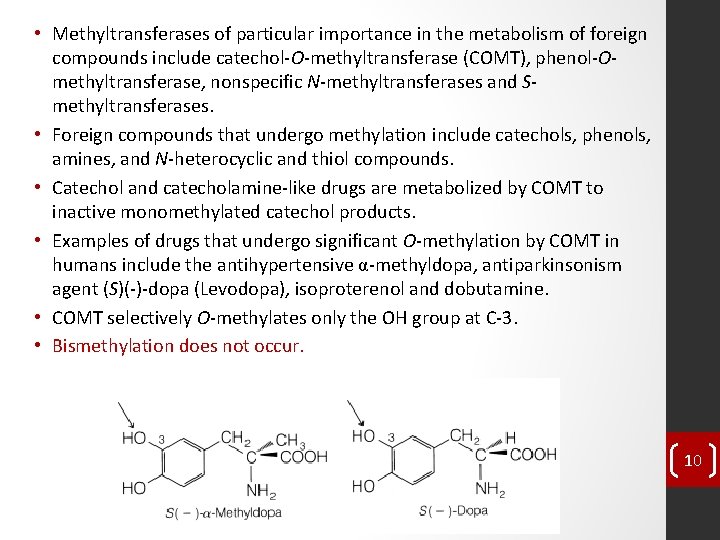
• Methyltransferases of particular importance in the metabolism of foreign compounds include catechol-O-methyltransferase (COMT), phenol-Omethyltransferase, nonspecific N-methyltransferases and Smethyltransferases. • Foreign compounds that undergo methylation include catechols, phenols, amines, and N-heterocyclic and thiol compounds. • Catechol and catecholamine-like drugs are metabolized by COMT to inactive monomethylated catechol products. • Examples of drugs that undergo significant O-methylation by COMT in humans include the antihypertensive α-methyldopa, antiparkinsonism agent (S)(-)-dopa (Levodopa), isoproterenol and dobutamine. • COMT selectively O-methylates only the OH group at C-3. • Bismethylation does not occur. 10
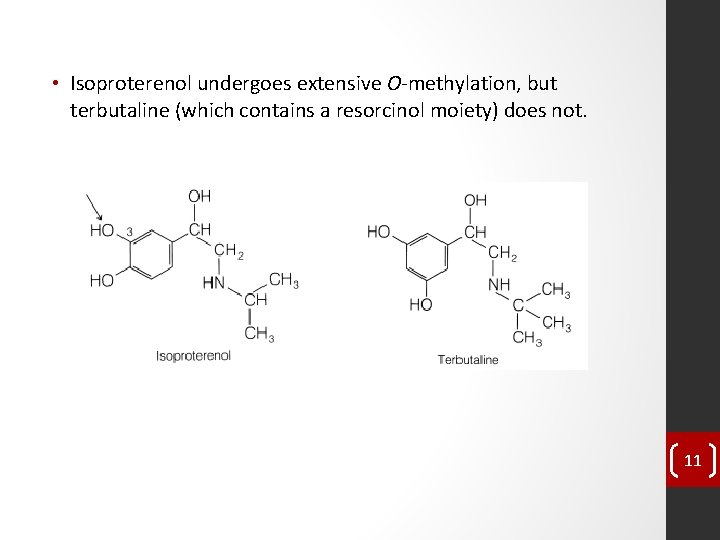
• Isoproterenol undergoes extensive O-methylation, but terbutaline (which contains a resorcinol moiety) does not. 11
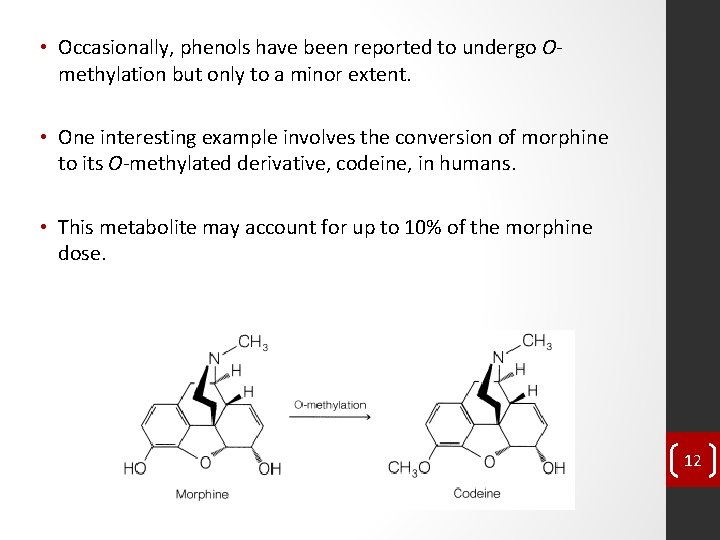
• Occasionally, phenols have been reported to undergo Omethylation but only to a minor extent. • One interesting example involves the conversion of morphine to its O-methylated derivative, codeine, in humans. • This metabolite may account for up to 10% of the morphine dose. 12

FACTORS AFFECTING DRUG METABOLISM 1 - Age Differences • 2 - Species and Strain Differences: The metabolism of many drugs and foreign compounds is often species dependent. Different animal species may biotransform a particular xenobiotic by similar or markedly different metabolic pathways. Even within the same species, individual variations (strain differences) may result in significant differences in a specific metabolic pathway. • Species variation has been observed in many oxidative biotransformation reactions. • For example, metabolism of amphetamine occurs by two main pathways: oxidative deamination or aromatic hydroxylation. • In human, rabbit, and guinea pig, oxidative deamination appears to be the predominant pathway; in the rat, aromatic hydroxylation appears to be the more important route. 3 - Hereditary or Genetic Factors 4 - Gender 13

5 - Enzyme Induction • The process by which the activity of drug-metabolizing enzymes is increased is termed enzyme induction. • Enzyme induction often increases the rate of drug metabolism and decreases the duration of drug action. (See Table 3. 4 for a list of clinically significant drug–drug interactions) • Inducing agents may increase the rate of their own metabolism as well. • Concomitant administration of two or more drugs often may lead to serious drug interactions as a result of enzyme induction. • A clinically critical drug interaction occurs with phenobarbital and warfarin. • Induction of microsomal enzymes by phenobarbital increases the metabolism of warfarin and, consequently, markedly decreases the anticoagulant effect. 14

6 - Enzyme Inhibition • Several drugs, other xenobiotics including grapefruit, and possibly other foods can inhibit drug metabolism. • With decreased metabolism, a drug often accumulates, leading to prolonged drug action and serious adverse effects. • The metabolism of phenytoin is inhibited by drugs such as chloramphenicol and isoniazid. • Several drugs, such as dicumarol, chloramphenicol and phenylbutazone, inhibit the biotransformation of tolbutamide, which may lead to a hypoglycemic response. • The grapefruit–drug interaction is complex. It may be caused by the bioflavonoids or the furanocoumarins. 15

7 - Miscellaneous Factors Affecting Drug Metabolism • Dietary factors. • Indoles present in vegetables such as Brussels sprouts, cabbage, and cauliflower, and polycyclic aromatic hydrocarbons present in charcoal broiled beef induce enzymes and stimulate the metabolism of some drugs. • Vitamins, minerals, starvation, and malnutrition also apparently influence drug metabolism. • Finally, physiological factors, such as the pathological state of the liver (e. g. , hepatic cancer, cirrhosis, hepatitis), pregnancy, hormonal disturbances may markedly affect drug metabolism. 16

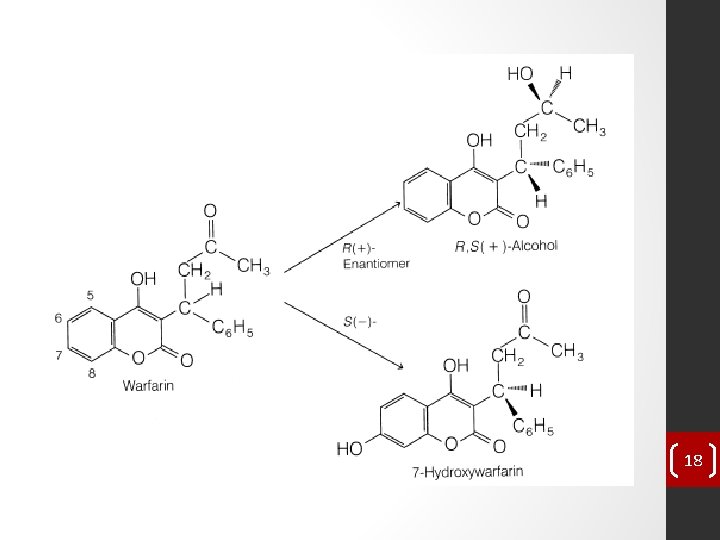
Stereochemical Aspects of Drug Metabolism • Many drugs (e. g. , warfarin, propranolol, hexobarbital, glutethimide, cyclophosphamide, ketamine, and ibuprofen) often are administered as racemic mixtures in humans. • The two enantiomers present in a racemic mixture may differ in pharmacological activity. • Usually, one enantiomer tends to be much more active than the other. • For example, the (S)(-) enantiomer of warfarin is 5 times more potent as an oral anticoagulant than the (R)(+) enantiomer. • Dramatic differences in the metabolic profile of two enantiomers of warfarin also have been noted. • In humans, the more active (S)(-)-isomer is 7 -hydroxylated (aromatic hydroxylation), whereas the (R)(+)-isomer undergoes keto reduction to yield primarily the (R, S) warfarin alcohol as the major plasma metabolite. 17
18

END 19
- Slides: 19