AP Chemistry Electronic Structure of Atoms electronic structure


















- Slides: 39

AP Chemistry Electronic Structure of Atoms electronic structure: the arrangement of electrons in an atom quantum mechanics: the physics that correctly describes atoms ?

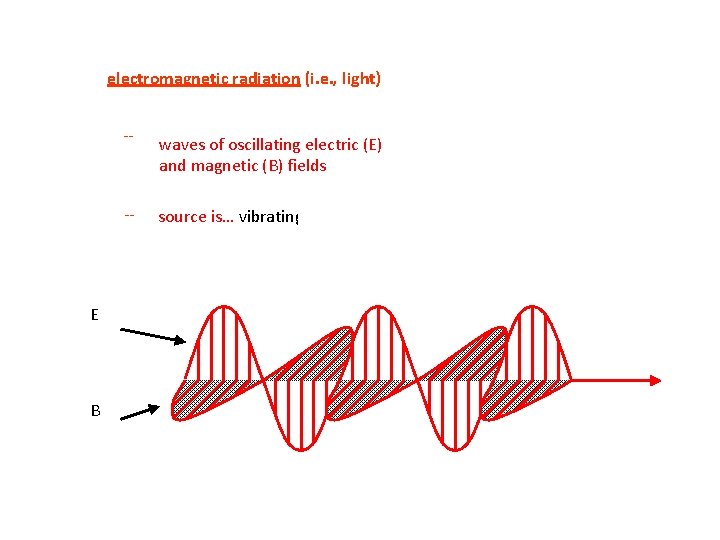
electromagnetic radiation (i. e. , light) E B -- waves of oscillating electric (E) and magnetic (B) fields -- source is… vibrating electric charges

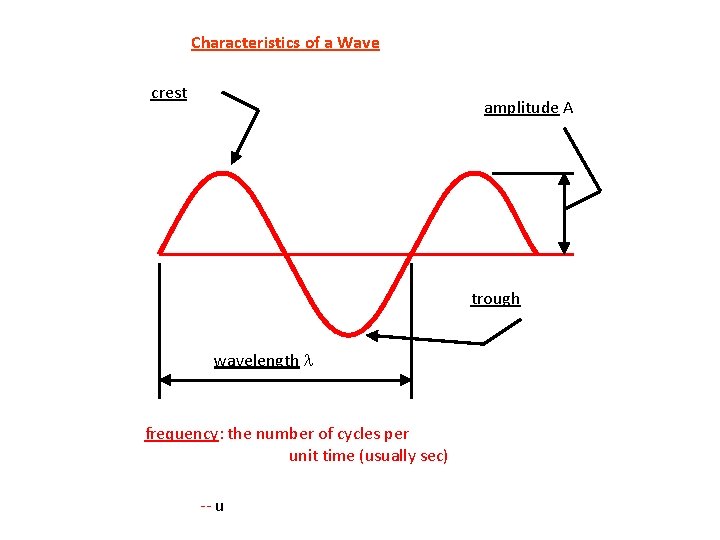
Characteristics of a Wave crest amplitude A trough wavelength l frequency: the number of cycles per unit time (usually sec) -- unit is Hz, or s– 1

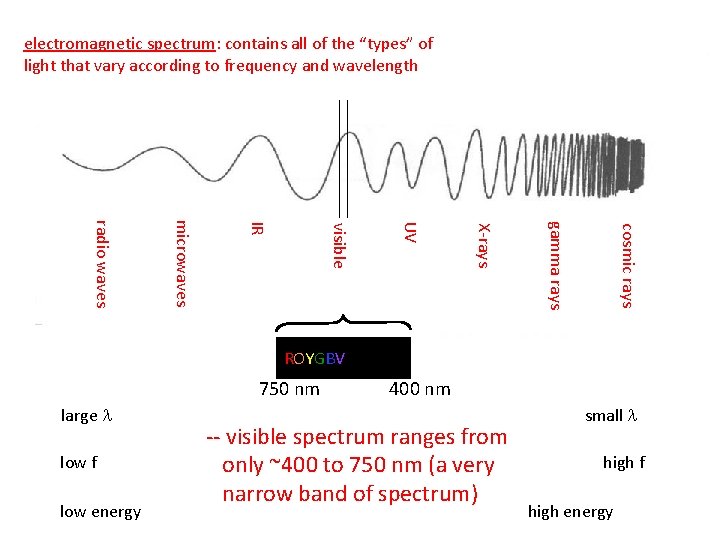
electromagnetic spectrum: contains all of the “types” of light that vary according to frequency and wavelength cosmic rays gamma rays X-rays UV visible IR microwaves radio waves ROYGBV 750 nm large l low f low energy 400 nm -- visible spectrum ranges from only ~400 to 750 nm (a very narrow band of spectrum) small l high f high energy

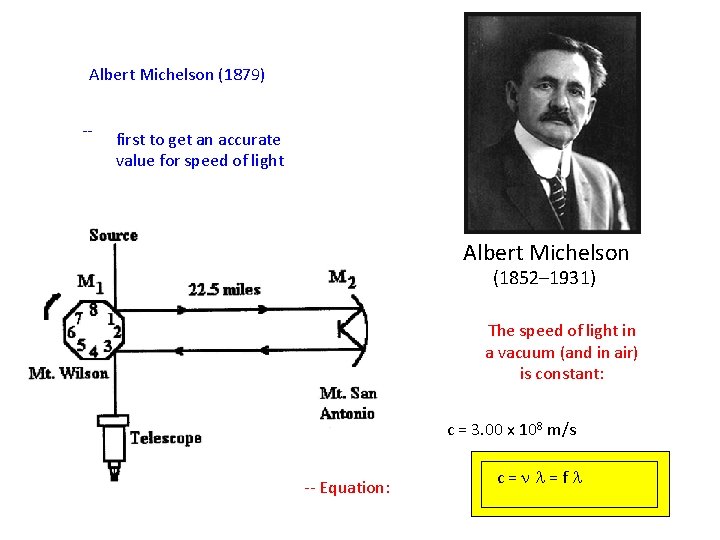
Albert Michelson (1879) -- first to get an accurate value for speed of light Albert Michelson (1852– 1931) The speed of light in a vacuum (and in air) is constant: c = 3. 00 x 108 m/s -- Equation: c=nl=fl

In 1900, Max Planck assumed that energy can be absorbed or released only in certain discrete amounts, which he called quanta. Later, Albert Einstein dubbed a light “particle” that carried a quantum of energy a photon. (1858– 1947) E=hn=hf -- Equation: E = energy, in J h = Planck’s constant = 6. 63 x Max Planck 10– 34 J-s (i. e. , J/Hz) Albert Einstein (1879– 1955)

A radio station transmits at 95. 5 MHz (FM 95. 5). Calculate the wavelength of this light and the energy of one of its photons. c=fl E=hf = 3. 00 x 108 m/s 95. 5 x 106 Hz = 6. 63 x 10– 34 J/Hz (95. 5 x 106 Hz) = 6. 33 x 10– 26 J = 3. 14 m

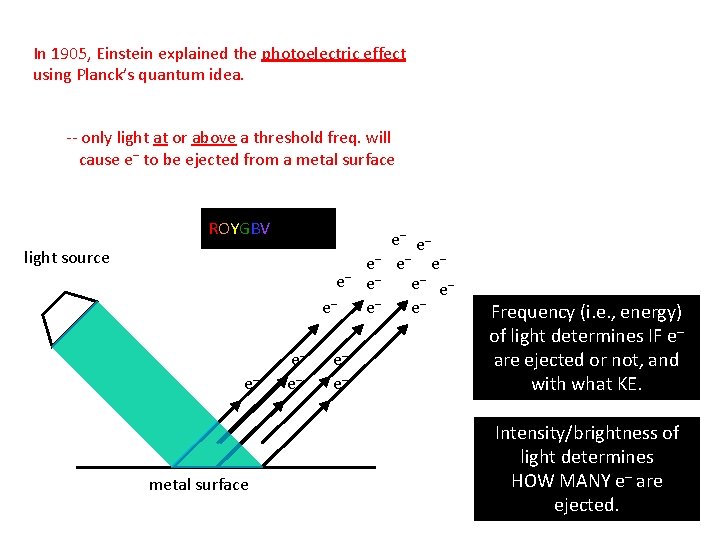
In 1905, Einstein explained the photoelectric effect using Planck’s quantum idea. -- only light at or above a threshold freq. will cause e– to be ejected from a metal surface ROYGBV light source e– e– e– metal surface e– e– e– e– Frequency (i. e. , energy) of light determines IF e– are ejected or not, and with what KE. Intensity/brightness of light determines HOW MANY e– are ejected.

Einstein also expanded Planck’s idea, saying that energy exists only in quanta. Light has both wavelike and particle-like qualities, and. . . so does matter. ?

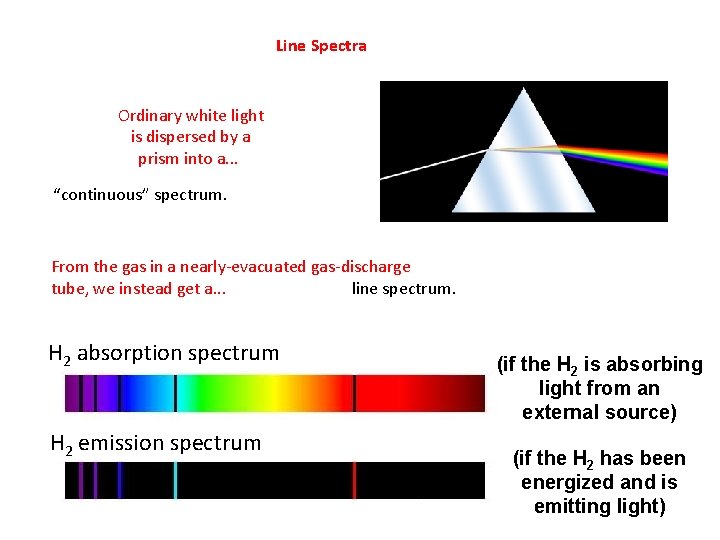
Line Spectra Ordinary white light is dispersed by a prism into a. . . “continuous” spectrum. From the gas in a nearly-evacuated gas-discharge tube, we instead get a. . . line spectrum. H 2 absorption spectrum H 2 emission spectrum (if the H 2 is absorbing light from an external source) (if the H 2 has been energized and is emitting light)

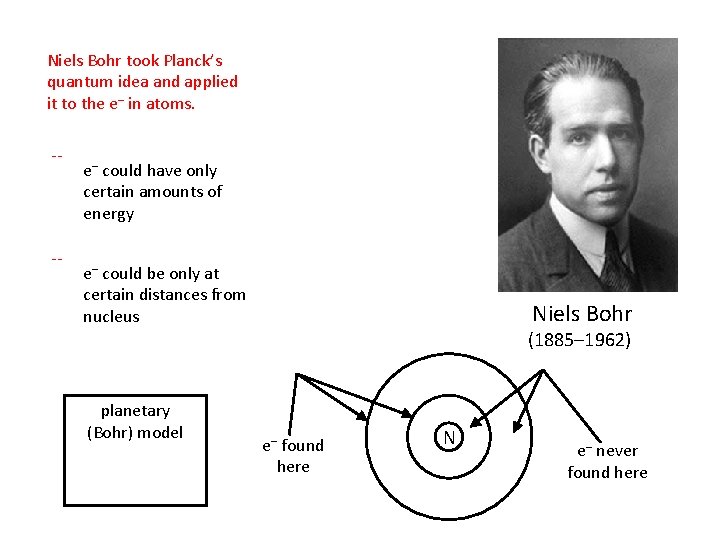
Niels Bohr took Planck’s quantum idea and applied it to the e– in atoms. -- -- e– could have only certain amounts of energy e– could be only at certain distances from nucleus planetary (Bohr) model Niels Bohr (1885– 1962) e– found here N e– never found here

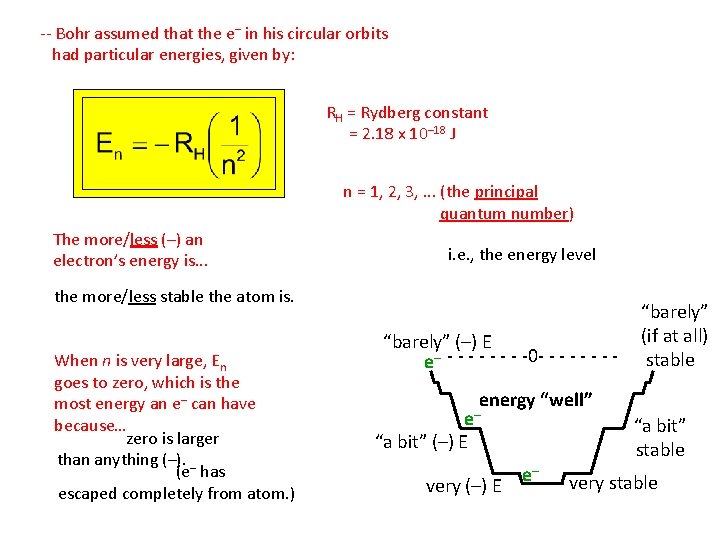
-- Bohr assumed that the e– in his circular orbits had particular energies, given by: RH = Rydberg constant = 2. 18 x 10– 18 J n = 1, 2, 3, . . . (the principal quantum number) The more/less (–) an electron’s energy is. . . i. e. , the energy level the more/less stable the atom is. When n is very large, En goes to zero, which is the most energy an e– can have because… zero is larger than anything (–). – (e has escaped completely from atom. ) “barely” (–) E e– - - - -0 - - - - “barely” (if at all) stable energy “well” e– “a bit” (–) E very (–) E e– “a bit” stable very stable

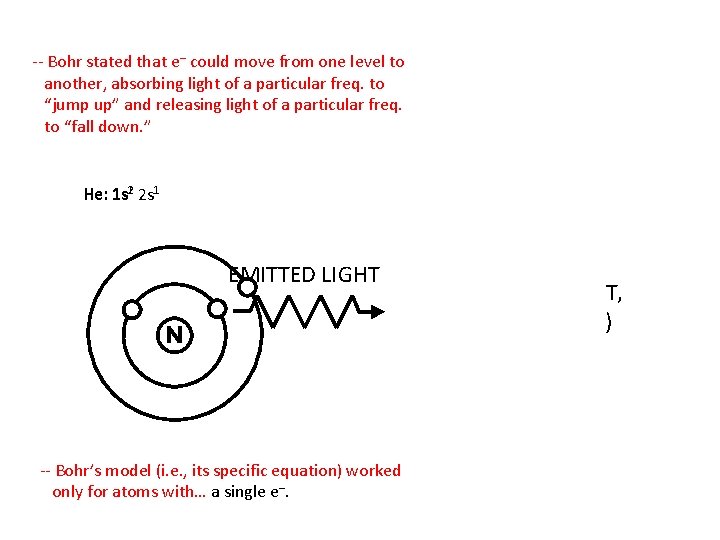
-- Bohr stated that e– could move from one level to another, absorbing light of a particular freq. to “jump up” and releasing light of a particular freq. to “fall down. ” He: 1 s 12 2 s 1 ENERGY EMITTED LIGHT -- Bohr’s model (i. e. , its specific equation) worked only for atoms with… a single e–. (HEAT, LIGHT, ELEC. , ETC. )

Find the wavelength, frequency, and energy of a photon of the emission line produced when an e– in a hydrogen atom makes the transition from n = 5 to n = 2. For n = 5… = – 8. 72 x 10– 20 J For n = 2… DE = 4. 58 x 10– 19 J E=hf c=fl E 2 = – 5. 45 x 10– 19 J = energy of photon = 6. 91 x 1014 Hz = 4. 34 x 10– 7 m

The Wave Behavior of Matter Since light (traditionally thought of as a wave) was found to behave as both a wave and a particle, Louis de Broglie suggested that matter (traditionally thought of as particles) might also behave like both a wave and a particle. He called these… matter waves. Louis de Broglie (1892– 1987) -- The wavelength of a moving sample of matter is given by: m = mass v = speed (kg) (m/s)

A proton moving at 1200 m/s would be associated with a matter wave of how many nanometers? In the absence of further info… mp ~ 1 amu = 1. 66 x 10– 27 kg = 3. 3 x 10– 10 m = 0. 33 nm (actual p+ mass is ~1. 673 x 10– 27 kg)

A wave is smeared out through space, i. e. , its location is not precisely defined. Since matter exhibits wave characteristics, there are limits to how precisely we can define a particle’s (e. g. , an e–’s) location. -- the limitation also applies to a particle’s. . . momentum -- Heisenberg’s uncertainty principle: It is impossible to know simultaneously BOTH the exact momentum of a particle AND its exact location in space. D = “uncertainty in” h = Planck’s const. (It is inappropriate to imagine an e– as a solid particle moving in a well-defined orbit. ) Werner Heisenber (1901– 1976)

Schrodinger’s wave equation (1926) accounts for both wave and particle behaviors of e–. w/no where h-bar is theof reduced Planck’s constant (=h/2 p), V is the potential energy, and Y iselectric the wave function) (for a non-relativistic particle mass m charge and zero spin, -- Solutions to the wave equation yield wave functions, symbolized by Y, which have no physical meaning, but Y 2 at any point in space gives the probability that you’ll find an e– at that point. Y 2 is called the probability density, which gives the electron density. Erwin Schrodinger (1887– 1961)

-- Orbitals describe a specific distribution of electron density in space. Each orbital has a characteristic shape and energy.

(“Not. ”) (Well, theory says that there ARE g orbitals, but they don’t look like this. ) We also g orbitals and they lookflike this… Thethink lanthanides andexist, actinides contain orbitals.

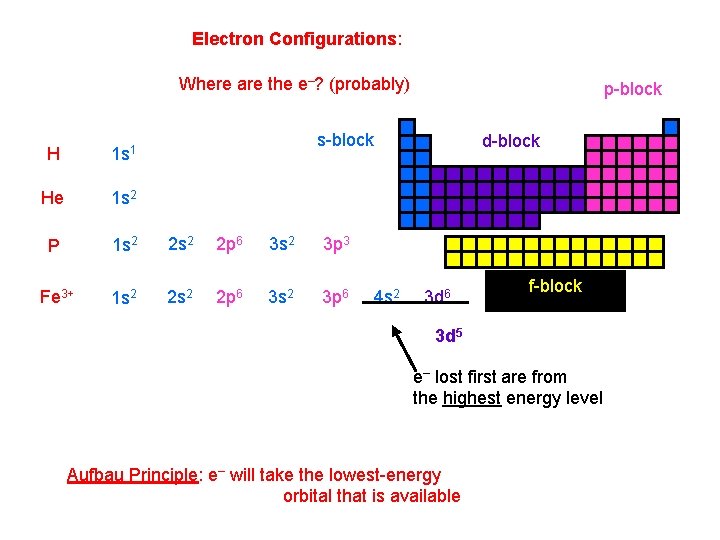
Electron Configurations: Where are the e–? (probably) p-block s-block H 1 s 1 He 1 s 2 P 1 s 2 2 p 6 3 s 2 3 p 3 Fe 3+ 1 s 2 2 p 6 3 s 2 3 p 6 d-block 4 s 2 3 d 6 f-block 3 d 5 e– lost first are from the highest energy level Aufbau Principle: e– will take the lowest-energy orbital that is available

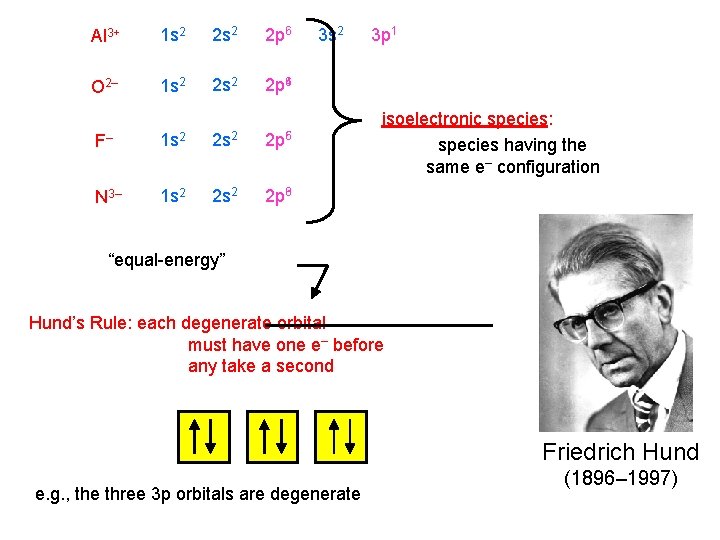
Al 3+ 1 s 2 2 p 6 O 2– 1 s 2 2 p 64 F– 1 s 2 2 p 56 N 3– 1 s 2 2 p 36 3 s 2 3 p 1 isoelectronic species: species having the same e– configuration “equal-energy” Hund’s Rule: each degenerate orbital must have one e– before any take a second Friedrich Hund e. g. , the three 3 p orbitals are degenerate (1896– 1997)

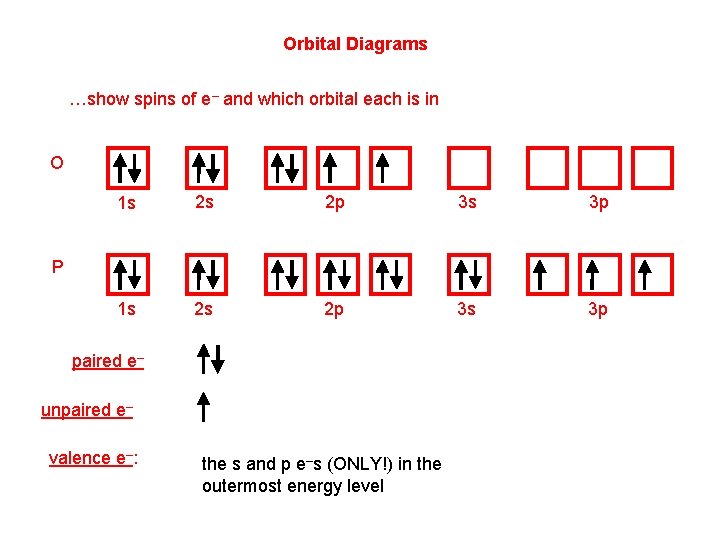
Orbital Diagrams …show spins of e– and which orbital each is in O 1 s 2 s 2 p 3 s 3 p P paired e– unpaired e– valence e–: the s and p e–s (ONLY!) in the outermost energy level

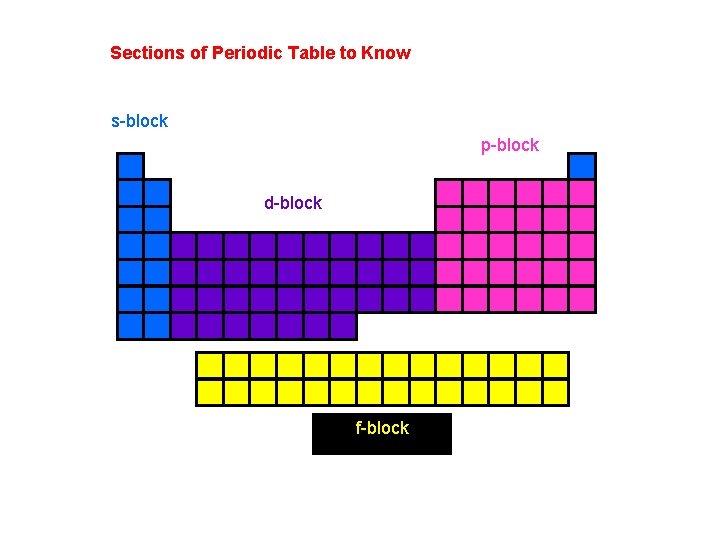
Sections of Periodic Table to Know s-block p-block d-block f-block

Shorthand Electron Configuration (S. E. C. ) 1. Put symbol of noble gas that precedes element in brackets. 2. Continue writing e– config. from that point. S [Ne] 3 s 2 3 p 4 Co [Ar] 4 s 2 3 d 7

In a many-electron atom, each e– is attracted to the nucleus and repelled by the other e–. -- effective nuclear charge, Zeff: the net (+) charge attracting an e– (a measure of how tightly particular e–s are held) Equation: Zeff = Z – S Z = atomic number 3 d 6 S = # of e– BETWEEN nucleus and e– in question (NOT e– in same subshell) -- Within a given electron shell, s e–s have the greatest Zeff, f e–s the least. 4 s 2 3 p 6 Fe 3 s 2 2 p 6 2 s 2 For Fe, the 3 p e–s have Zeff = 26 – 12 = 14; the 3 d e–s have Zeff = 26 – 18 = 8. 1 s 2 nuc.

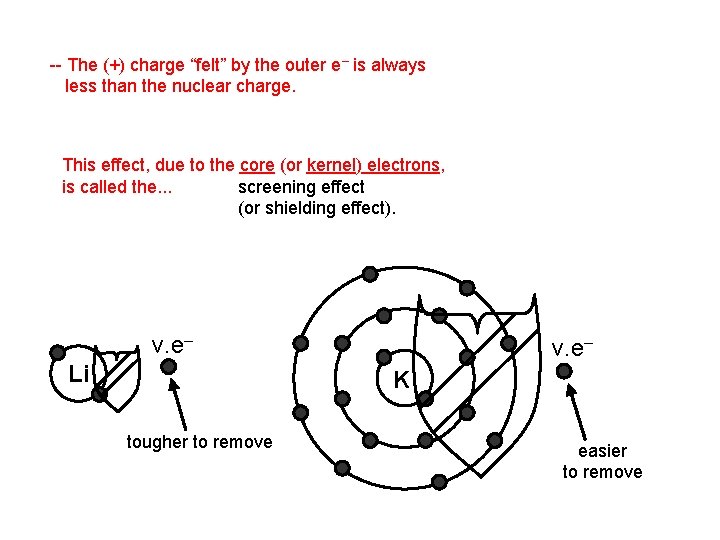
-- The (+) charge “felt” by the outer e– is always less than the nuclear charge. This effect, due to the core (or kernel) electrons, is called the. . . screening effect (or shielding effect). v. e– Li v. e– K tougher to remove easier to remove

Quantum numbers are used to describe where an e– is in an atom. 1. principal quantum number, n (n = integers 1, 2, 3, . . . ) -- correspond to the energy level of the electrons -- All orbitals having the same n are called an electron shell (e. g. , 2 s and 2 p). 2. angular momentum quantum number, l (l = integers from 0 up to (n – 1)) -- This number defines the type of subshell: s = 0, p = 1, d = 2, f = 3 -- For a given shell, the energies of orbitals go: s<p<d<f

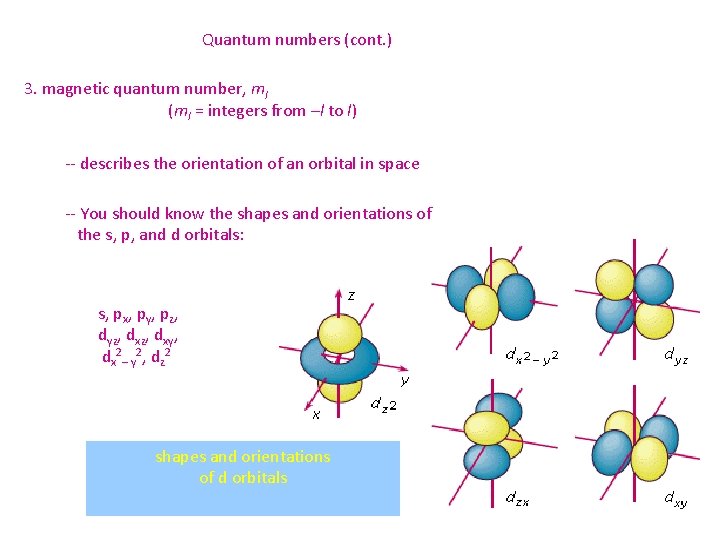
Quantum numbers (cont. ) 3. magnetic quantum number, ml (ml = integers from –l to l) -- describes the orientation of an orbital in space -- You should know the shapes and orientations of the s, p, and d orbitals: s, px, py, pz, dyz, dxy, dx 2– y 2, dz 2 shapes and orientations of d orbitals

4. electron spin quantum number, ms -- only two values: +½ or –½ (“spin-up” and “spin-down”) -- Pauli exclusion principle: No two electrons in an atom may have the same set of four quantum numbers (i. e. , an orbital may hold only two electrons, and they must have opposite spins). Wolfgang Pauli (1900– 1958)

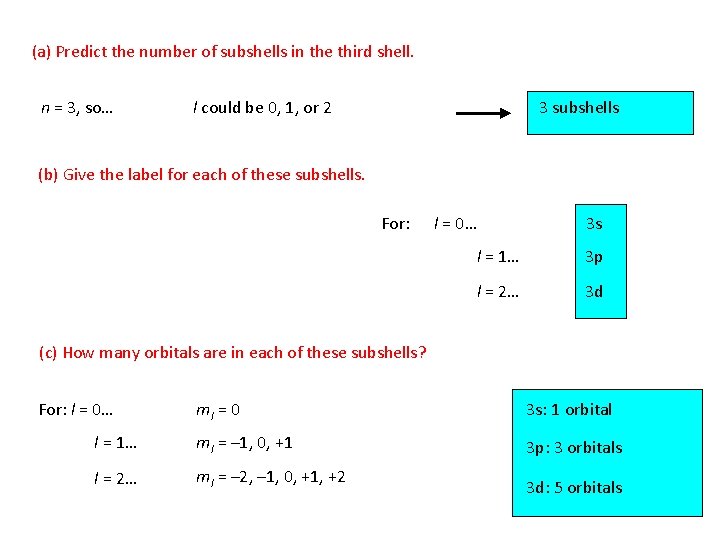
(a) Predict the number of subshells in the third shell. n = 3, so… l could be 0, 1, or 2 3 subshells (b) Give the label for each of these subshells. For: l = 0… 3 s l = 1… 3 p l = 2… 3 d (c) How many orbitals are in each of these subshells? For: l = 0… ml = 0 3 s: 1 orbital l = 1… ml = – 1, 0, +1 3 p: 3 orbitals l = 2… ml = – 2, – 1, 0, +1, +2 3 d: 5 orbitals

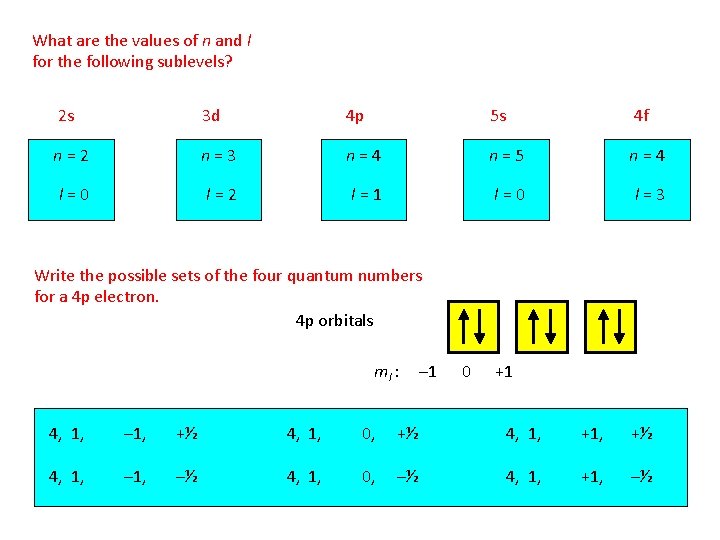
What are the values of n and l for the following sublevels? 2 s 3 d 4 p 5 s 4 f n=2 n=3 n=4 n=5 n=4 l=0 l=2 l=1 l=0 l=3 Write the possible sets of the four quantum numbers for a 4 p electron. 4 p orbitals ml : – 1 0 +1 4, 1, – 1, +½ 4, 1, 0, +½ 4, 1, –½ 4, 1, 0, –½ 4, 1, +1, –½

Write the four quantum numbers of each of the six 3 d electrons of an iron atom. 3 d orbitals ml : – 2 3, 2, – 2, +½ 3, 2, – 1, +½ 3, 2, 0, +½ 3, 2, +1, +½ 3, 2, ? , –½ – 1 0 +1 +2 ?

Brief Review of the Periodic Table metals: left side of Table; form cations properties: lustrous (shiny) good conductors (heat and electricity) ductile (can pull into wire) malleable (can hammer into shape)

Brief Review of the Periodic Table (cont. ) nonmetals: right side of Table; form anions properties: good insulators gases or brittle solids neon sulfur iodine bromine Ne S 8 I 2 Br 2

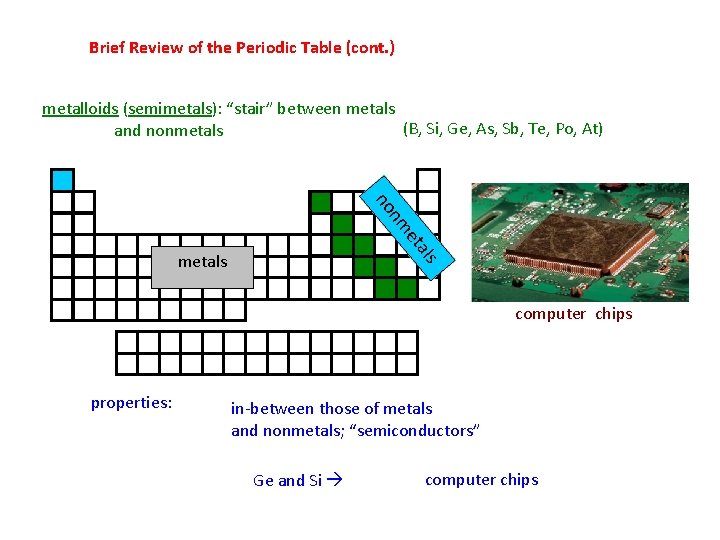
Brief Review of the Periodic Table (cont. ) metalloids (semimetals): “stair” between metals (B, Si, Ge, As, Sb, Te, Po, At) and nonmetals et nm no Si and Ge metals computer chips properties: in-between those of metals and nonmetals; “semiconductors” Ge and Si computer chips

alkali metals: group 1 (except H); 1+ charge; very reactive alkaline earth metals: groups 3– 12; variable charges transition elements: chalcogens: halogens: noble gases: lanthanides: actinides: group 2; 2+ charge; less reactive than alkalis group 16; 2– charge; reactive group 17; 1– charge; very reactive group 18; no charge; unreactive elements 58– 71 contain f orbitals elements 90– 103 main block (representative) elements: groups 1, 2, 13– 18

What family of elements has an ns 2 valence electron configuration? alkaline earth metals

Anomalies in the Electron Configurations Your best guide to writing e– configs is “The Table, ” but there a few exceptions. e. g. , Cr: [ Ar ] 4 s 1 3 d 5 Cu: [ Ar ] 4 s 1 3 d 10 These exceptions are due to the closeness in energy of the upper-level orbitals. Other exceptions are… Mo, Ru, Rh, and Ag. All of these exceptions have a single valence-level s electron. “Ru. Rh…!!”