Alkynes C C Synthesis of Acetylene Heating coke



















- Slides: 61

Alkynes C C

Synthesis of Acetylene Heating coke with lime in an electric furnace to forms calcium carbide. Then drip water on the calcium carbide. coke lime * *This reaction was used to produce light for miners’ lamps and for the stage.

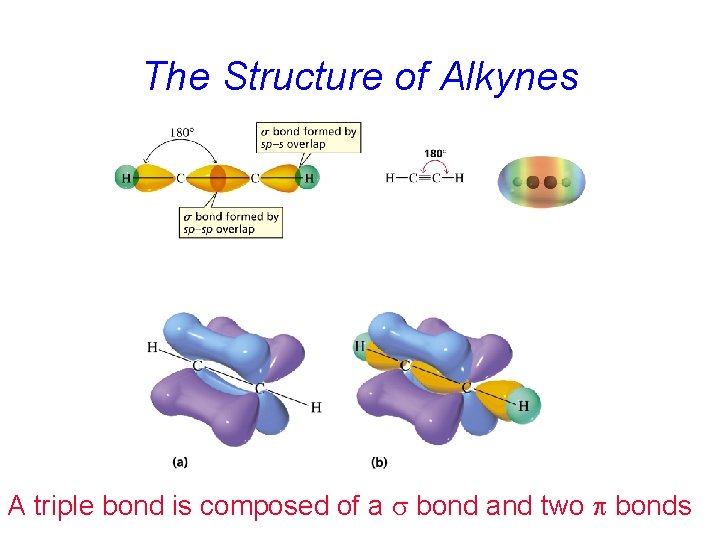
The Structure of Alkynes A triple bond is composed of a s bond and two p bonds

Acidity of Acetylene and Terminal Alkynes H C C

Acidity of Hydrocarbons In general, hydrocarbons are excedingly weak acids Compound p. Ka HF 3. 2 H 2 O 16 NH 3 36 H 2 C CH 2 45 CH 4 60

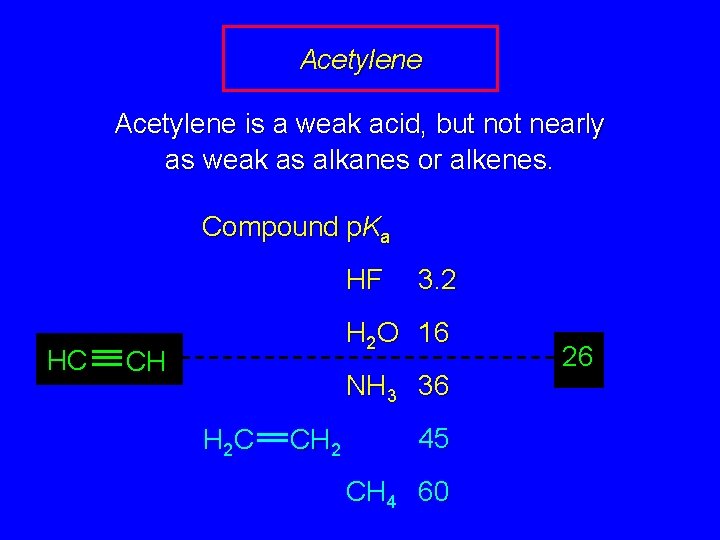
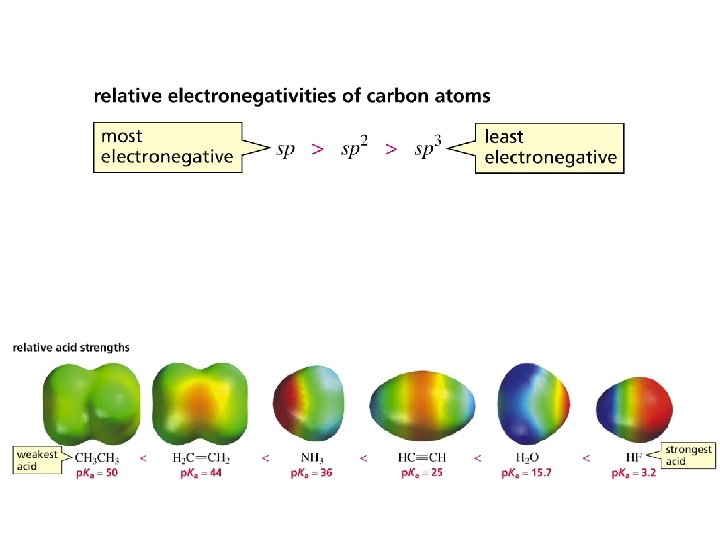
Acetylene is a weak acid, but not nearly as weak as alkanes or alkenes. Compound p. Ka HF HC 3. 2 H 2 O 16 CH NH 3 36 H 2 C CH 2 45 CH 4 60 26


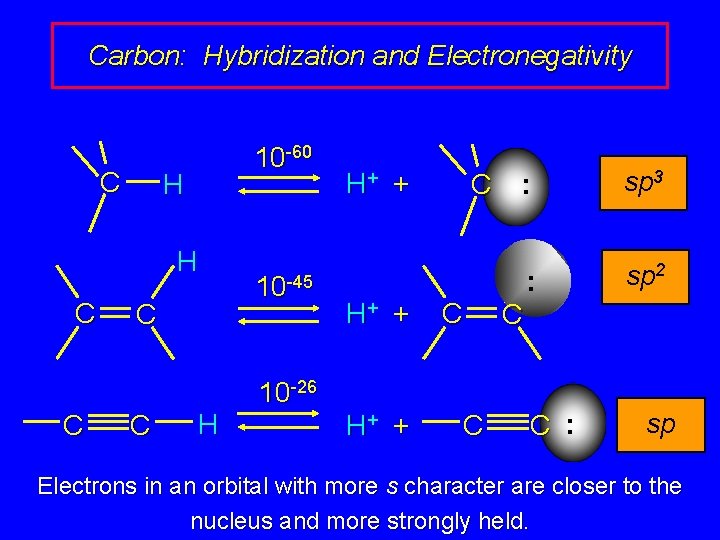
Carbon: Hybridization and Electronegativity C 10 -60 H H C C H 10 -45 H+ + C C 10 -26 H+ + C : sp 3 : sp 2 C C : sp Electrons in an orbital with more s character are closer to the nucleus and more strongly held.

The stronger the acid, the weaker its conjugate base top 252

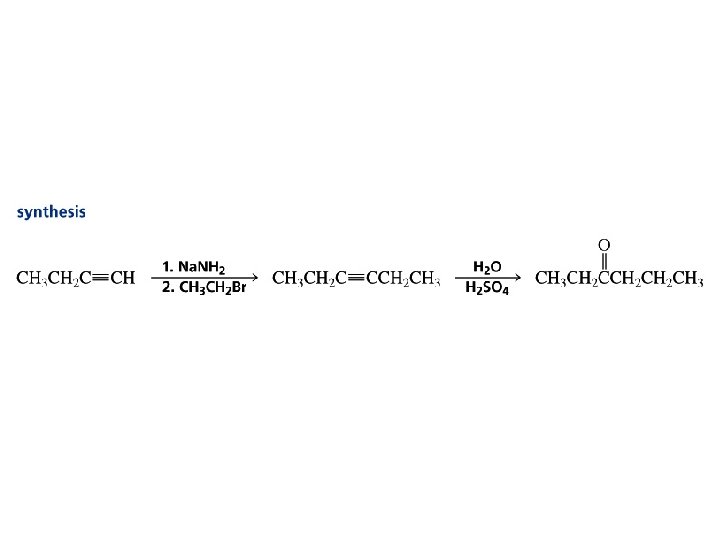
Sodium Acetylide Solution: Use a stronger base. Sodium amide is a stronger base than sodium hydroxide. Na. NH 2 + HC CH Na. C CH + NH 3. . – H 2 N : + H C CH stronger acid p. Ka = 26 . . H 2 N – H + : C weaker acid p. Ka = 36 Ammonia is a weaker acid than acetylene. The position of equilibrium lies to the right. CH

Preparation of Various Alkynes by alkylation reactions with Acetylide or Terminal Alkynes

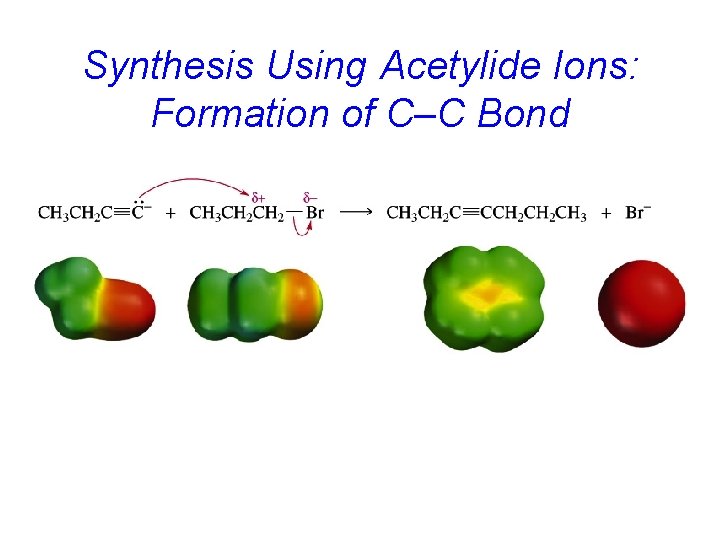
Synthesis Using Acetylide Ions: Formation of C–C Bond

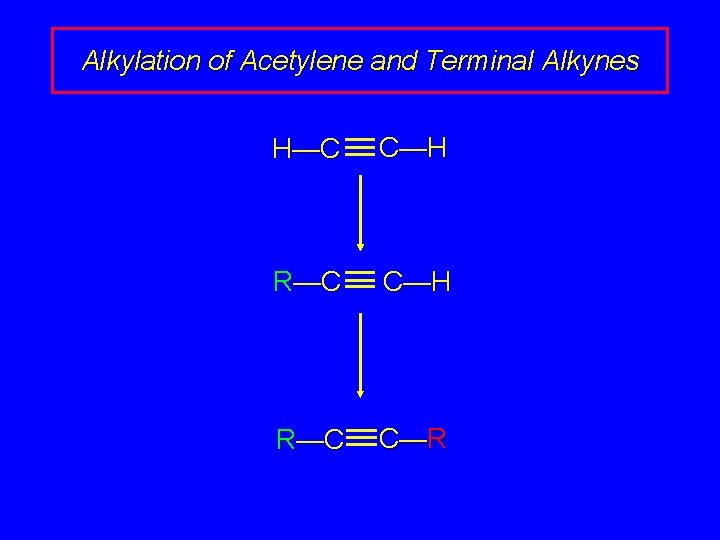
Alkylation of Acetylene and Terminal Alkynes H—C C—H R—C C—R

Alkylation of Acetylene and Terminal Alkynes H—C – C: + R X S N 2 H—C C—R + : X– The alkylating agent is an alkyl halide, and the reaction is nucleophilic substitution. The nucleophile is sodium acetylide or the sodium salt of a terminal (monosubstituted) alkyne.

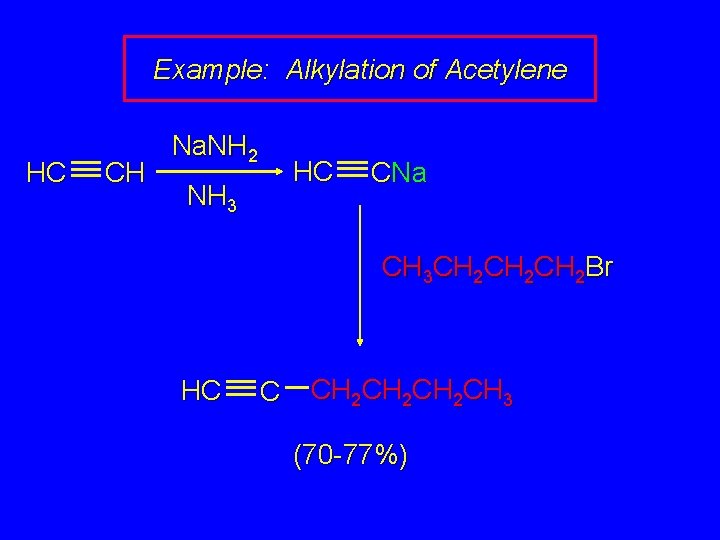
Example: Alkylation of Acetylene HC CH Na. NH 2 HC NH 3 CNa CH 3 CH 2 CH 2 Br HC C CH 2 CH 2 CH 3 (70 -77%)

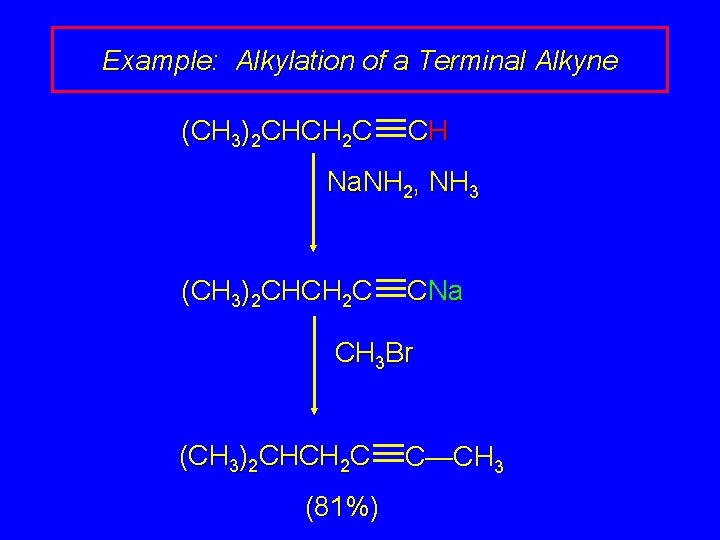
Example: Alkylation of a Terminal Alkyne (CH 3)2 CHCH 2 C CH Na. NH 2, NH 3 (CH 3)2 CHCH 2 C CNa CH 3 Br (CH 3)2 CHCH 2 C (81%) C—CH 3

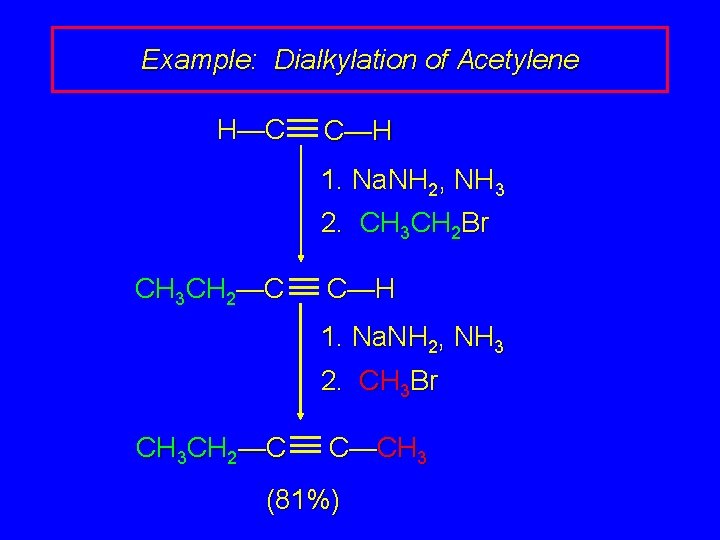
Example: Dialkylation of Acetylene H—C C—H 1. Na. NH 2, NH 3 2. CH 3 CH 2 Br CH 3 CH 2—C C—H 1. Na. NH 2, NH 3 2. CH 3 Br CH 3 CH 2—C C—CH 3 (81%)

Limitation Effective only with primary alkyl halides Secondary and tertiary alkyl halides undergo elimination

Reactions of Alkynes

Reactions of Alkynes Acidity Hydrogenation Metal-Ammonia Reduction Addition of Hydrogen Halides Hydration

Hydrogenation of Alkynes

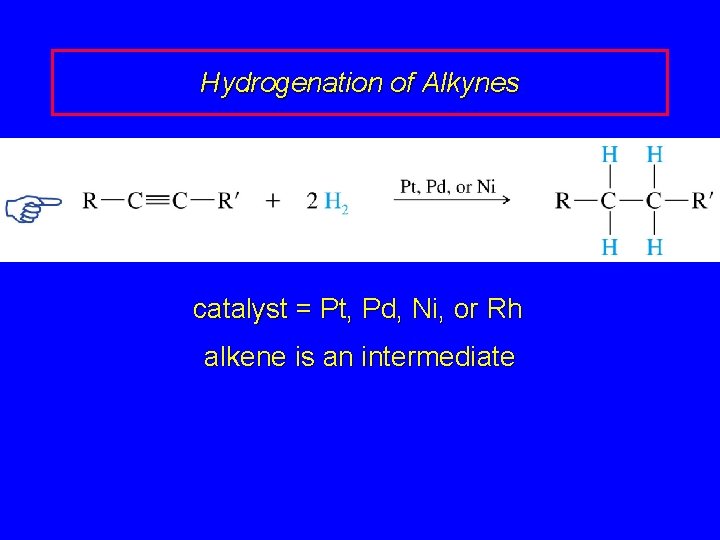
Hydrogenation of Alkynes RC CR' + 2 H 2 cat RCH 2 R' catalyst = Pt, Pd, Ni, or Rh alkene is an intermediate

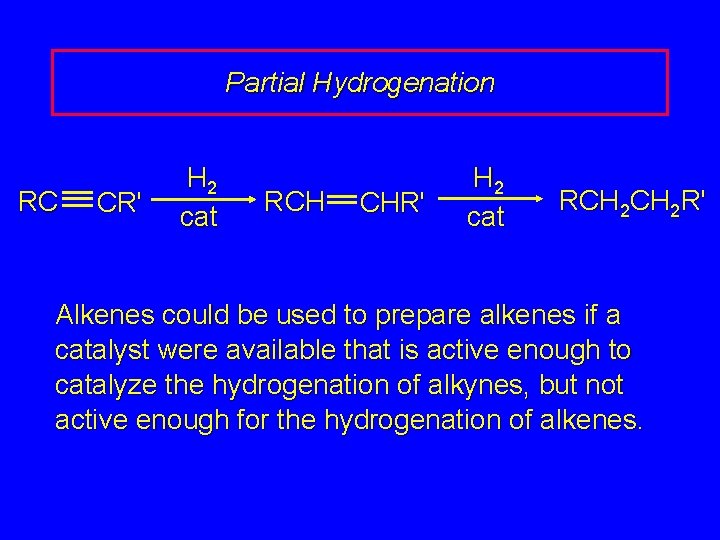
Partial Hydrogenation RC CR' H 2 cat RCH CHR' H 2 cat RCH 2 R' Alkenes could be used to prepare alkenes if a catalyst were available that is active enough to catalyze the hydrogenation of alkynes, but not active enough for the hydrogenation of alkenes.

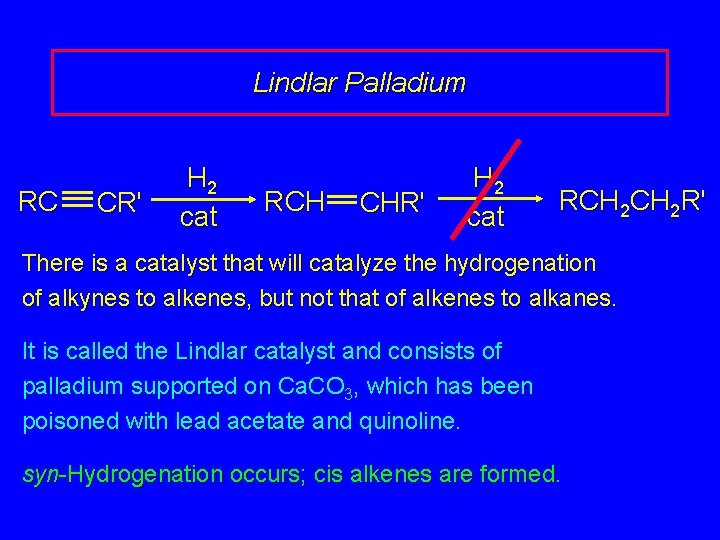
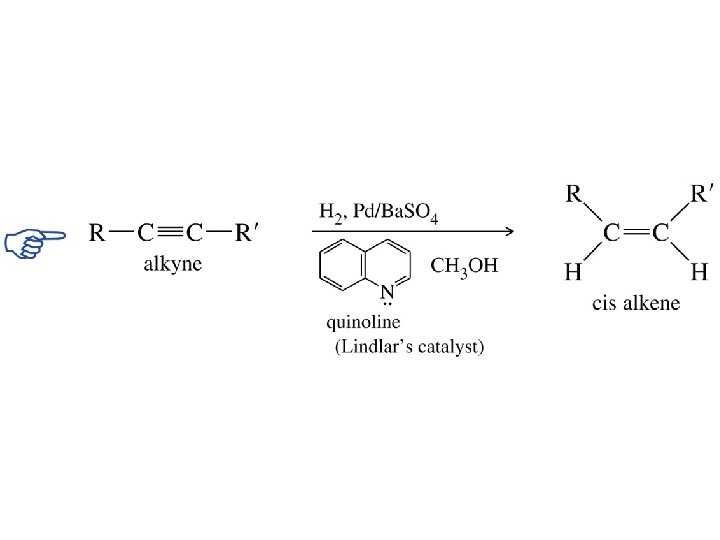
Lindlar Palladium RC CR' H 2 cat RCH CHR' H 2 cat RCH 2 R' There is a catalyst that will catalyze the hydrogenation of alkynes to alkenes, but not that of alkenes to alkanes. It is called the Lindlar catalyst and consists of palladium supported on Ca. CO 3, which has been poisoned with lead acetate and quinoline. syn-Hydrogenation occurs; cis alkenes are formed.

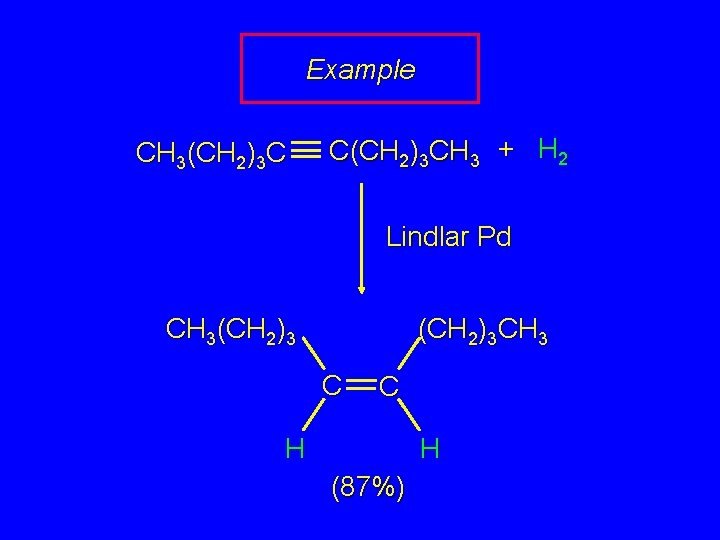
Example CH 3(CH 2)3 C C(CH 2)3 CH 3 + H 2 Lindlar Pd CH 3(CH 2)3 CH 3 C C H H (87%)



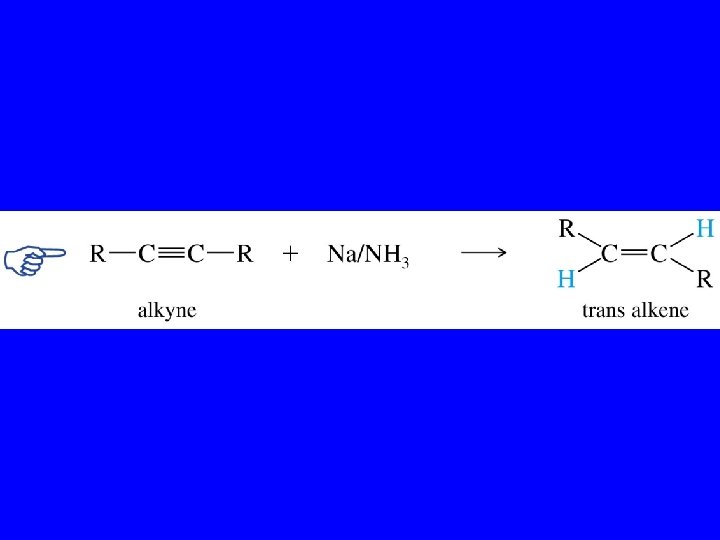
Metal-Ammonia Reduction of Alkynes trans-Alkenes

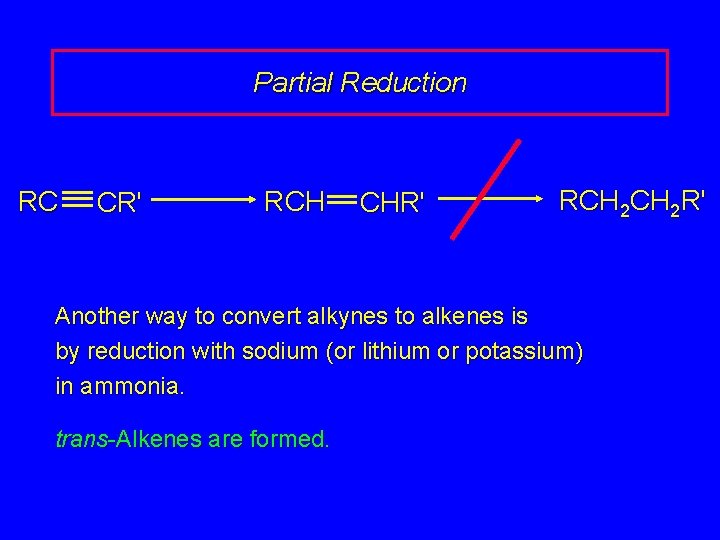
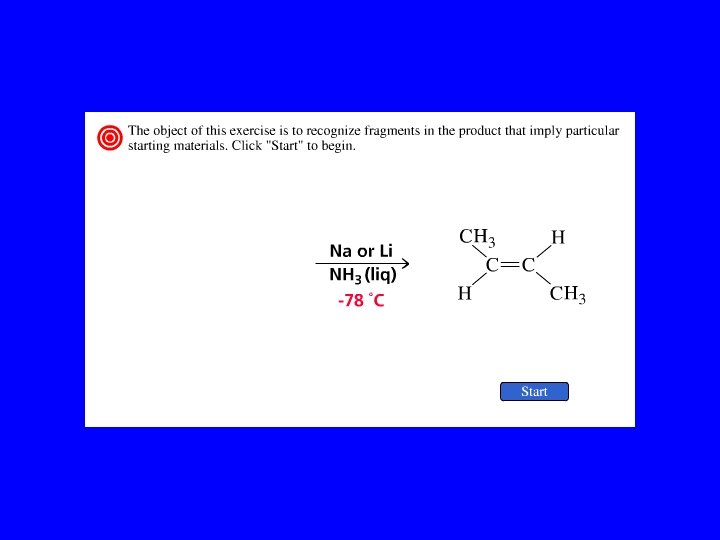
Partial Reduction RC CR' RCH CHR' RCH 2 R' Another way to convert alkynes to alkenes is by reduction with sodium (or lithium or potassium) in ammonia. trans-Alkenes are formed.


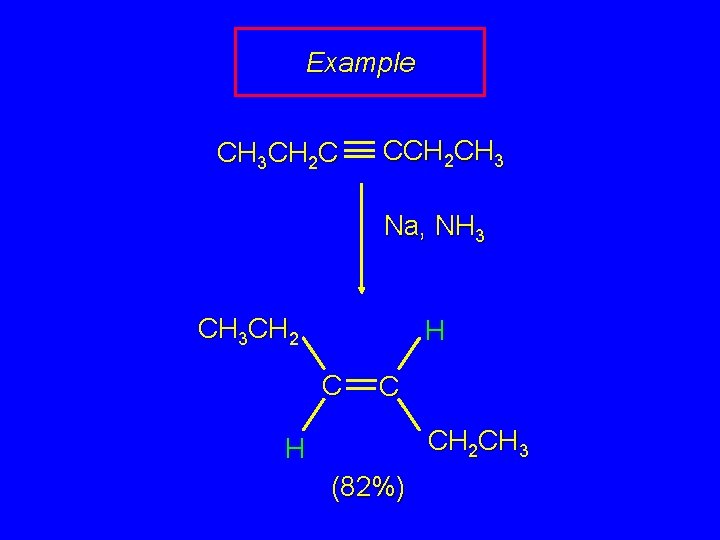
Example CH 3 CH 2 C CCH 2 CH 3 Na, NH 3 CH 3 CH 2 H C C CH 2 CH 3 H (82%)


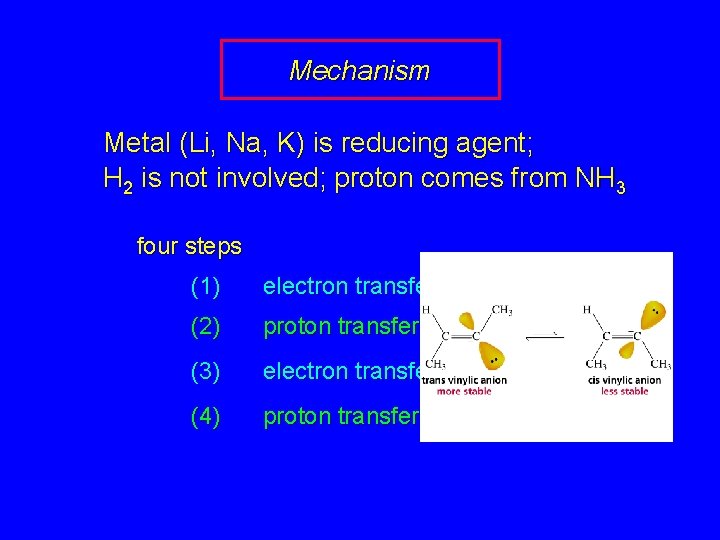
Mechanism Metal (Li, Na, K) is reducing agent; H 2 is not involved; proton comes from NH 3 four steps (1) electron transfer (2) proton transfer (3) electron transfer (4) proton transfer

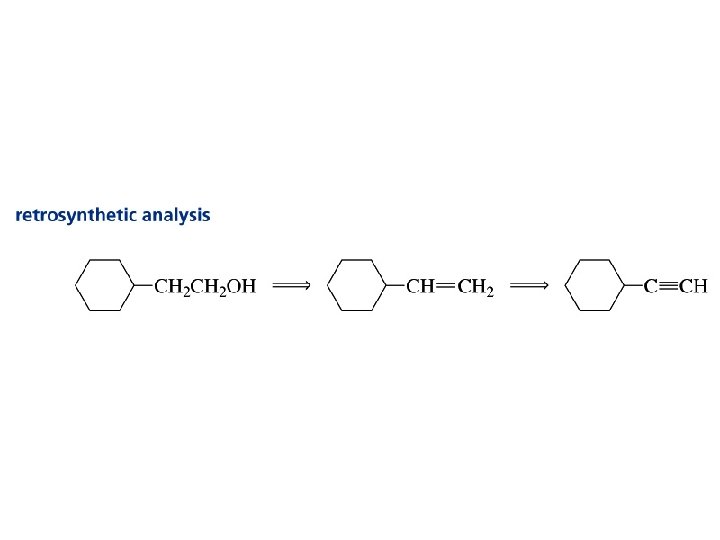
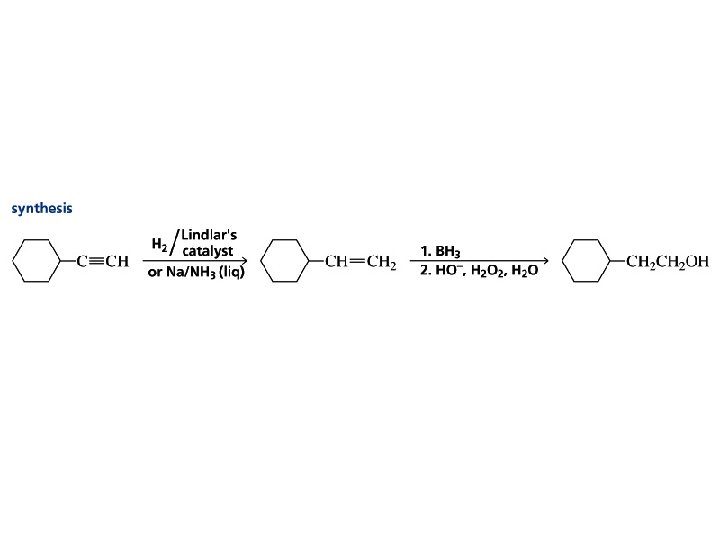
Problem Suggest an efficient syntheses of (E)- and (Z)-2 heptene from propyne and any necessary organic or inorganic reagents.

Problem Strategy

Problem Strategy

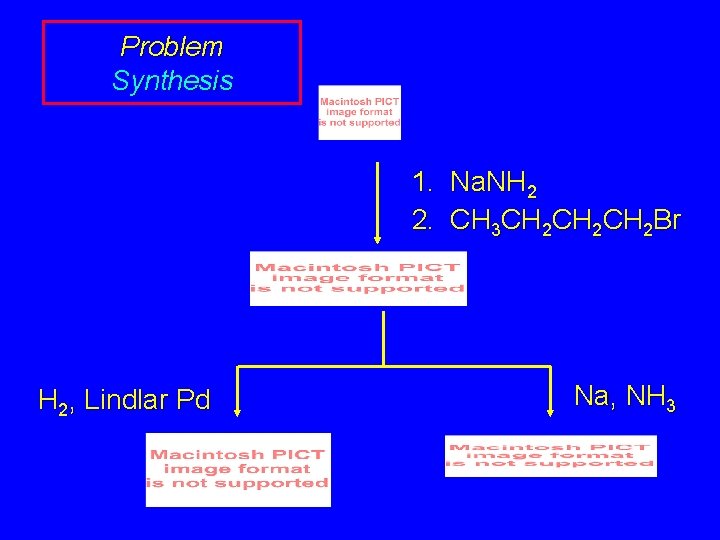
Problem Synthesis 1. Na. NH 2 2. CH 3 CH 2 CH 2 Br H 2, Lindlar Pd Na, NH 3

Addition of Hydrogen Halides to Alkynes

Follows Markovnikov's Rule CH 3(CH 2)3 C CH HBr CH 3(CH 2)3 C CH 2 Br (60%) Alkynes are slightly less reactive than alkenes

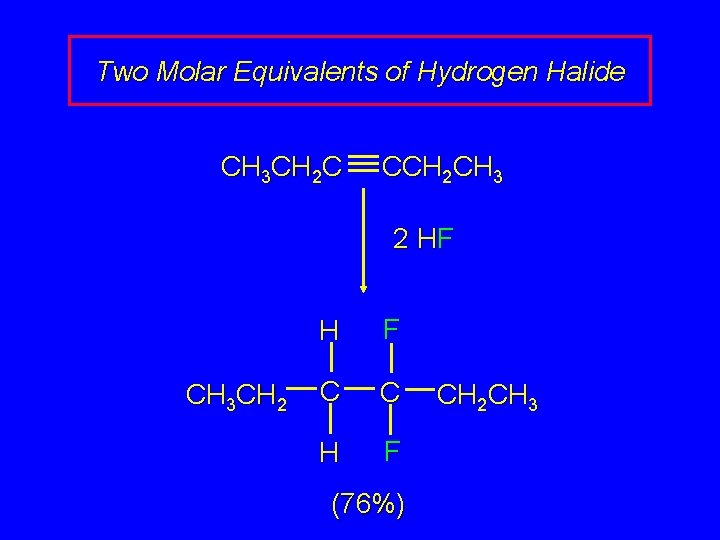
Two Molar Equivalents of Hydrogen Halide CH 3 CH 2 C CCH 2 CH 3 2 HF CH 3 CH 2 H F C C H F (76%) CH 2 CH 3

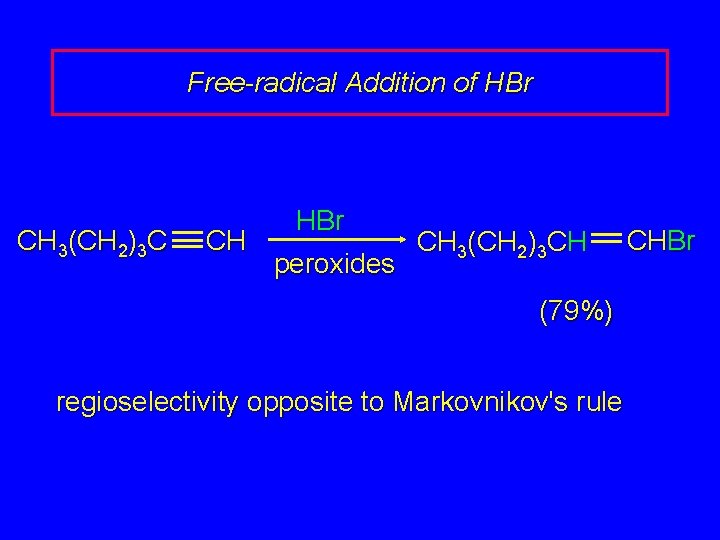
Free-radical Addition of HBr CH 3(CH 2)3 C CH HBr peroxides CH 3(CH 2)3 CH (79%) regioselectivity opposite to Markovnikov's rule CHBr

Hydration of Alkynes


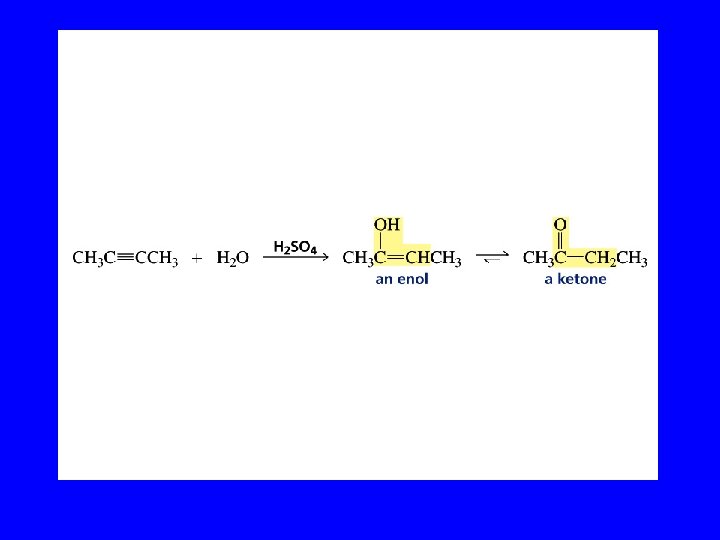
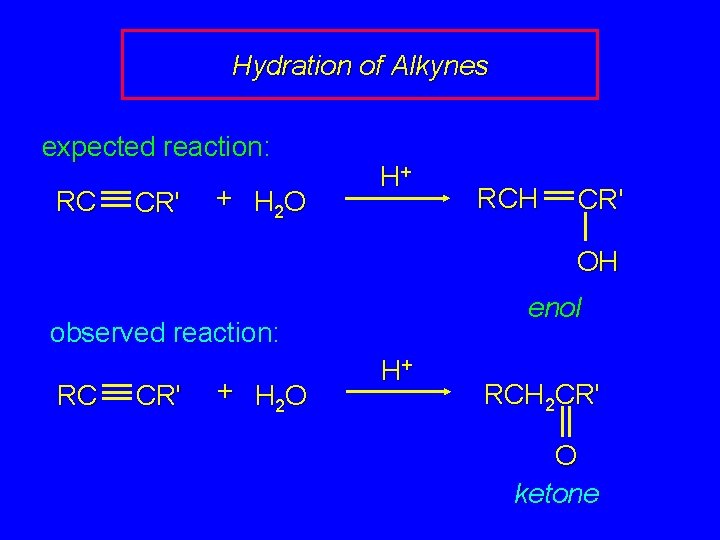
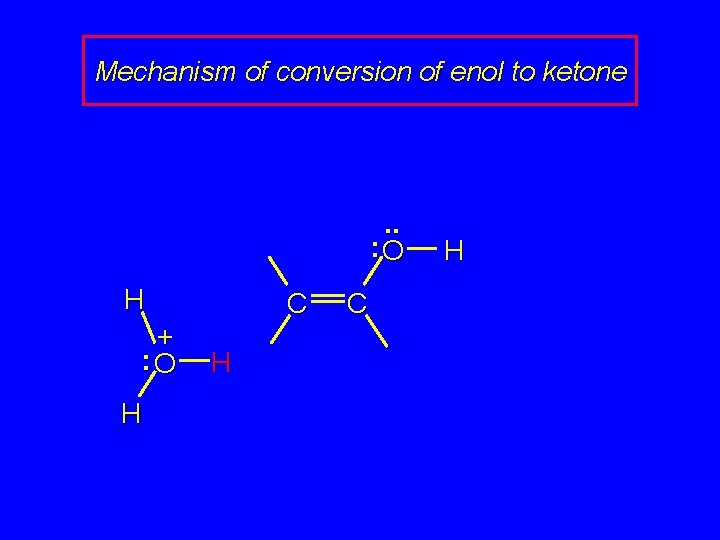
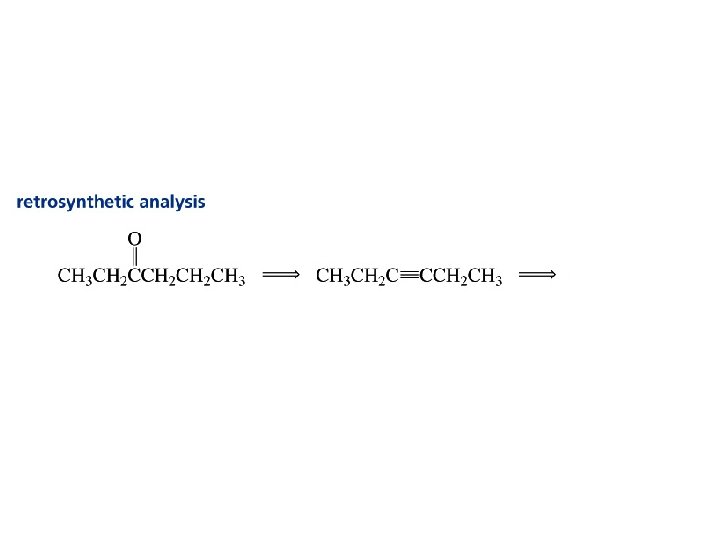
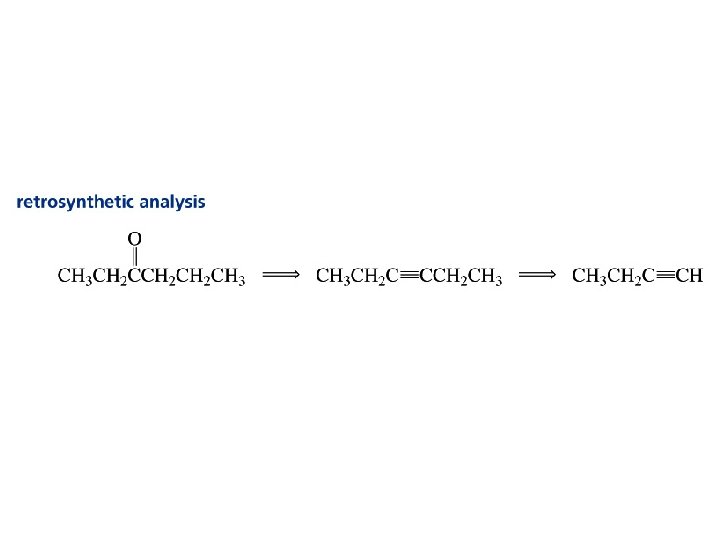
Hydration of Alkynes expected reaction: RC CR' + H 2 O H+ RCH CR' OH enol observed reaction: RC CR' + H 2 O H+ RCH 2 CR' O ketone

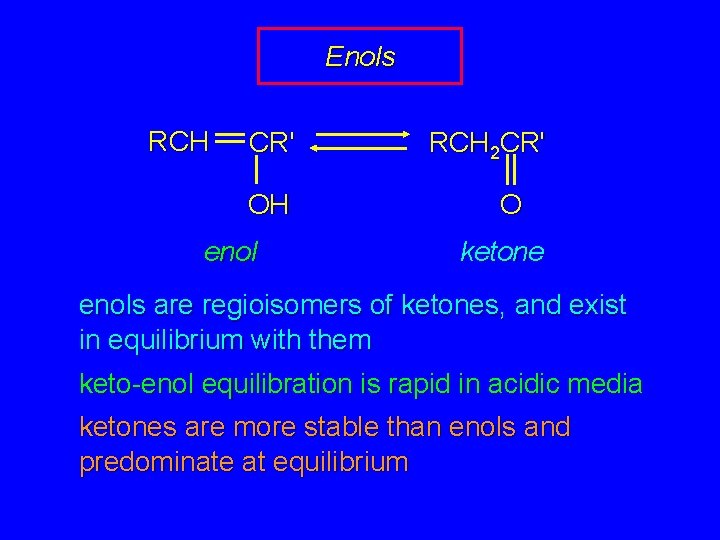
Enols RCH CR' OH enol RCH 2 CR' O ketone enols are regioisomers of ketones, and exist in equilibrium with them keto-enol equilibration is rapid in acidic media ketones are more stable than enols and predominate at equilibrium


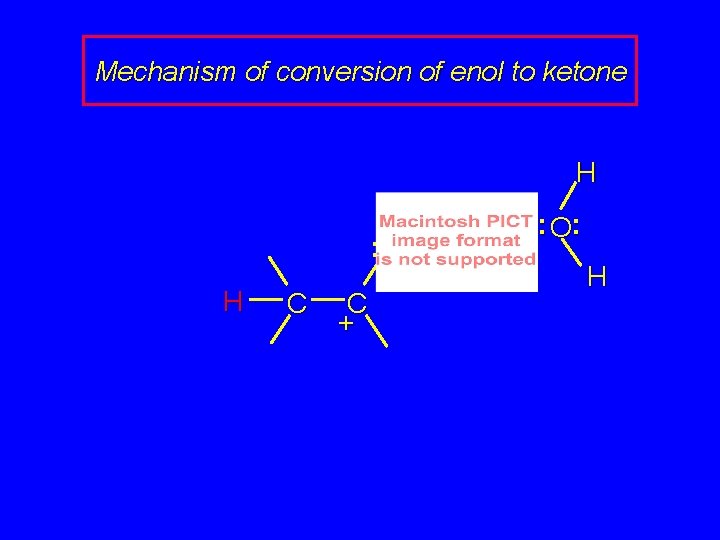
Mechanism of conversion of enol to ketone . . : O H + : O H C H

Mechanism of conversion of enol to ketone . . : O H + : O H C H

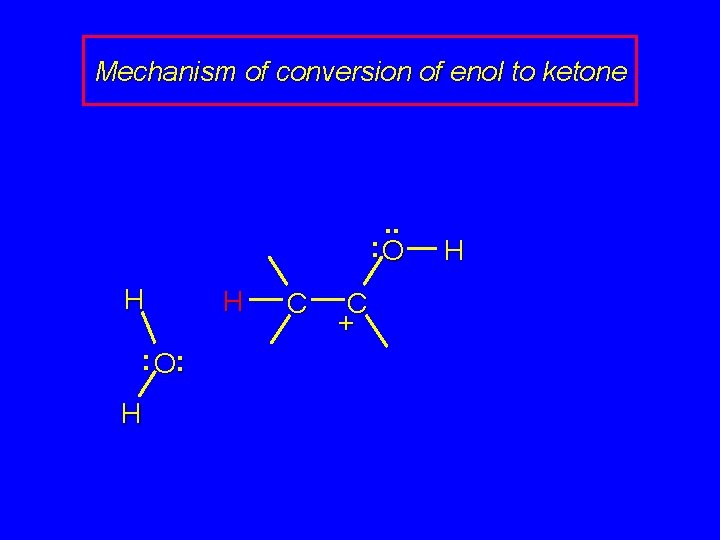
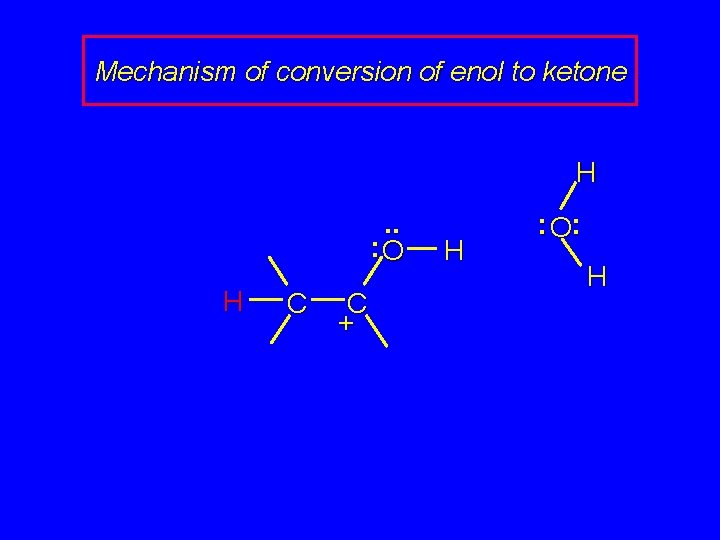
Mechanism of conversion of enol to ketone . . : O H : O: H H C C + H

Mechanism of conversion of enol to ketone H. . : O H C C + H : O: H

Mechanism of conversion of enol to ketone H. . : O H C C + H : O: H

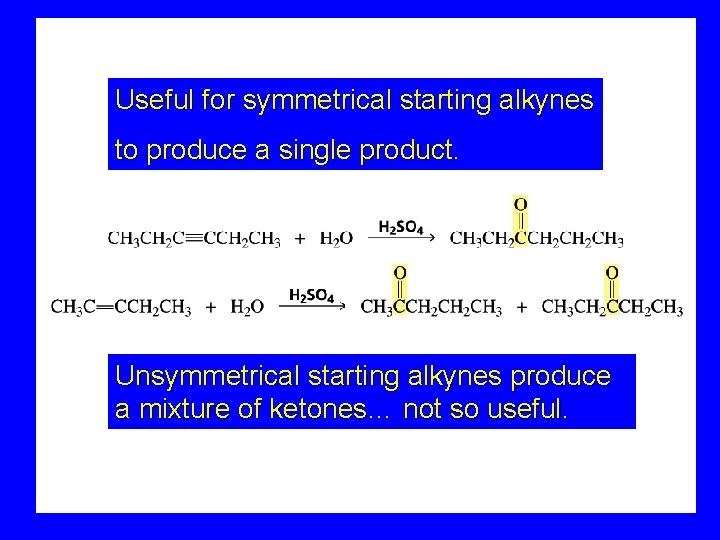
Useful for symmetrical starting alkynes to produce a single product. Unsymmetrical starting alkynes produce a mixture of ketones… not so useful.

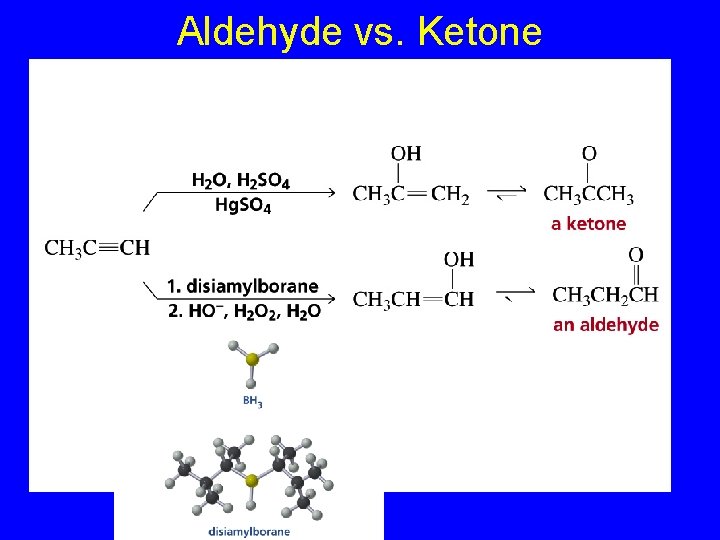
Aldehyde vs. Ketone

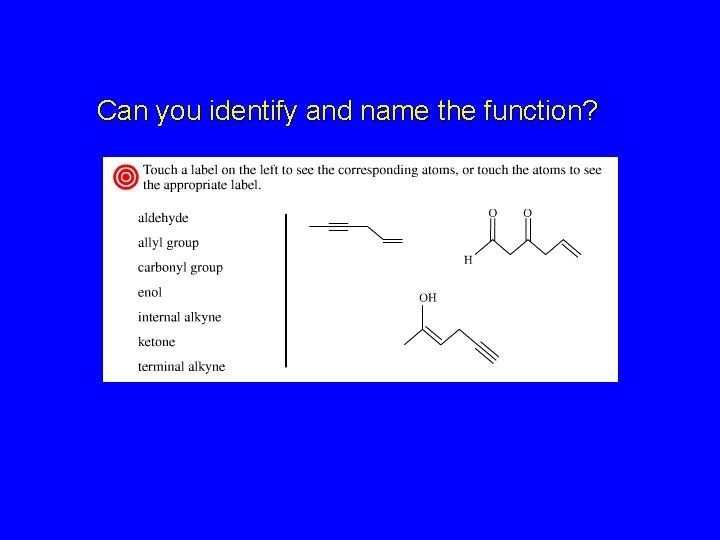
Can you identify and name the function?





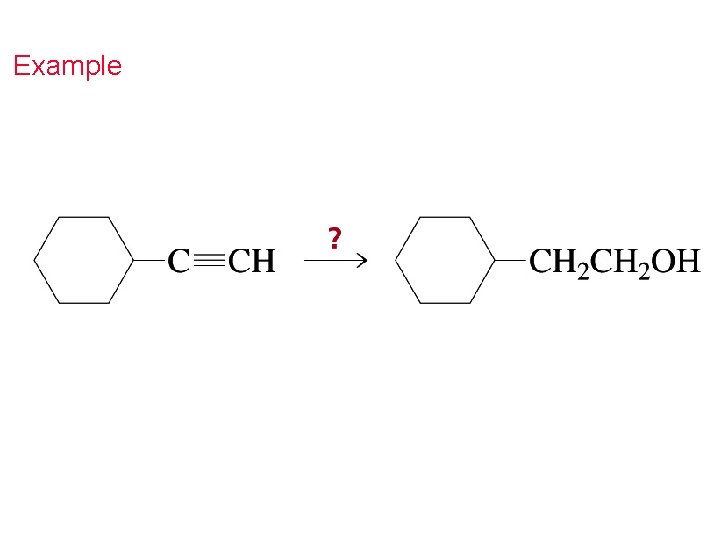
Example

