Synthesis of pyridine 1 By heating the hydrochloride

Synthesis of pyridine 1 – By heating the hydrochloride of pentamethylene diamine and oxidizing the product piperidine with concentrated sulphuric acid at 300 cº. lec 8 1
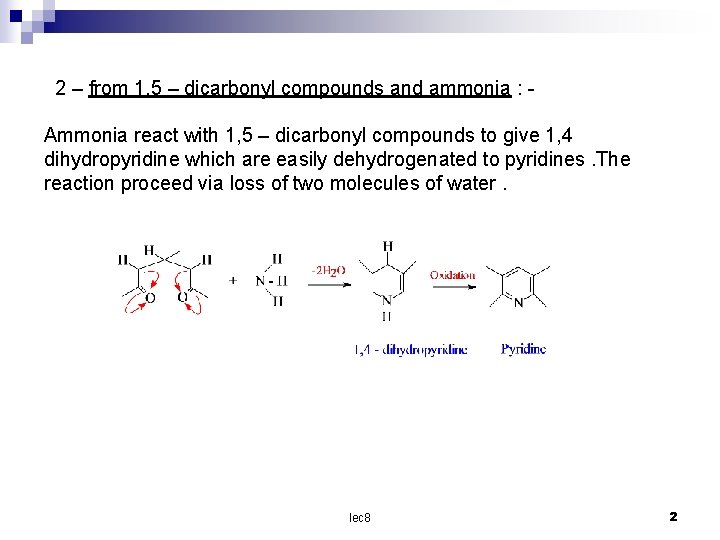
2 – from 1, 5 – dicarbonyl compounds and ammonia : Ammonia react with 1, 5 – dicarbonyl compounds to give 1, 4 dihydropyridine which are easily dehydrogenated to pyridines. The reaction proceed via loss of two molecules of water. lec 8 2
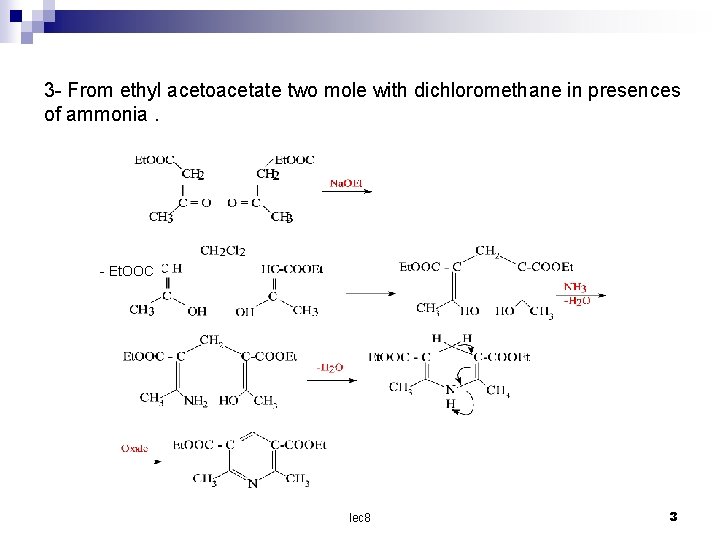
3 - From ethyl acetoacetate two mole with dichloromethane in presences of ammonia. - Et. OOC lec 8 3
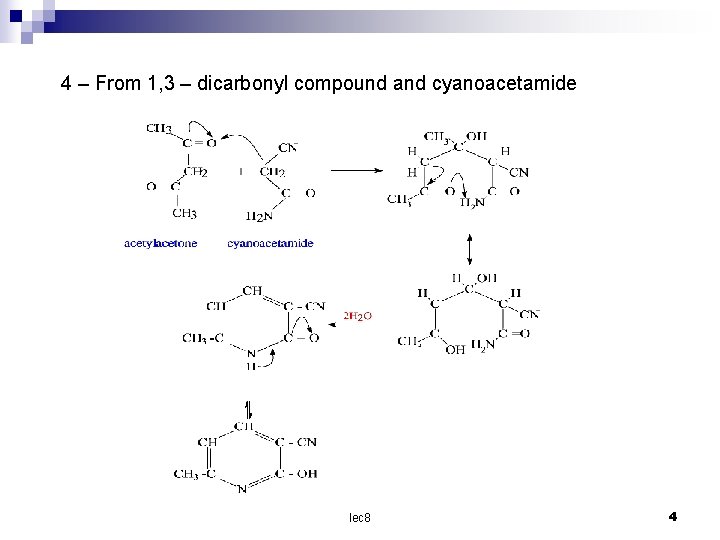
4 – From 1, 3 – dicarbonyl compound and cyanoacetamide lec 8 4

Chemical reactions Basicity of pyridine Pyridine behaves as abase , It react with acids to from fairly stable salt. The reason for the basic character of pyridine is that the nitrogen lone pair being in sp 2 hybrid orbital is not involve in the delocalized π molecular orbital. It is readily available for the formation of a new p N – H bond with proton. Pyridine is a stronger base than pyrrole in which the basicity is reduced by delocalization of the nitrogen lone pair Pyrrole < Pyridine < aliphatic amine Basicity increase lec 8 5
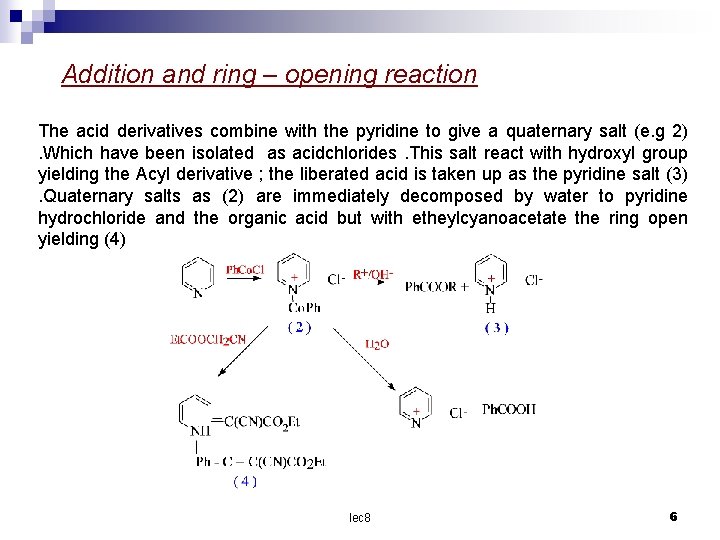
Addition and ring – opening reaction The acid derivatives combine with the pyridine to give a quaternary salt (e. g 2). Which have been isolated as acidchlorides. This salt react with hydroxyl group yielding the Acyl derivative ; the liberated acid is taken up as the pyridine salt (3). Quaternary salts as (2) are immediately decomposed by water to pyridine hydrochloride and the organic acid but with etheylcyanoacetate the ring open yielding (4) lec 8 6

The ring is comparatively easily opened by nucleophilic reagent 2, 4 – Dinitrophenylpyridinum chloride (5) is a colourless crystalline solid which is formed from pyridine and 2, 4 – dinitrochlorobenzene at 100 cº this reaction is reversed at 200 cº with water at 150 cº yield pyridine hydrochloride and 2, 4 – dinitrophenol , but with cold aqueous alkali a deep red compound (6) is formed which on successive treatment with dilute aniline and acid yields 2, 4 – dinitroaniline and glutaconic aldehyde dianil (7). lec 8 7
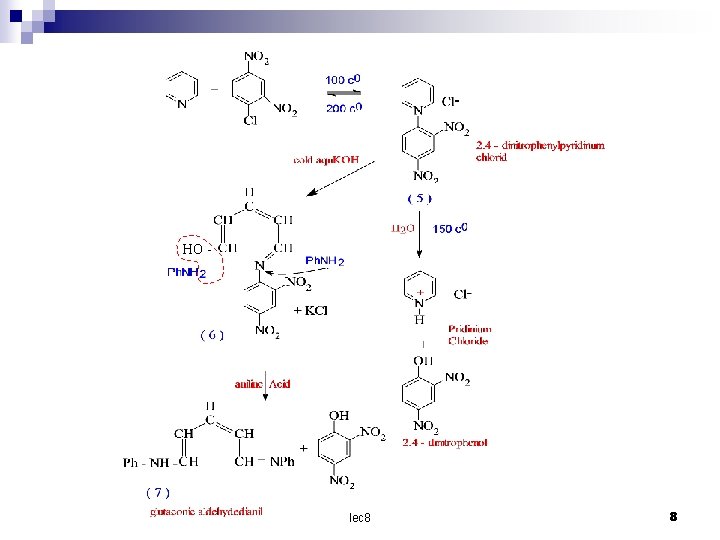
HO - lec 8 8

Reduction Pyridine is easily reduce to hexahydropyridin or piperidine by a variety of method including hydrogen over Raney nickel , rubidium at 60 ºc palladium charcoal with acetic acid. 1, 4 – dihydropyridine has however been obtained by the reduction of pyridine with trimethylsilane. The pyridine is cleaved by ultrasonic waves giving acetylene and hydrogen cyanide. lec 8 9
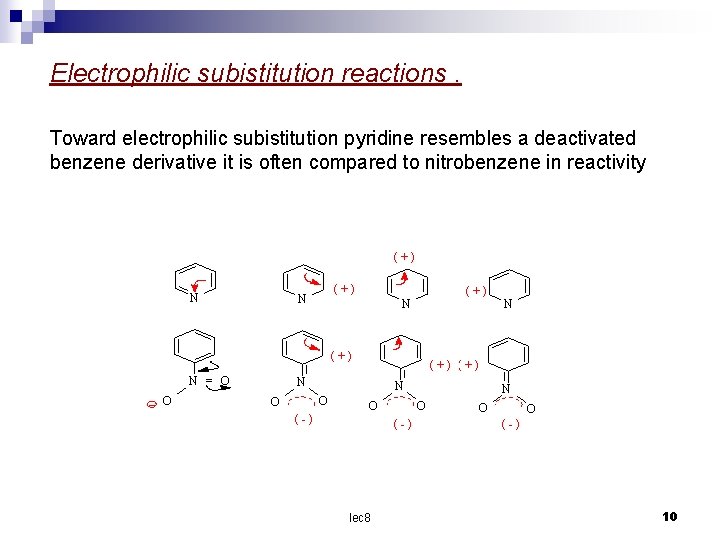
Electrophilic subistitution reactions. Toward electrophilic subistitution pyridine resembles a deactivated benzene derivative it is often compared to nitrobenzene in reactivity (+) N = O O (+) N N O O (-) lec 8 N O O (-) 10
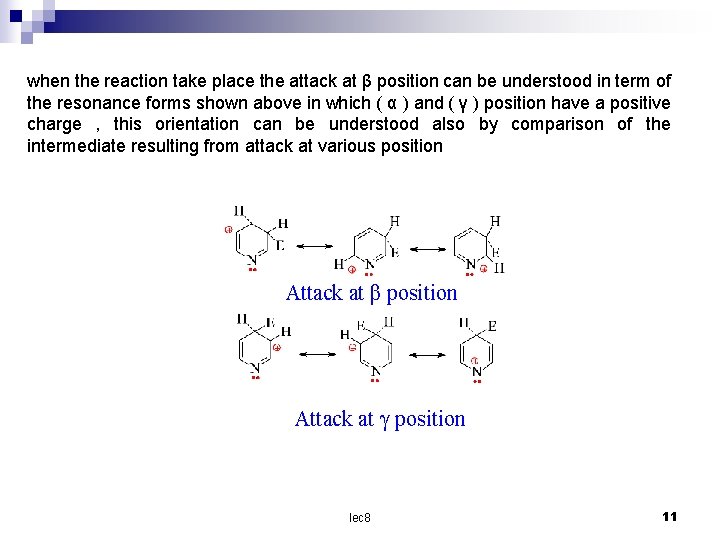
when the reaction take place the attack at β position can be understood in term of the resonance forms shown above in which ( α ) and ( γ ) position have a positive charge , this orientation can be understood also by comparison of the intermediate resulting from attack at various position Attack at β position Attack at γ position lec 8 11
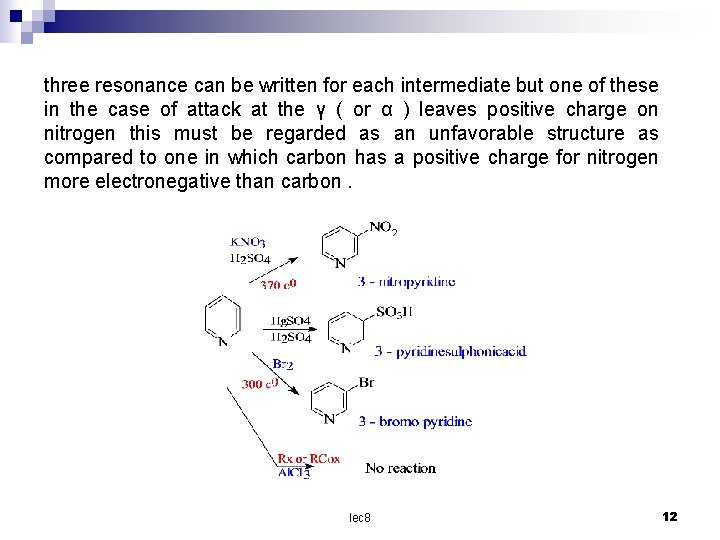
three resonance can be written for each intermediate but one of these in the case of attack at the γ ( or α ) leaves positive charge on nitrogen this must be regarded as an unfavorable structure as compared to one in which carbon has a positive charge for nitrogen more electronegative than carbon. lec 8 12
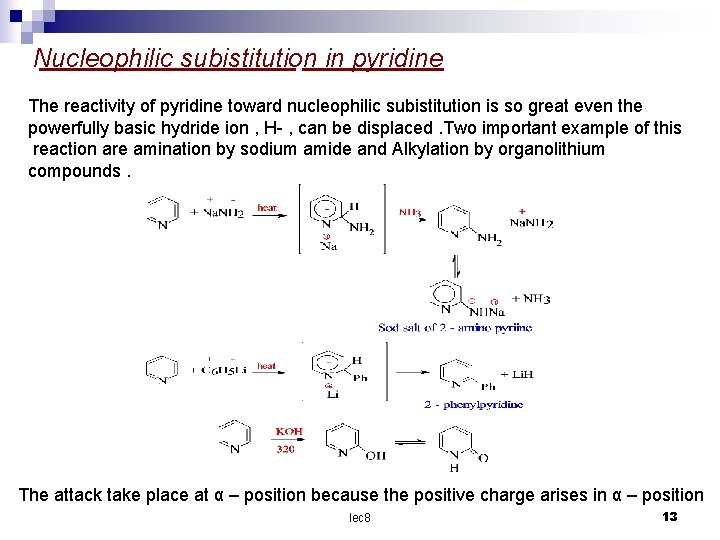
Nucleophilic subistitution in pyridine The reactivity of pyridine toward nucleophilic subistitution is so great even the powerfully basic hydride ion , H- , can be displaced. Two important example of this reaction are amination by sodium amide and Alkylation by organolithium compounds. The attack take place at α – position because the positive charge arises in α – position lec 8 13

Pyrylium salt and pyrans Very few simple derivatives of the aromatic pyrylium cation (1) are known , although benzopyrylium are widely distributed as flower petal colouring matters. The potentially very reactive 4 -pyran (3) has been obtained recently and the pyranes ( 4 and 5 ) are well known. 2, 3 – Dihydro – 4 – pyran (6) has received some attention now it is easily available , and tetrahydropyran ( 7 ) is used as a synthetic intermediate. The sulphur analogues of these compounds have received little attention until very recently lec 8 14

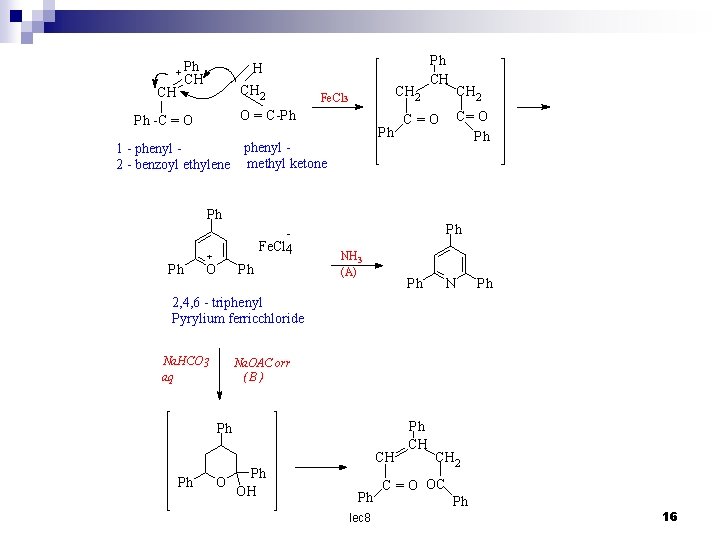
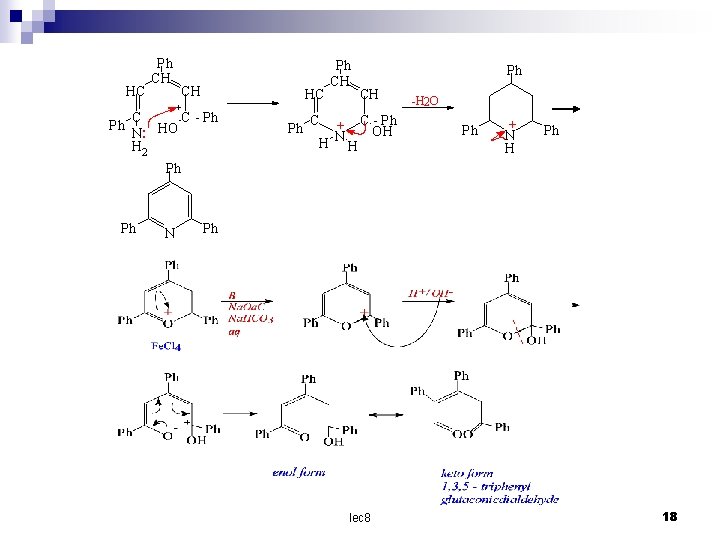
Pyrylium salts The sodium salt of glutaconic aldehyde with perchloric acid at – 20 cº gives ared oxonium salt , which on standing at o cº cyclizes to the colourless pyrylium perchlorate (8). This perchlorate has received little attention , but with ammonia it yields 2, 4, 6 – triphenylpyrylium ferrichloride ( 9 ) can be prepared easily. It is stable in acid solution and nitrates. with ammonia it yields 2, 4, 6 – triphenylpyridine while with alkali the ring is opened , yielding (II) through the intermediate (10 ) - + CH CH CH = O Na O CH 6 4 O CH H Cl. O 4 O 2 - HO CH CH + CH = OH CH c 00 H-2 O - Cl. O 4 sod. salt of glutaconic aldehyde Oxonium salt 5 CH 3 2 - Cl. O 4 1 (8) Pyrylium perchlorate lec 8 15
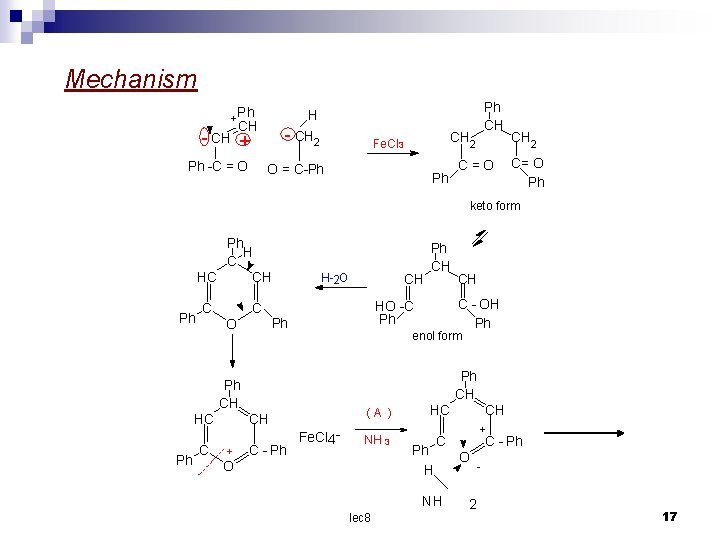
+ CH Ph CH H CH 2 Fe. Cl 3 O = C-Ph Ph -C = O Ph phenyl 1 - phenyl 2 - benzoyl ethylene methyl ketone Ph CH Ph - Ph Fe. Cl 4 O Ph C=O Ph + CH 2 NH 3 (A) Ph N Ph 2, 4, 6 - triphenyl Pyrylium ferricchloride Na. HCO 3 aq Na. OAC orr (B) Ph CH Ph OH Ph lec 8 Ph CH CH 2 C = O OC Ph 16
Mechanism Ph CH H + - CH 2 Ph -C = O O = C-Ph CH 2 Fe. Cl 3 Ph C=O CH 2 C= O Ph keto form Ph H C HC CH Ph C C O Ph Ph C enol form Ph CH CH + CH C - OH Ph HO -C Ph Ph CH HC CH H-2 O Ph CH C - Ph Fe. Cl 4 - (A ) HC NH 3 C O Ph H NH lec 8 CH + C - Ph O 2 - 17
HC Ph CH CH HC + Ph C C - Ph HO N: H 2 Ph C H Ph CH + N Ph CH C - Ph OH H -H 2 O Ph + N H Ph Ph Ph N Ph lec 8 18

Reaction of pyrylium cation A–Reaction with electrophilic reagent 2, 4, 6 –Triphenylpyrylium undergoes exchange at 3 – and 5 – position in hot deuteroacetic acid Ph Ph ACDO Ph + O Ph -) ( + ACO Ph OAC Ph O Ph D + Ph 5 + O D H OAC Ph B – No nitration of pyrylium are known lec 8 19
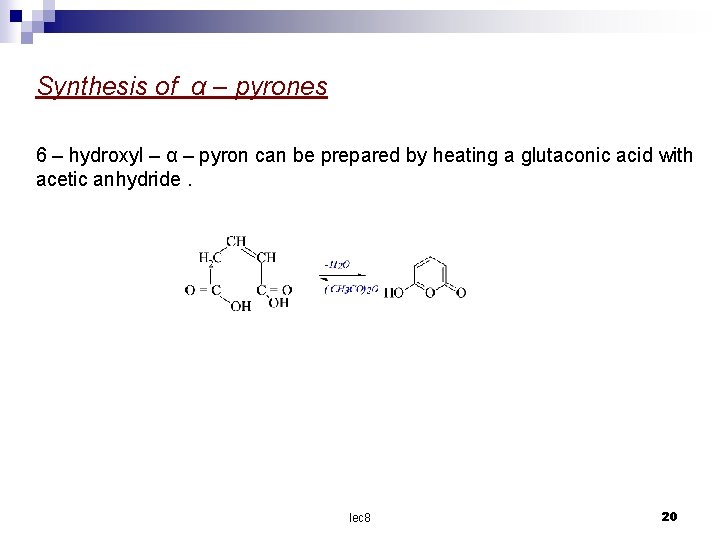
Synthesis of α – pyrones 6 – hydroxyl – α – pyron can be prepared by heating a glutaconic acid with acetic anhydride. lec 8 20
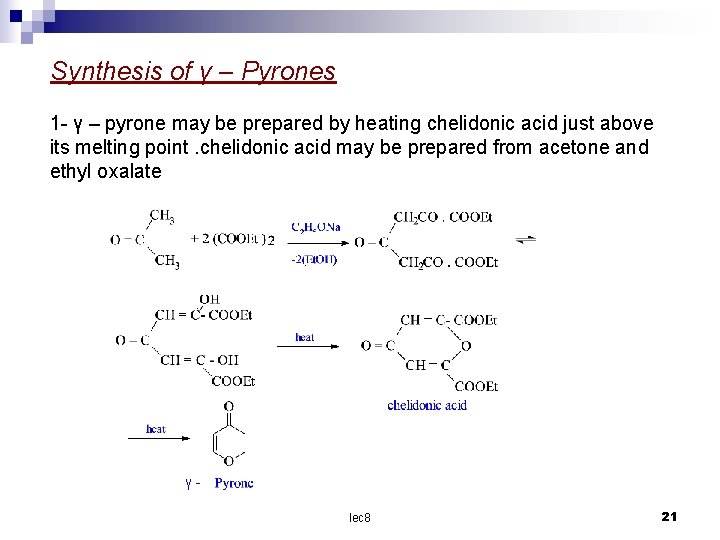
Synthesis of γ – Pyrones 1 - γ – pyrone may be prepared by heating chelidonic acid just above its melting point. chelidonic acid may be prepared from acetone and ethyl oxalate γlec 8 21

2 – The dimethyl γ – pyrone may be prepared from the copper salt of ethylacetoacetate as follows lec 8 22

Reaction of α and γ –Pyrone. α lec 8 23
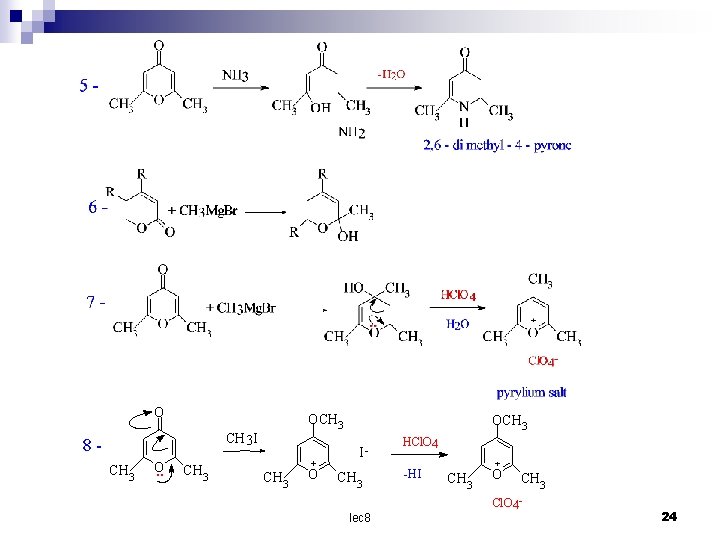
O OCH 3 I 8 CH 3 O. . CH 3 + O OCH 3 I- CH 3 lec 8 HCl. O 4 -H I + CH 3 O CH 3 Cl. O 4 - 24
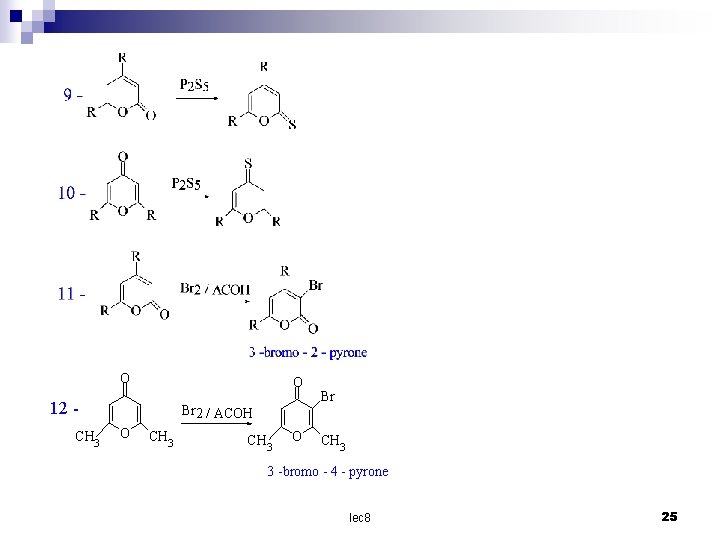
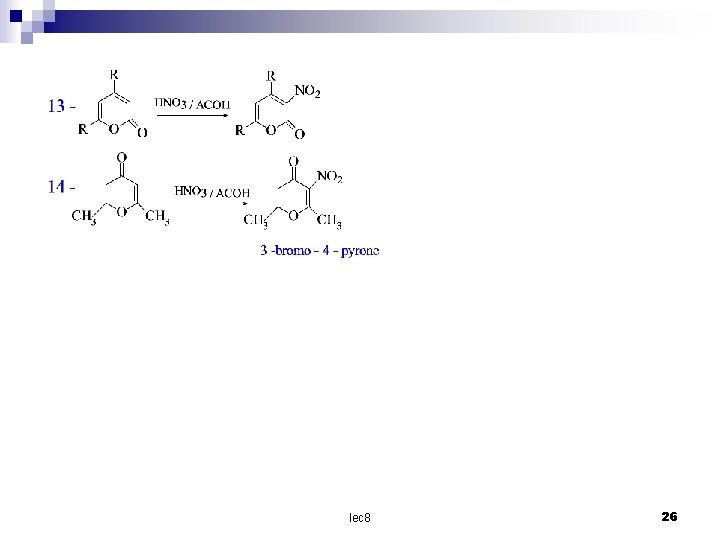
O O 12 CH 3 Br 2 / ACOH O CH 3 O Br CH 3 3 -bromo - 4 - pyrone lec 8 25
lec 8 26
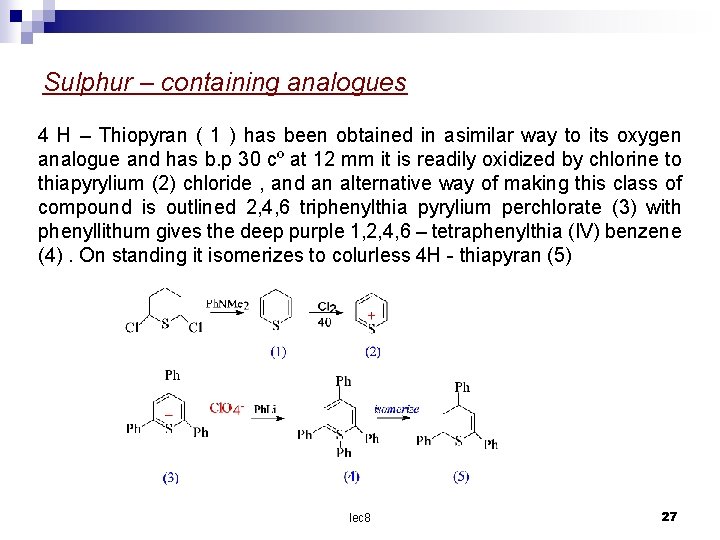
Sulphur – containing analogues 4 H – Thiopyran ( 1 ) has been obtained in asimilar way to its oxygen analogue and has b. p 30 cº at 12 mm it is readily oxidized by chlorine to thiapyrylium (2) chloride , and an alternative way of making this class of compound is outlined 2, 4, 6 triphenylthia pyrylium perchlorate (3) with phenyllithum gives the deep purple 1, 2, 4, 6 – tetraphenylthia (IV) benzene (4). On standing it isomerizes to colurless 4 H - thiapyran (5) lec 8 27

1 - methyl – 3, 5 – diphenylthia(IV)benzene 1 - oxide (6) has been obtained as indicated below. It has m. p 148º and can be sublimed at near this temperature at 0 – 0. 5 mm pressure. The compound is therefore very much more stable than (IV) benzene such as (4) lec 8 28
- Slides: 28