Alkynes Alkynes Hydrocarbons with a carboncarbon triple bond

Alkynes

Alkynes Hydrocarbons with a carbon–carbon triple bond are alkynes. Noncyclic alkynes have the molecular formula Cn. H 2 n-2. Acetylene (HC≡ CH) is the simplest alkyne. Compounds with the triple bond at the end of a carbon chain (RC≡ CH) are monosubstituted, or terminal, alkynes. Disubstituted alkynes (RC≡ CR′) have internal triple bonds.
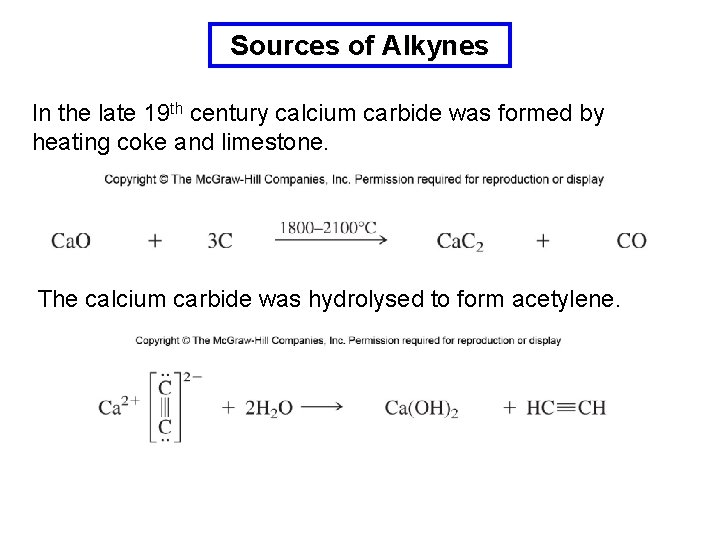
Sources of Alkynes In the late 19 th century calcium carbide was formed by heating coke and limestone. The calcium carbide was hydrolysed to form acetylene.

Sources of Alkynes One alternative synthesis is the dehydration of ethylene. The endothermic reaction favors acetylene at high temperature. Enzymes known as acetylenases catalyze this reaction in nature.
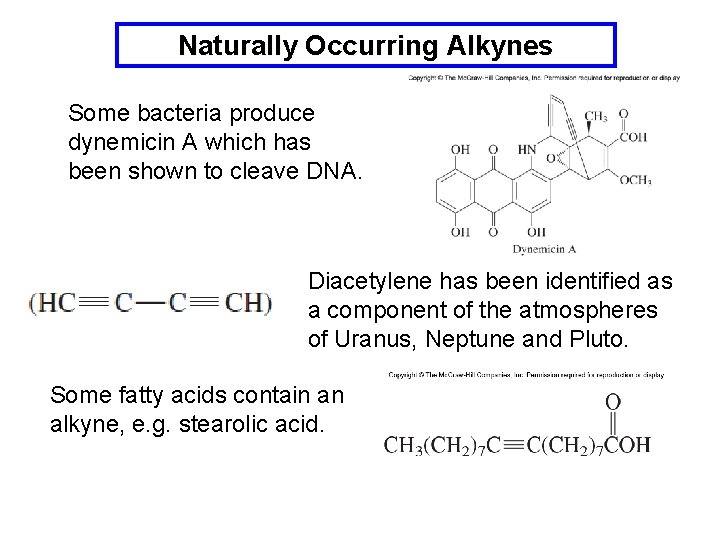
Naturally Occurring Alkynes Some bacteria produce dynemicin A which has been shown to cleave DNA. Diacetylene has been identified as a component of the atmospheres of Uranus, Neptune and Pluto. Some fatty acids contain an alkyne, e. g. stearolic acid.
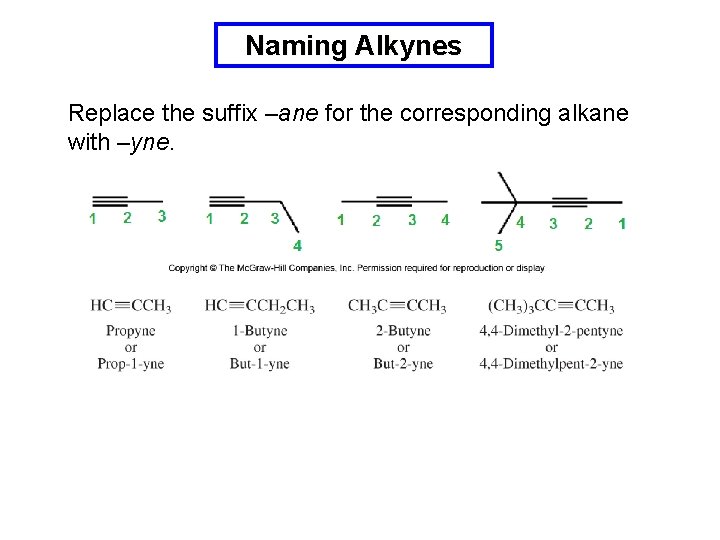
Naming Alkynes Replace the suffix –ane for the corresponding alkane with –yne.

Naming Alkynes Compounds that contain both an alkene and an alkyne are numbered to give the first multiple bond the lowest number and are named as an –enynes. hept-1 -en-5 -yne

Physical Properties of Alkynes resemble alkanes and alkenes in physical properties. Low density and low water solubility. Boiling points similar to the corresponding alkane.
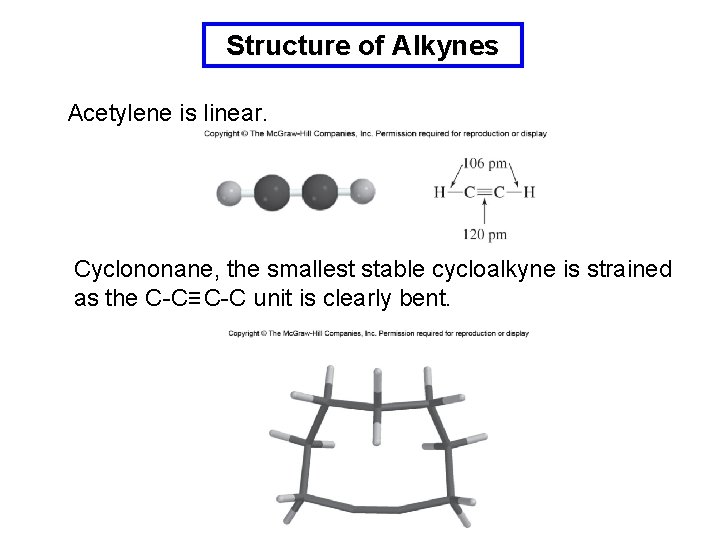
Structure of Alkynes Acetylene is linear. Cyclononane, the smallest stable cycloalkyne is strained as the C-C≡ C-C unit is clearly bent.
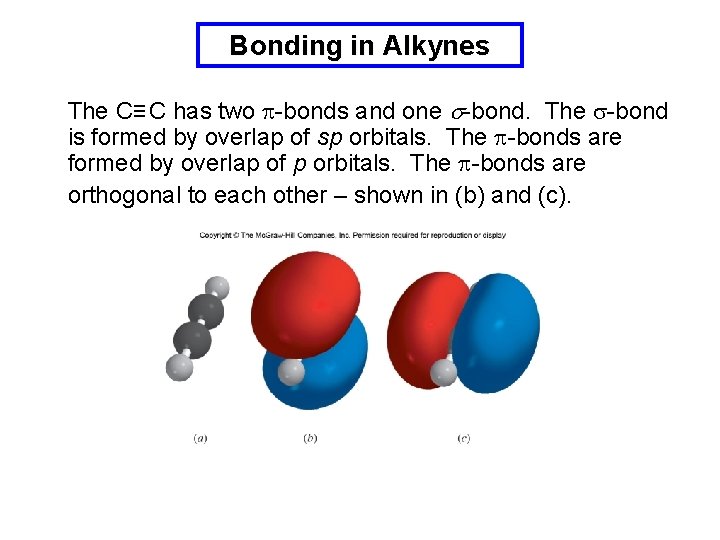
Bonding in Alkynes The C≡ C has two p-bonds and one s-bond. The s-bond is formed by overlap of sp orbitals. The p-bonds are formed by overlap of p orbitals. The p-bonds are orthogonal to each other – shown in (b) and (c).
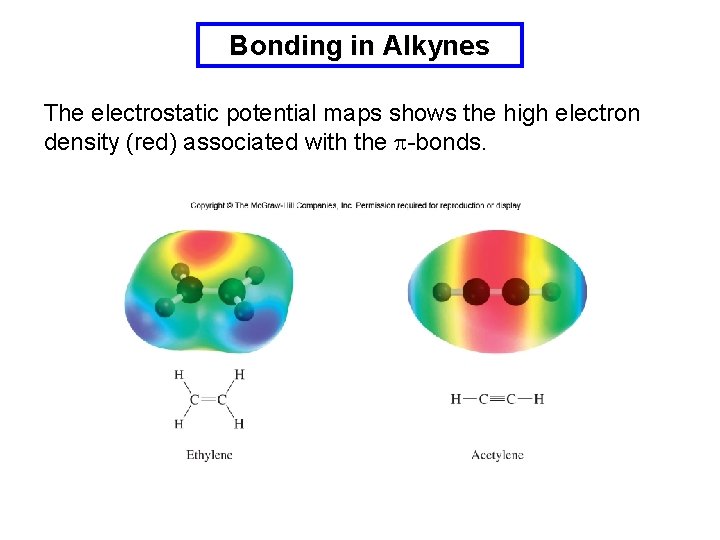
Bonding in Alkynes The electrostatic potential maps shows the high electron density (red) associated with the p-bonds.
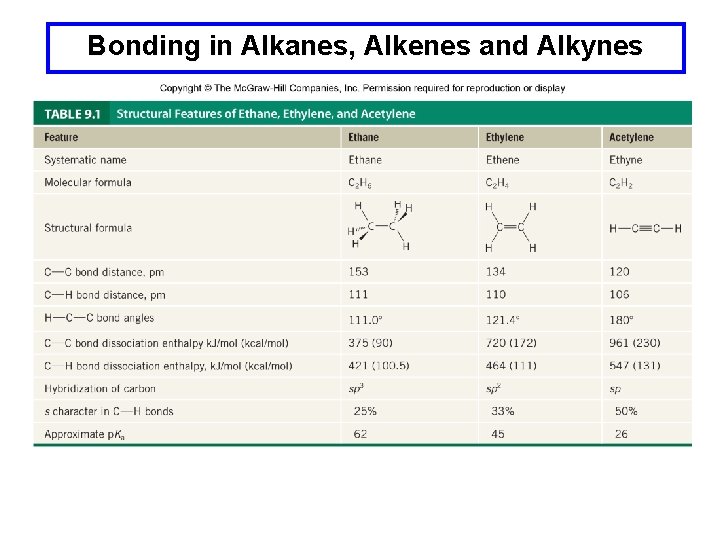
Bonding in Alkanes, Alkenes and Alkynes

Bonding in Alkanes, Alkenes and Alkynes For the series ethane → ethylene → acetylene the general trends are: 1. The geometry at carbon changes from tetrahedral → trigonal planar → linear. 2. The C-C and C-H bonds become shorter and stronger. 3. The acidity of the C-H bonds increases.
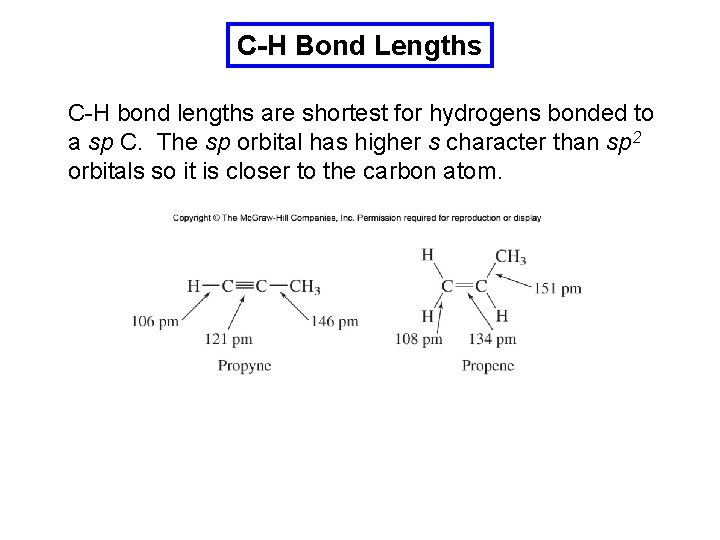
C-H Bond Lengths C-H bond lengths are shortest for hydrogens bonded to a sp C. The sp orbital has higher s character than sp 2 orbitals so it is closer to the carbon atom.
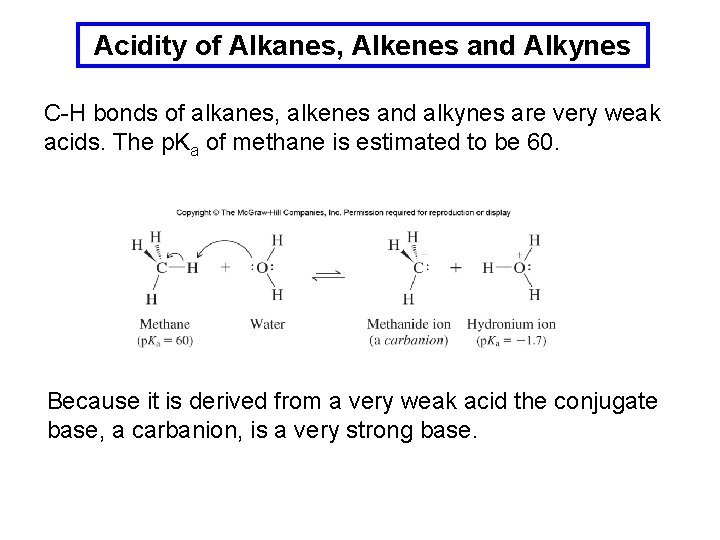
Acidity of Alkanes, Alkenes and Alkynes C-H bonds of alkanes, alkenes and alkynes are very weak acids. The p. Ka of methane is estimated to be 60. Because it is derived from a very weak acid the conjugate base, a carbanion, is a very strong base.
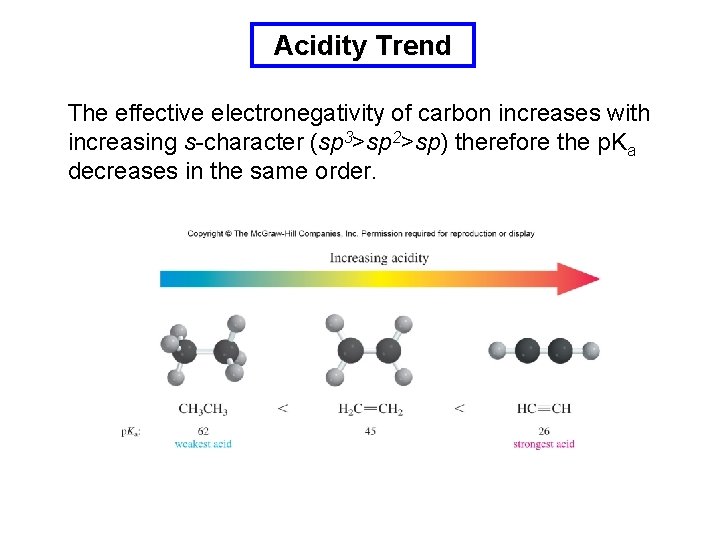
Acidity Trend The effective electronegativity of carbon increases with increasing s-character (sp 3>sp 2>sp) therefore the p. Ka decreases in the same order.

Acidity of Alkynes Terminal alkynes RC≡ CH are similar to acetylene in acidity. After deprotonation the acetylide electron pair is in a sp orbital.

Acid-Base Reactions of Alkynes In order to form the acetylide anion the base must be strong enough. The conjugate acid of the base should have p. Ka greater than the p. Ka of the alkyne. Hydroxide is too weak and the equilibrium lies to the left. Amide is strong enough and the equilibrium lies to the right.

Preparation of Alkynes by Alkylation of Acetylene and Terminal Alkynes

Acetylide Anions as Nucleophiles Acetylide anions react with methyl and primary alkyl halides to form substituted alkynes. This is an SN 2 type of reaction.
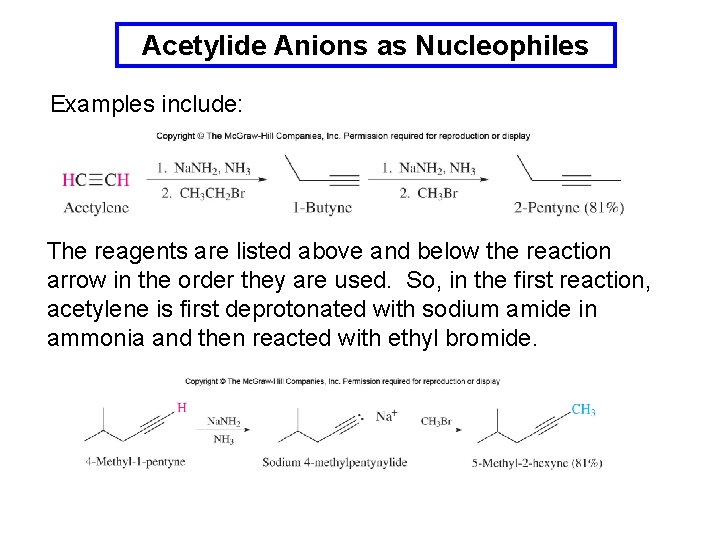
Acetylide Anions as Nucleophiles Examples include: The reagents are listed above and below the reaction arrow in the order they are used. So, in the first reaction, acetylene is first deprotonated with sodium amide in ammonia and then reacted with ethyl bromide.

Elimination Reactions with Acetylide Anions Acetylide anions are very basic so they act as bases if the alkyl halide is secondary or tertiary.

Preparation of Alkynes by Elimination Alkynes can be prepared by double dehydrohalogenation of either geminal dihalides or vicinal dihalides.
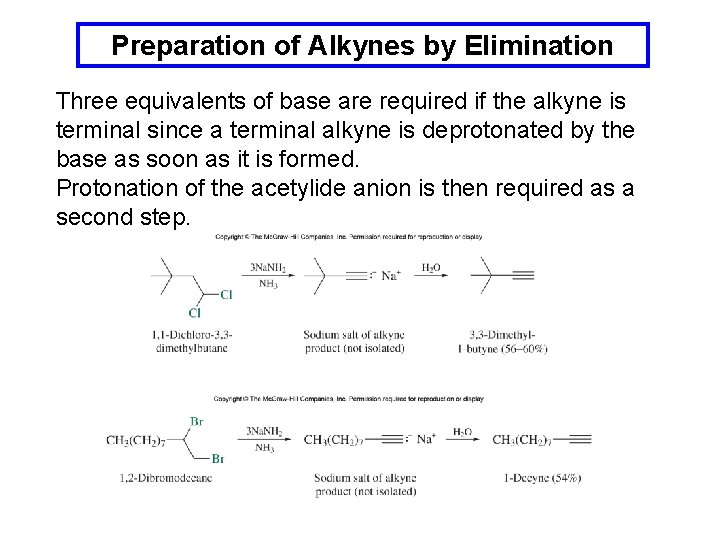
Preparation of Alkynes by Elimination Three equivalents of base are required if the alkyne is terminal since a terminal alkyne is deprotonated by the base as soon as it is formed. Protonation of the acetylide anion is then required as a second step.
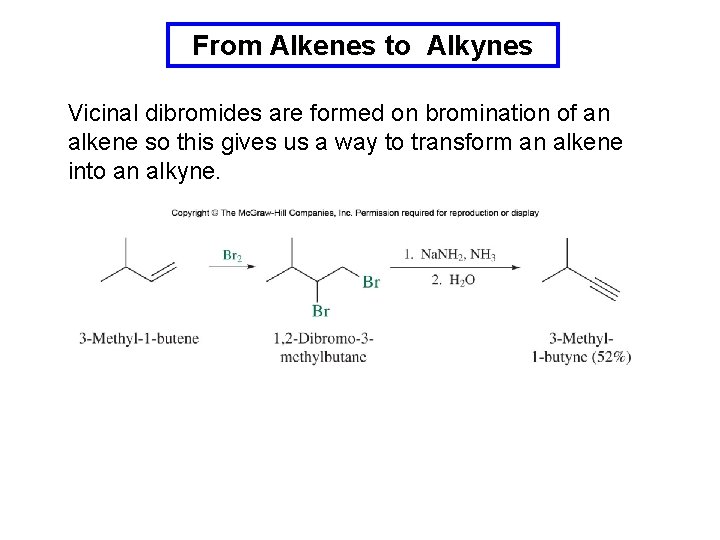
From Alkenes to Alkynes Vicinal dibromides are formed on bromination of an alkene so this gives us a way to transform an alkene into an alkyne.

Reactions of Alkynes Reactions of alkynes are similar to reaction of alkenes: hydrogenation, hydration, halogenation, ozonolysis.

Hydrogenation of Alkynes are reduced to alkanes using hydrogen and a transition metal catalyst. For example:

Relative Stability of Alkynes The heat of hydrogenation of 1 - and 2 -butyne can be used to determine the relative stability of an internal and a terminal alkyne (both yield butane as product). Less heat is released by 2 -butyne so that is the more stable isomer.
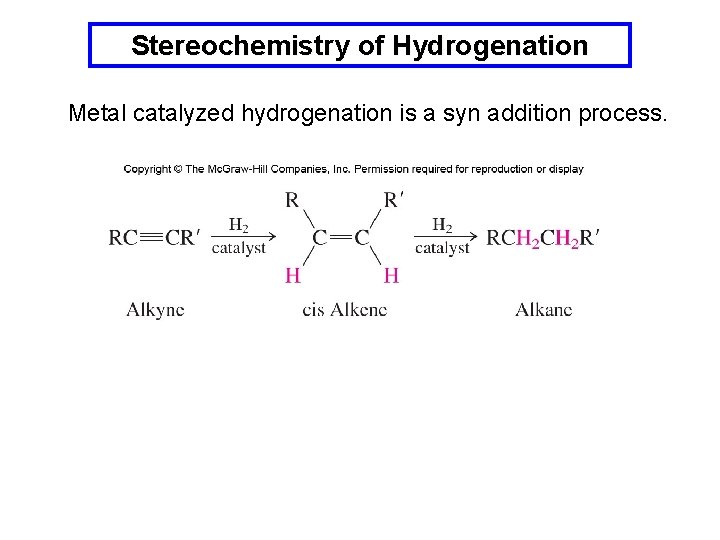
Stereochemistry of Hydrogenation Metal catalyzed hydrogenation is a syn addition process.
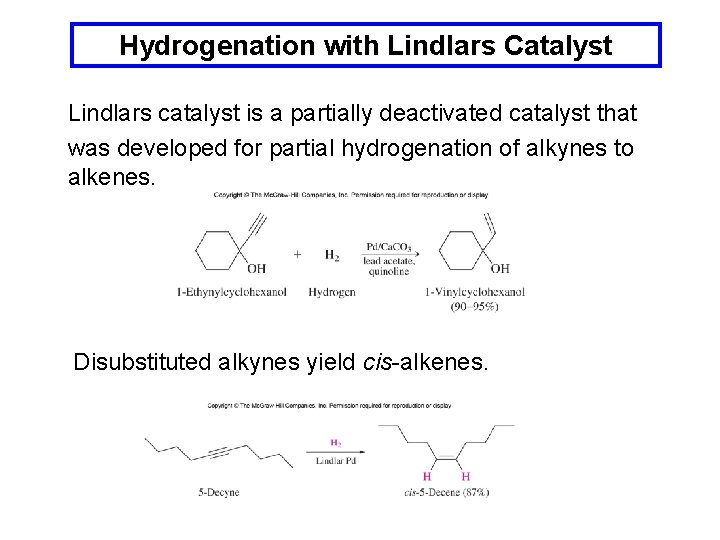
Hydrogenation with Lindlars Catalyst Lindlars catalyst is a partially deactivated catalyst that was developed for partial hydrogenation of alkynes to alkenes. Disubstituted alkynes yield cis-alkenes.
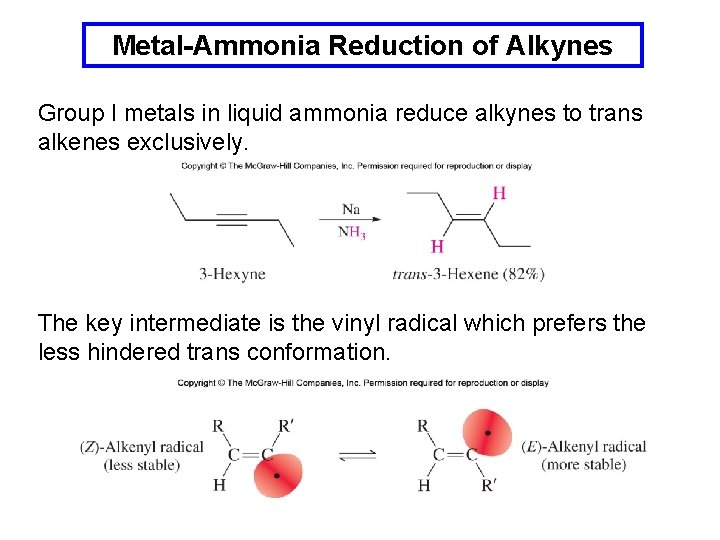
Metal-Ammonia Reduction of Alkynes Group I metals in liquid ammonia reduce alkynes to trans alkenes exclusively. The key intermediate is the vinyl radical which prefers the less hindered trans conformation.

Mechanism of Metal-Ammonia Reduction Sodium atoms dissolved in liquid ammonia dissociate into sodium ions and electrons, both solvated by ammonia. The solvated electrons are represented in e–(am). Step 1. Electron transfer. Step 2. Proton transfer.

Mechanism of Metal-Ammonia Reduction Step 3. Electron transfer. Step 4. Proton transfer.

Addition of Hydrogen Halides Hydrogen halides add to alkynes to form alkenyl halides. The reaction follows Markovkovs Rule (the proton adds to the carbon that initially has the most hydrogens).
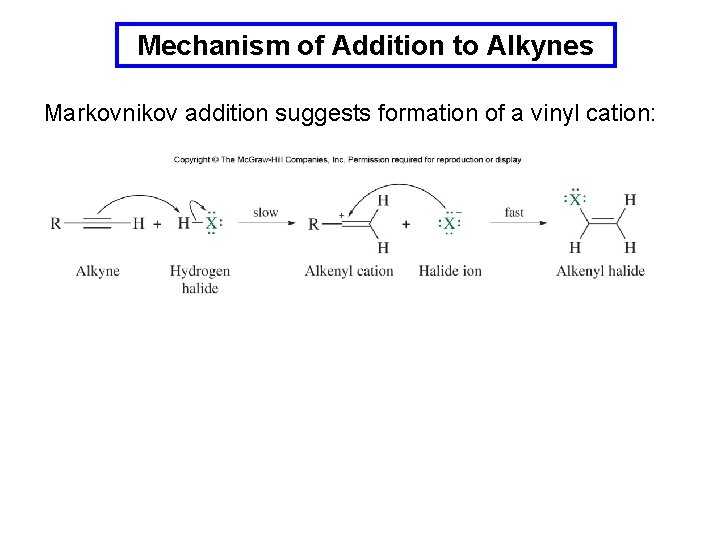
Mechanism of Addition to Alkynes Markovnikov addition suggests formation of a vinyl cation:
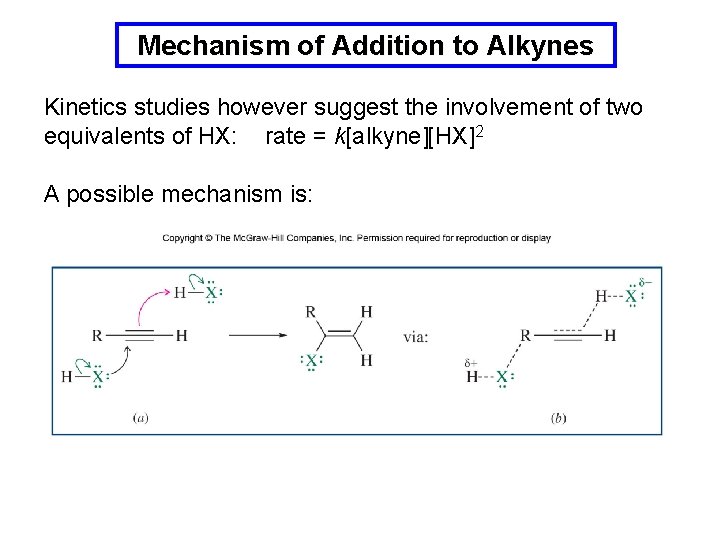
Mechanism of Addition to Alkynes Kinetics studies however suggest the involvement of two equivalents of HX: rate = k[alkyne][HX]2 A possible mechanism is:
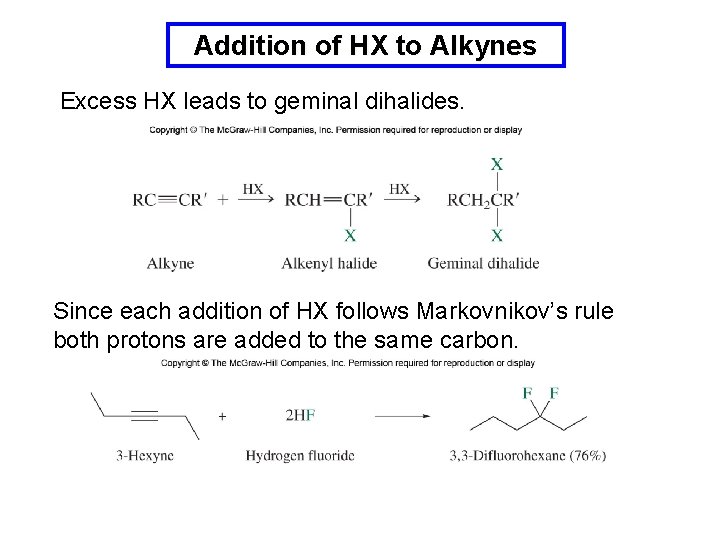
Addition of HX to Alkynes Excess HX leads to geminal dihalides. Since each addition of HX follows Markovnikov’s rule both protons are added to the same carbon.
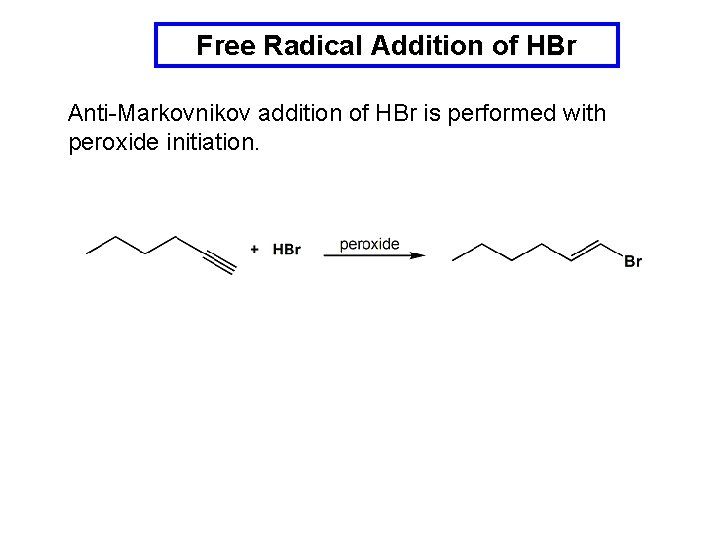
Free Radical Addition of HBr Anti-Markovnikov addition of HBr is performed with peroxide initiation.
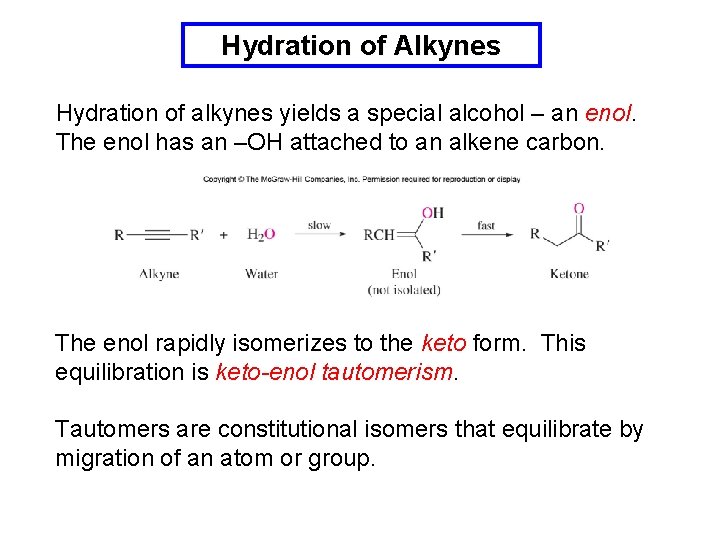
Hydration of Alkynes Hydration of alkynes yields a special alcohol – an enol. The enol has an –OH attached to an alkene carbon. The enol rapidly isomerizes to the keto form. This equilibration is keto-enol tautomerism. Tautomers are constitutional isomers that equilibrate by migration of an atom or group.
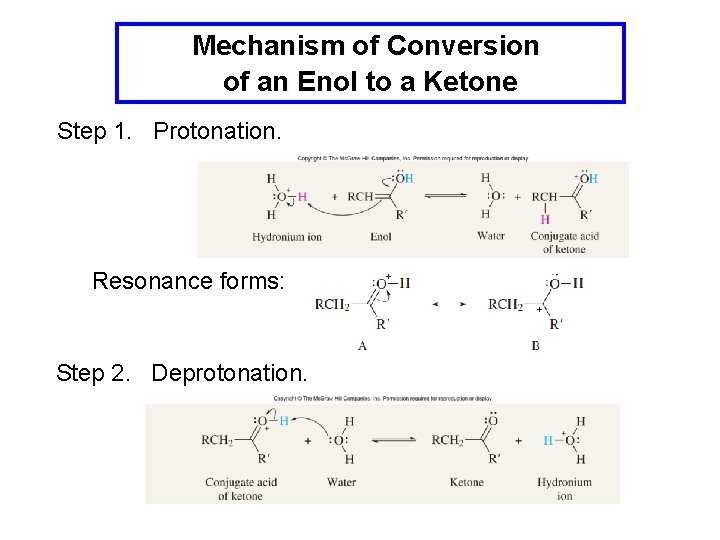
Mechanism of Conversion of an Enol to a Ketone Step 1. Protonation. Resonance forms: Step 2. Deprotonation.

Hydration of Alkynes In general the keto form is more stable so it will be drawn as the product of a hydration reaction. The reaction follows Markovnikov’s rule so terminal alkynes yield methyl ketones not aldehydes.
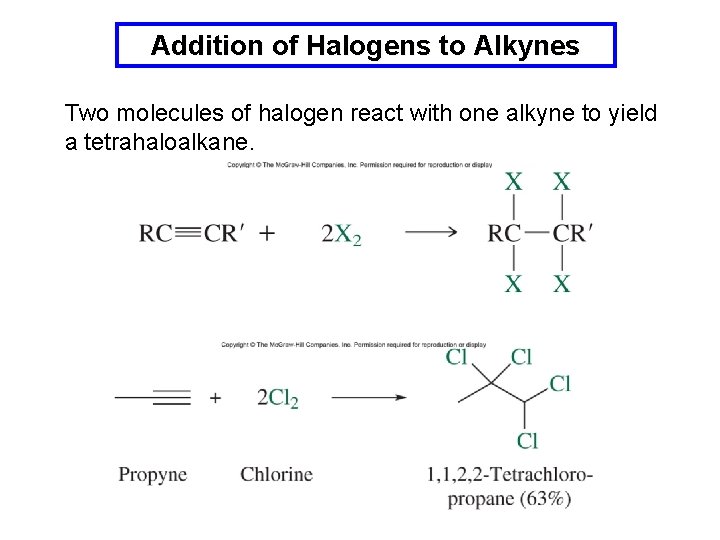
Addition of Halogens to Alkynes Two molecules of halogen react with one alkyne to yield a tetrahaloalkane.
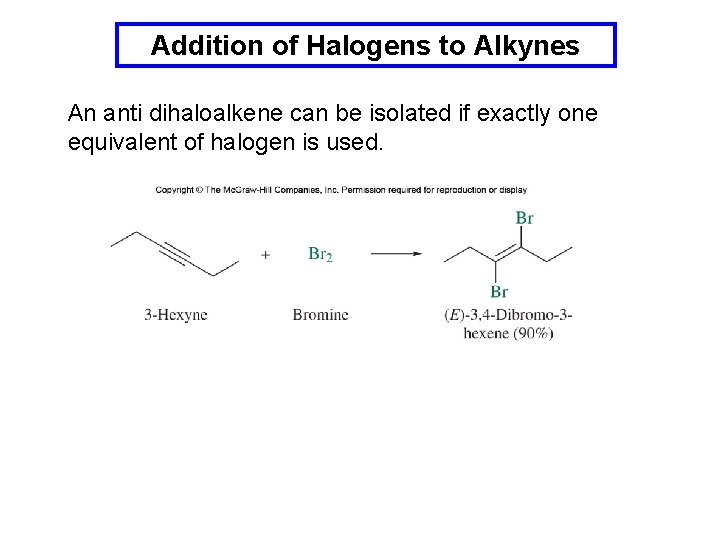
Addition of Halogens to Alkynes An anti dihaloalkene can be isolated if exactly one equivalent of halogen is used.

What Could be Made From Acetylene but Isn’t We can write simple reactions to produce these useful monomers from acetylene. In reality they are actually made from ethylene because it is so much cheaper and easier to prepare than acetylene!
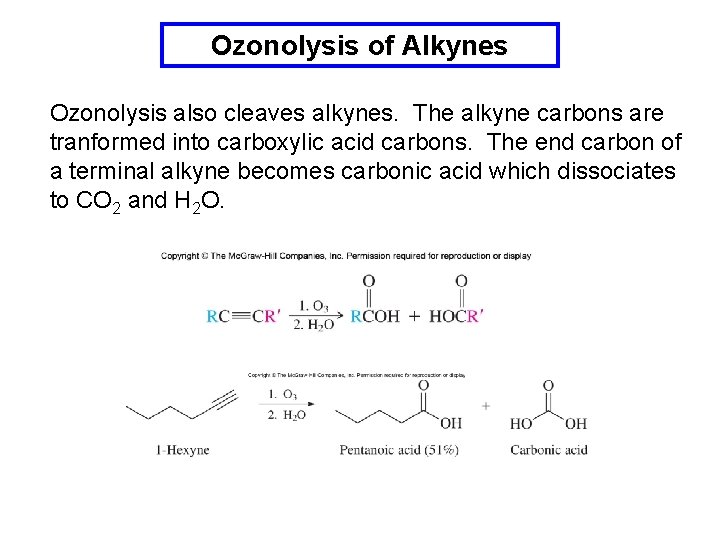
Ozonolysis of Alkynes Ozonolysis also cleaves alkynes. The alkyne carbons are tranformed into carboxylic acid carbons. The end carbon of a terminal alkyne becomes carbonic acid which dissociates to CO 2 and H 2 O.

Alkynes in Synthesis and Retrosynthesis

Applications of Alkynes in Synthesis Example: Outline a synthesis of 1, 2 -epoxybutane using ethyl bromide and acetylene as sources for all the carbon atoms. The epoxide may be formed from the corresponding alkene: The alkene could be made from the corresponding alkyne which could itself be prepared from an acetylide anion:
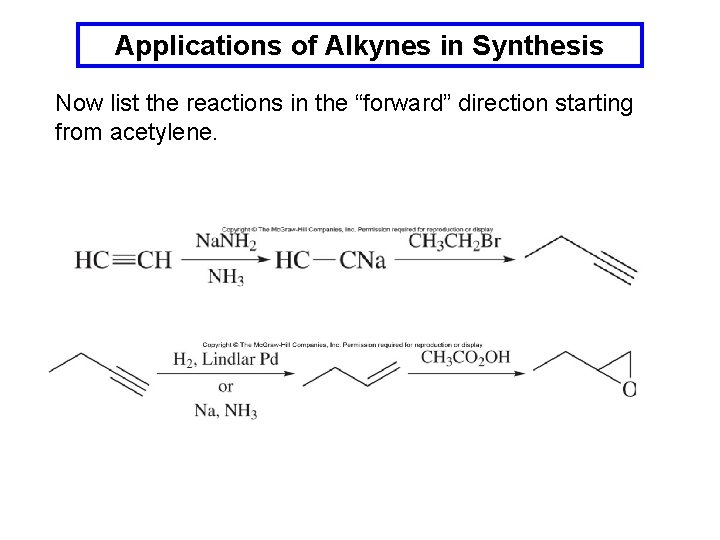
Applications of Alkynes in Synthesis Now list the reactions in the “forward” direction starting from acetylene.
- Slides: 48