ALKENES AND ALKYNES Alkenes contain one or more






- Slides: 19

ALKENES AND ALKYNES

• Alkenes contain one or more double bonds • Alkynes contain one or more triple bonds • Since alkenes and alkynes are NOT bonded to the maximum possible number of atoms, these compounds are often referred to as unsaturated hydrocarbons Boiling points and melting points: alkynes < alkenes < alkanes Reactivity: alkynes > alkenes > alkanes

• alkenes are used to synthesize polymers because they are reactive • alkynes are very unstable. As a result, they rarely occur in nature • When naming these molecules, there a few extra rules to follow: your main chain must include double or triple bonds at lowest position number (indicate position #) when numbering the main chain, double and triple bonds have priority over alkyl groups

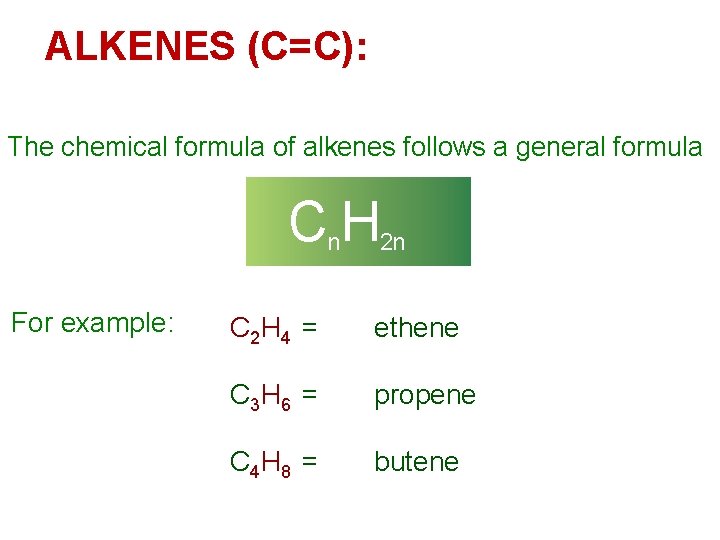
ALKENES (C=C): The chemical formula of alkenes follows a general formula Cn. H 2 n For example: C 2 H 4 = ethene C 3 H 6 = propene C 4 H 8 = butene

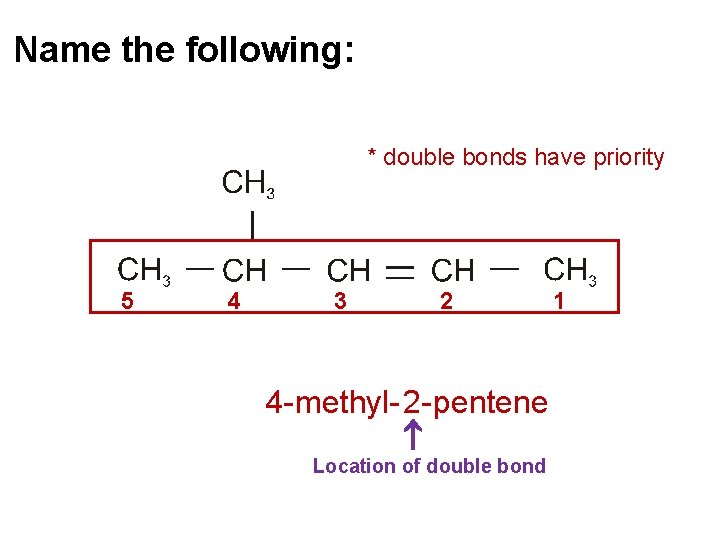
Name the following: * double bonds have priority 5 4 3 2 4 - methyl- 2 - pentene Location of double bond 1

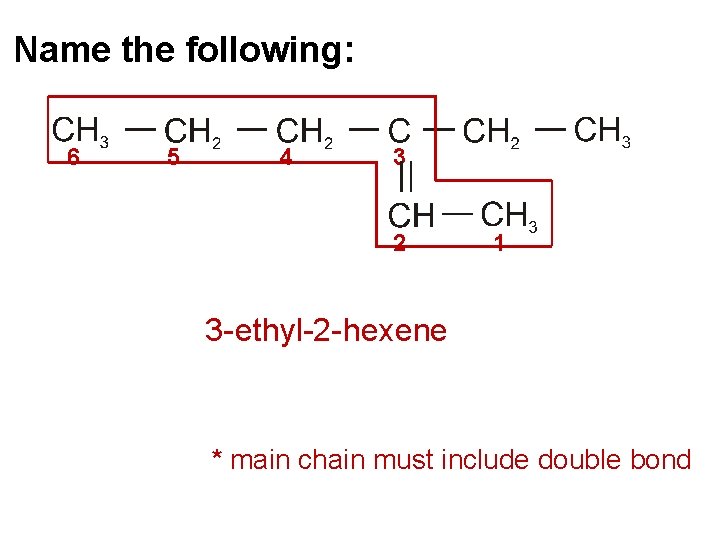
Name the following: 6 5 4 3 2 1 3 - ethyl-2 - hexene * main chain must include double bond

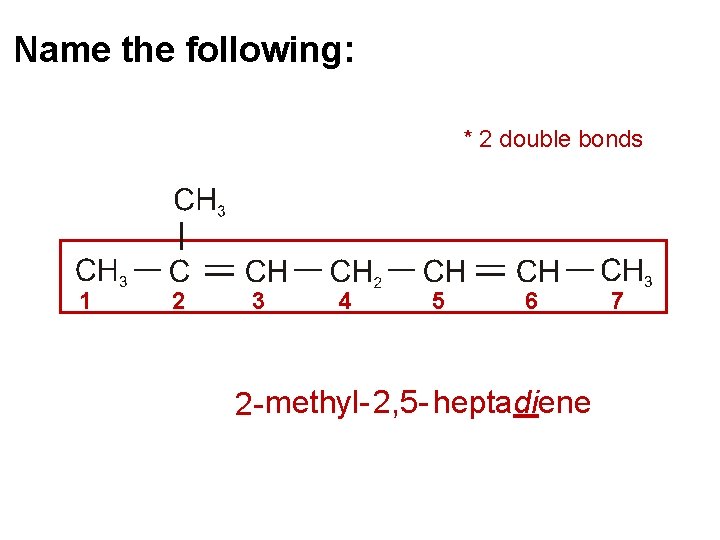
Name the following: * 2 double bonds 1 2 3 4 5 6 2 - methyl- 2, 5 - heptadiene 7

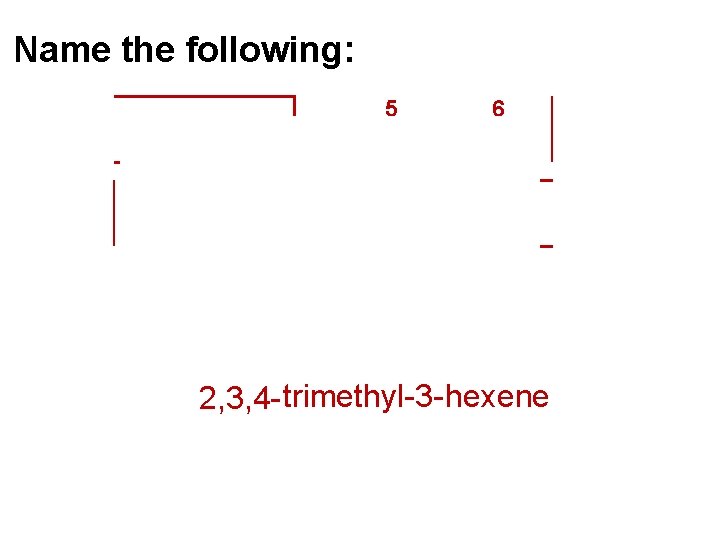
Name the following: 5 1 2 3 6 4 2, 3, 4 - trimethyl-3 - hexene

Name the following: 1 2 3 4 5 6 3 - isopropyl-1, 3 - hexadiene

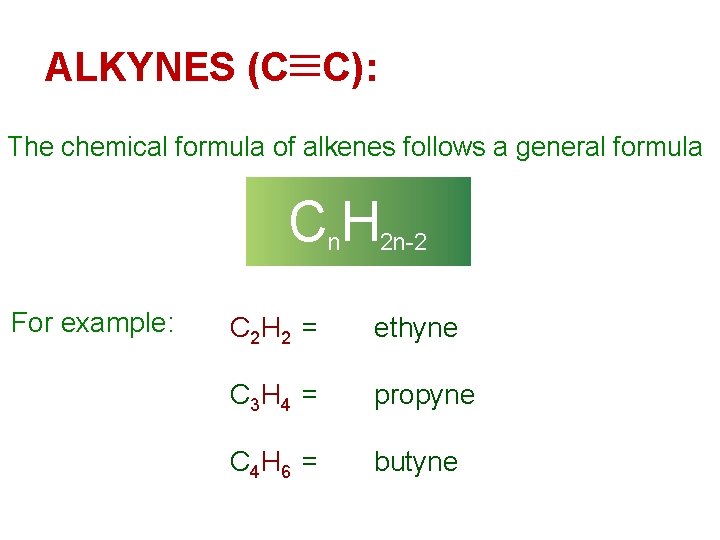
ALKYNES (C C): The chemical formula of alkenes follows a general formula Cn. H 2 n-2 For example: C 2 H 2 = ethyne C 3 H 4 = propyne C 4 H 6 = butyne

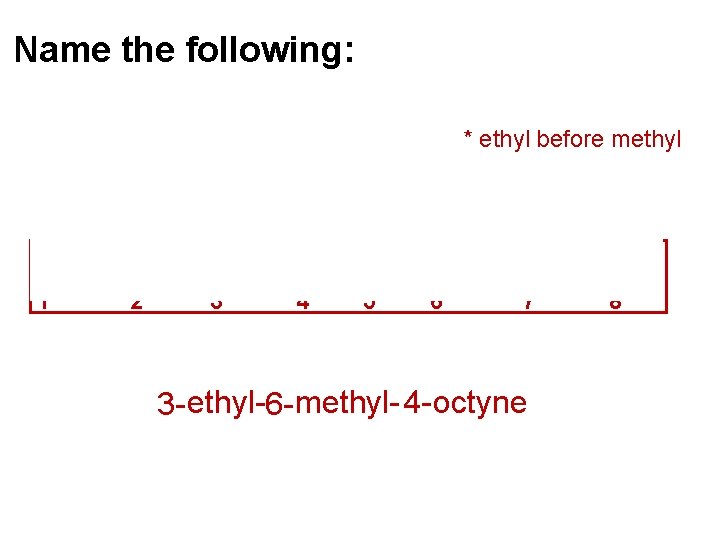
Name the following: * ethyl before methyl 1 2 3 4 5 6 7 3 - ethyl-6 - methyl- 4 - octyne 8

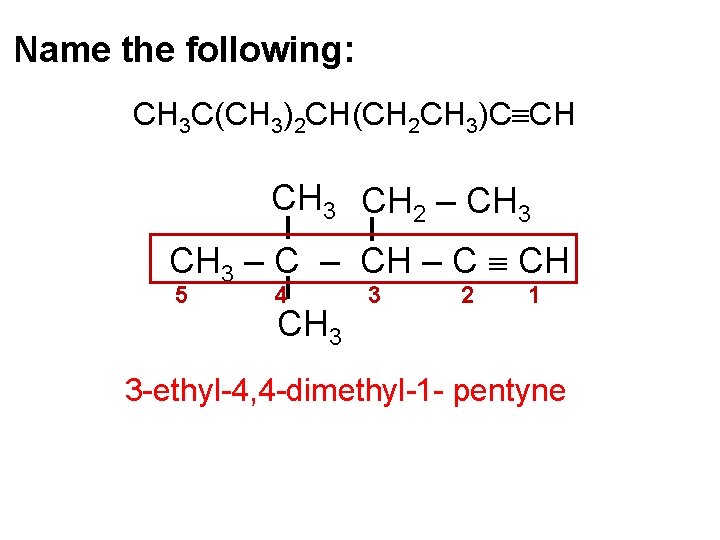
Name the following: CH 3 C(CH 3)2 CH(CH 2 CH 3)C CH CH 3 CH 2 – CH 3 – CH – C CH 5 4 CH 3 3 2 1 3 -ethyl-4, 4 -dimethyl-1 - pentyne

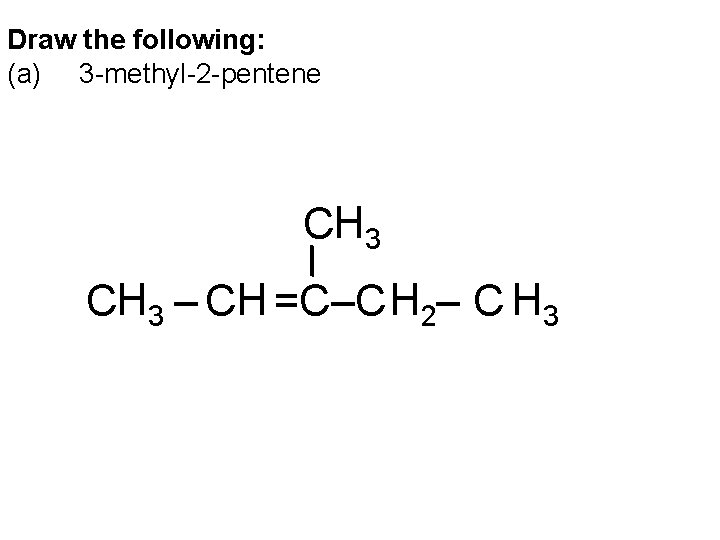
Draw the following: (a) 3 -methyl-2 -pentene CH 3 – CH =C–C H 2– C H 3

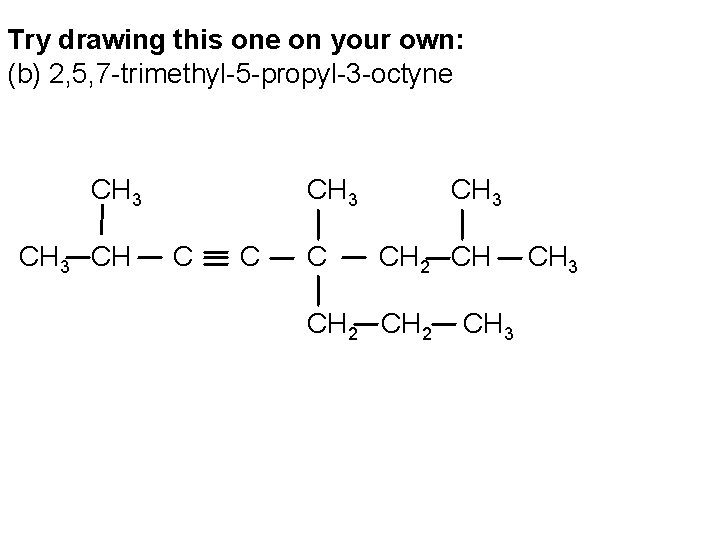
Try drawing this one on your own: (b) 2, 5, 7 -trimethyl-5 -propyl-3 -octyne CH 3 CH CH 3 CH 2 CH 3

Cyclic Hydrocarbons

• Cyclic hydrocarbons are hydrocarbon ring structures • When naming these molecules, the cyclic structure is always the main chain • Number the carbons, in either direction, so that the branches have the lowest possible position number • Double bonds have priority over alkyl groups • have the prefix “cyclo”

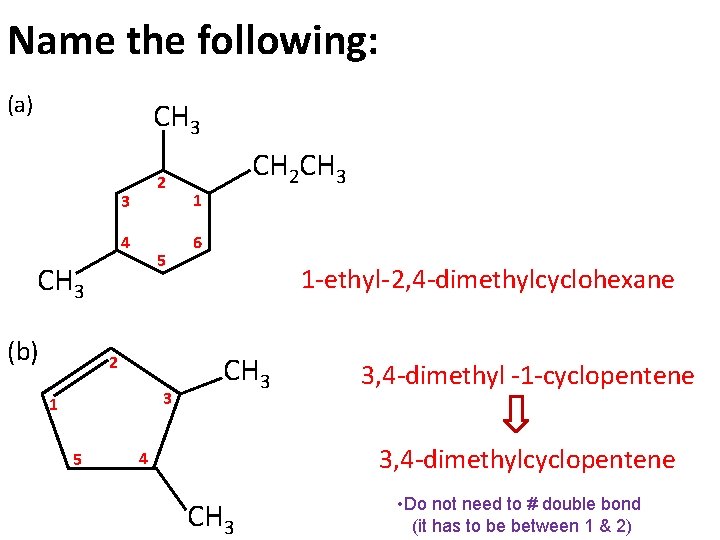
Name the following: (a) CH 3 2 3 4 5 CH 3 (b) 2 3 1 5 CH 2 CH 3 1 6 1 -ethyl- 2, 4 -dimethylcyclohexane CH 3 3, 4 -dimethyl -1 -cyclopentene 3, 4 -dimethylcyclopentene 4 CH 3 • Do not need to # double bond (it has to be between 1 & 2)

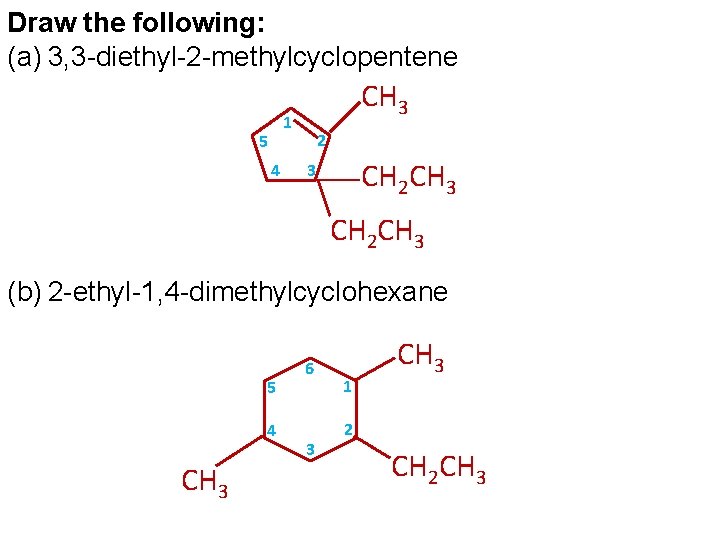
Draw the following: (a) 3, 3 -diethyl-2 -methylcyclopentene CH 3 1 5 4 2 CH 2 CH 3 3 CH 2 CH 3 (b) 2 -ethyl-1, 4 -dimethylcyclohexane 5 4 CH 3 6 3 1 CH 3 2 CH 2 CH 3

HOMEWORK WS “Naming and Drawing Alkenes” WS “Naming and Drawing Alkynes WS “Naming and Drawing Cyclic molecules”