Unit 6 Kinetic Molecular Theory and Gas Laws





























- Slides: 30

Unit 6: Kinetic Molecular Theory and Gas Laws CH 1120

Gases � Vary with respect to chemical properties but share some physical properties ◦ ◦ ◦ Low molar masses Non-metallic Expand spontaneously to fill container Volume of gas = volume of container Compressible Form homogeneous mixtures readily � All physical properties arise due to molecules being relatively far apart

Gases � At ◦ ◦ ◦ ◦ ◦ room temperature: He Ne Ar Kr Xe H 2 N 2 O 2 F 2 Cl 2

Kinetic Molecular Theory (Gases) � An increase in volume at a constant temperature causes pressure to decrease ◦ Syringe filled with air �A temperature increase at constant volume causes pressure to increase ◦ Balloon in winter

Pressure �

Common Units of Pressure � SI unit = Pascal (Pa) � 1 Pa = 1 N/m 2 � 1 bar = 105 Pa � 1 atm = 760 torr = 760 mm. Hg � 1 atm = 101. 3 Pa

Boyle’s Law (V & P) �

Boyle’s Law �

Kelvin � Remember �K how to calculate K = ℃ + 273. 15 � Used in gas law equations

Charles’s Law �

Charles’s Law �

Charles’s and Boyle’s Laws �

Combined Gas Law �

Combined Gas Law � An inflated balloon has a volume of 6. 0 L at sea level (1. 0 atm) and is allowed to ascend until the pressure is 0. 45 atm. During ascent, the temperature of the gas falls from 22℃ to -21℃. Calculate the volume of the balloon at its final altitude.

Dalton’s Law of Partial Pressures �

Dalton’s Law of Partial Pressures � Each gas behaves independently of other gases in a mixture � At constant temperature and constant volume the total pressure of a gas sample is determined by the total number of moles of gas present

Avogadro’s Hypothesis �

Ideal-Gas Equation (Law) �

Ideal-Gas Equation (Law) �

Ideal-Gas Equation (Law) �

What is an Ideal Gas? � Hypothetical gas whose pressure, volume, and temperature relationship are described completely by the ideal gas equation ◦ The molecules do not interact with one another ◦ The volume of the molecules is much smaller than the volume the gas occupies so we assume the molecules take up no space in the container � Very small error is introduced with these assumptions so it is acceptable ◦ When accurate calculations are needed we can correct for the assumptions

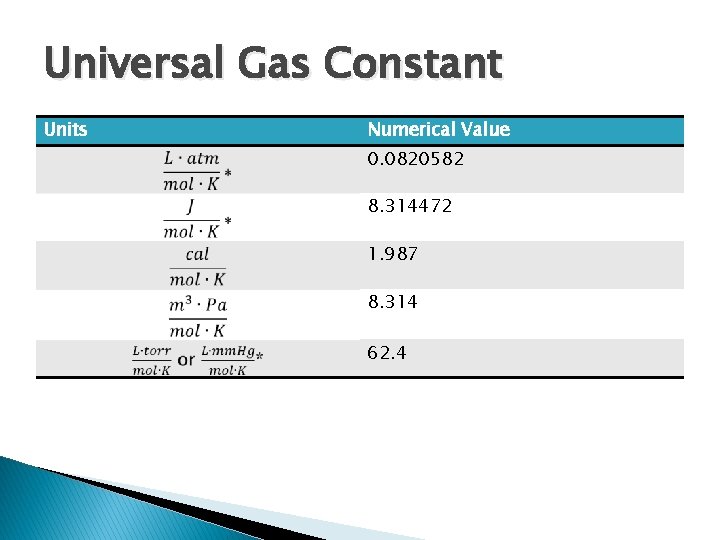
Universal Gas Constant � Unit depend on units of P, V, n, and T �T in Kelvin �n is in mols �P usually atm �V usually L

Universal Gas Constant Units Numerical Value 0. 0820582 8. 314472 1. 987 8. 314 62. 4

Calculations with Gas Laws and Stoichiometry � Calcium carbonate, Ca. CO 3(s) decomposes upon heating to Ca. O(s) and CO 2(g). A sample of Ca. CO 3 is decomposed, and the carbon dioxide is collected in a 250 m. L flask. After decomposition is complete, the gas has a pressure of 1. 3 atm at a temperature of 31℃. How many moles of CO 2 gas were generated? � Use R to determine the units for all of the other components of your equation.

Calculations with Gas Laws and Stoichiometry � When silver oxide is heated, it decomposes according to the reaction below. If 5. 76 g of Ag 2 O is heated and the O 2 gas produced by the reaction is collected in a 0. 65 L flask at 25℃ what is the pressure of the O 2 in the flask? 2 Ag 2 O(s) 4 Ag(s) + O 2(g)

Partial Pressure and Mole Fractions �

Partial Pressure and Mole Fraction �

Standard Temperature and Pressure � 0℃ and 1 atm are referred to as STP ◦ Standard temperature and pressure

Molar Volume �

Molar Volume �A sample of pure helium gas occupies a volume of 6. 8 L at STP, how many grams of helium are present?