Toxic Alcohols John Kashani D O Attending St









































- Slides: 56

Toxic Alcohols John Kashani D. O. Attending, St. Joseph’s Emergency Department Staff Toxicologist, New Jersey Poison Center

Case • An 18 year old male is brought into the ED by his mother when he was difficult to awaken in the AM • He was partying the night before, he is not able to provide a history • He becomes progressively more obtunded while in the ED

Case • A 22 year old frustrated medical student drinks a bottle of formaldehyde he stole from gross anatomy lab • He complains of throat and esophageal irritation and has had multiple episodes of emesis

Case • A 65 year old man is found comatosed • His wife states that he has been depressed recently and has been drinking heavily • An empty bottle of antifreeze was found in his kitchen garbage can

Case • A 17 year old female ingests a bottle of rubbing alcohol • She appears drunk, has multiple episodes of emesis and complains of abdominal pain

Case • A 25 year old man presents to the ED with blurry vision • For the past few days he has been feeling “cruddy” • He admits to the ingestion of homemade everclear 3 days prior

Objectives • Outline the “toxic” alcohols and potentially toxic alcohols • Discuss the pharmacology, kinetics and pathophysiology of the toxic alcohols • Discuss the clinical manifestations, diagnosis and management of patients poisoned by these agents

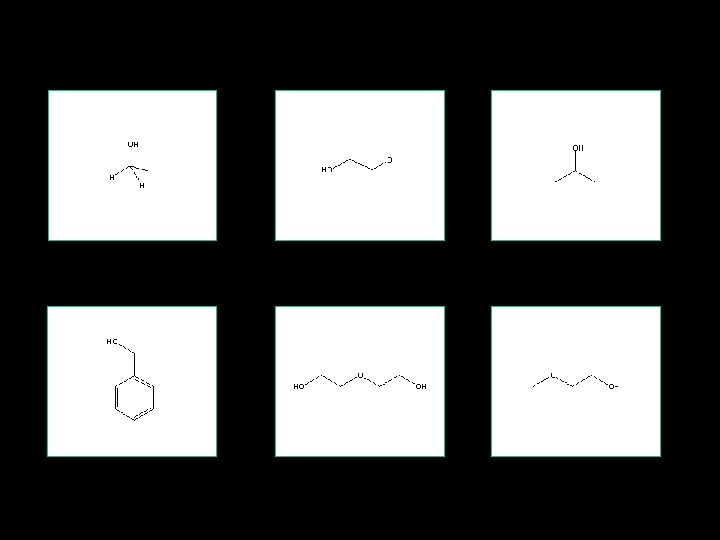
Introduction • Alcohols are hydrocarbons that contain a hydroxyl group • A compound with two hydroxyl groups is called a diol or a glycol • Toxic alcohols commonly refer to methanol, ethylene glycol and isopropyl alcohol

Introduction • Less common but potentially toxic alcohols include diethylene glycol, benzyl alcohol and the glycol ethers


Ethylene Glycol • • Coolant mixtures Antifreeze Air craft de-icing solutions Solvent (inks, pesticides and adhesives) Brake fluid Heat exchangers and condensers Glycerin substitute

Propylene glycol • Commonly used as a diluent for parental preparations • Environmentally safe alternative to ethylene glycol antifreeze

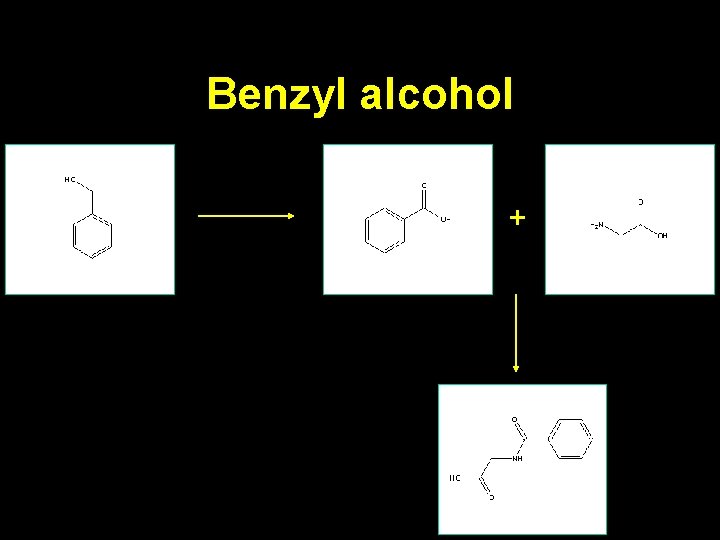
Benzyl alcohol +

Methanol • • Antifreeze (window washer fluid) Anti icing agent Octane booster Ethanol denaturant Extraction agent Solvent Fuel source

Methanol • Varnish and paint removers • Industrial solvent • Manufacture of acetic acid, formaldehyde and inorganic acids

Isopropanol • • • Synthesis of acetone, glycerin Solvent for oils, gums and resins Deicing agent Rubbing alcohol Hair care products, skin lotion and aerosols

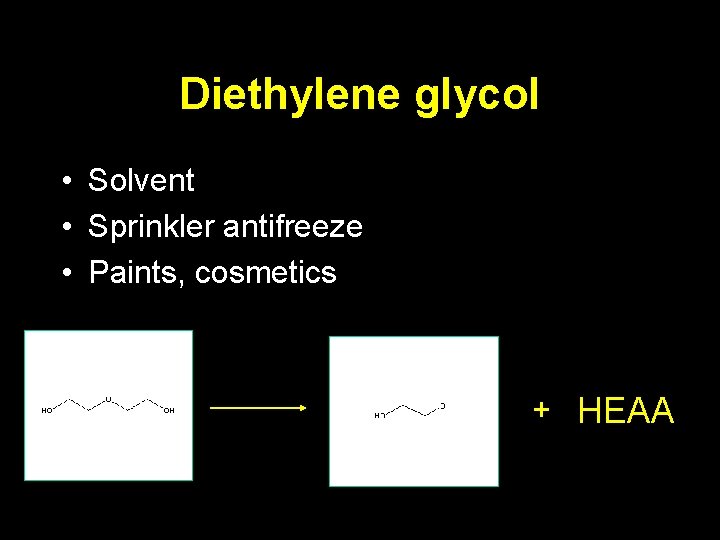
Diethylene glycol • Solvent • Sprinkler antifreeze • Paints, cosmetics + HEAA

Glycol ethers • • • Solvents Semiconductor industry Fingernail polishes and removers Dyes, ink, cleaners, degreasers Brake fluid, car wax, injector cleaner Various household cleaning products

Pharmacology and Kinetics • Exposure may occur dermally, pulmonary and GI – Pulmonary absorption depends on vapor pressure • Rapidly absorbed by the gastrointestinal route

Pharmacology and Kinetics • Time to peak concentration – Ethylene glycol = 1 - 4 hrs – Methanol, isopropyl alcohol = 30 - 60 minutes • VD is 0. 6 L/kg

Pharmacology and Kinetics • Ethylene glycol and methanol are metabolized by alcohol dehyrogenase and aldehyde dehydrogenase • Isopropanol is metabolized by alcohol dehydrogenase • Binding affinities for – ethanol>methanol>ethylene glycol

Pharmacology and Kinetics • Methanol metabolism may be delayed (up to 72 hours) • The volatility of methanol contributes to its pulmonary excretion (10 -20%) • Ethylene glycol is metabolized over 3 – 8 hours – Undergoes multiple oxidations

Pharmacology and Kinetics • Ethylene glycol is not appreciably excreted by the lungs • Isopropanol is rapidly metabolized to acetone via alcohol dehyrogenase • 20% is excreted unchanged • Acetone is predominantly renally excreted

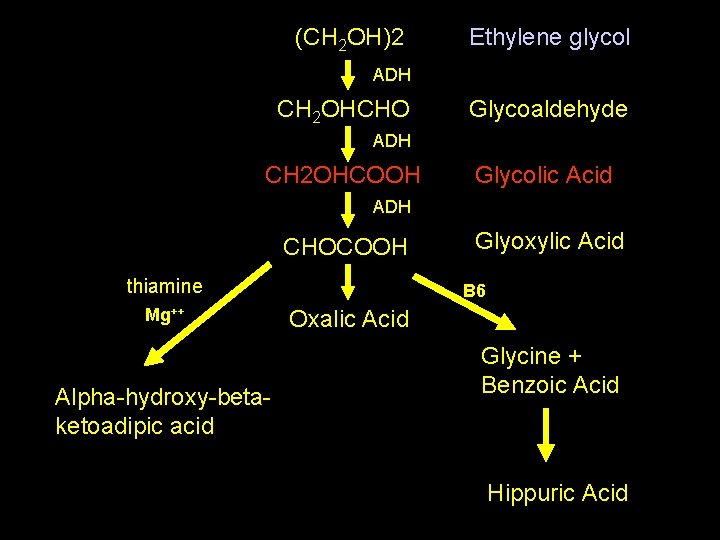
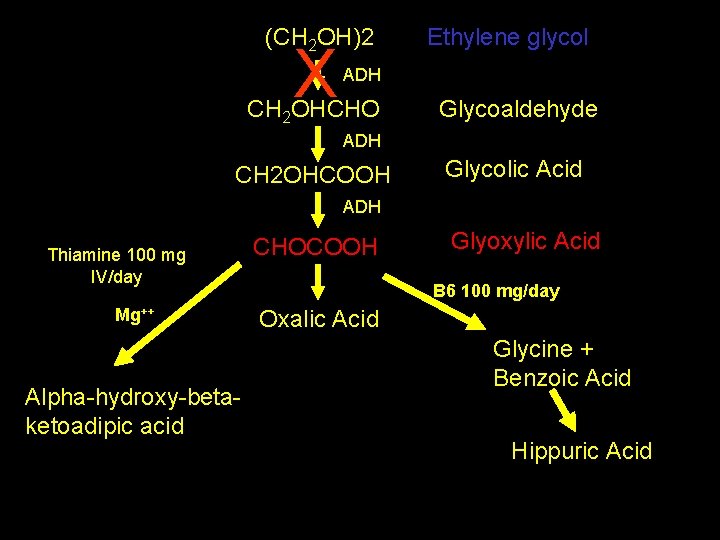
(CH 2 OH)2 Ethylene glycol ADH CH 2 OHCHO Glycoaldehyde ADH CH 2 OHCOOH Glycolic Acid ADH CHOCOOH thiamine Mg++ Alpha-hydroxy-betaketoadipic acid Glyoxylic Acid B 6 Oxalic Acid Glycine + Benzoic Acid Hippuric Acid

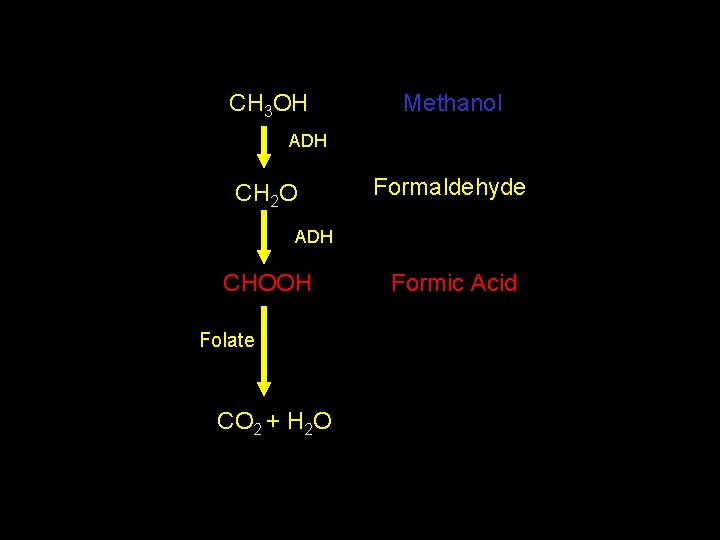
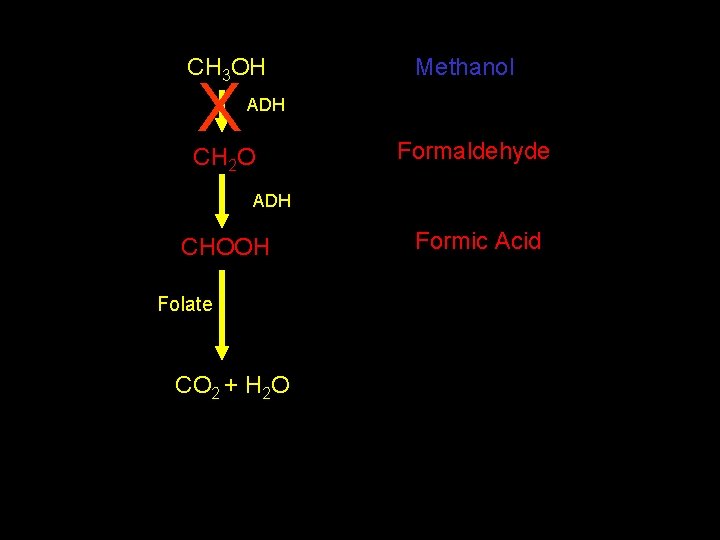
CH 3 OH Methanol ADH CH 2 O Formaldehyde ADH CHOOH Folate CO 2 + H 2 O Formic Acid

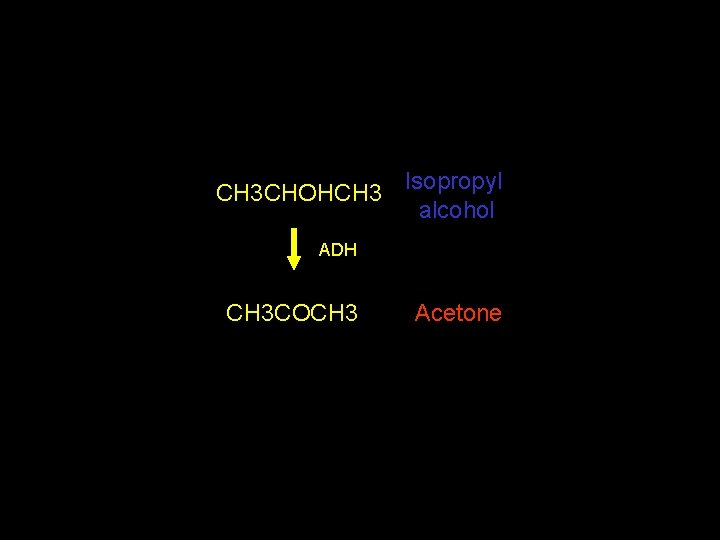
CH 3 CHOHCH 3 Isopropyl alcohol ADH CH 3 COCH 3 Acetone

The Usual Suspects

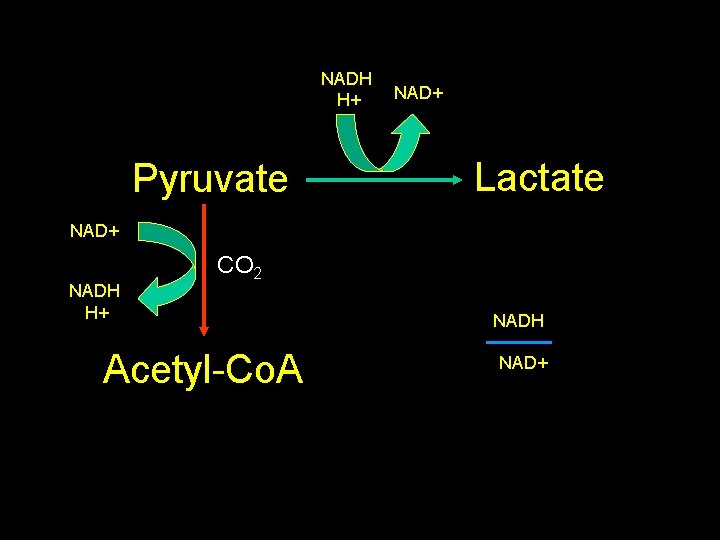
Formic acid • Metabolic acidosis • Inhibits cytochrome oxidase: – Decreased ATP production • Increased anaerobic glycolysis & lactate

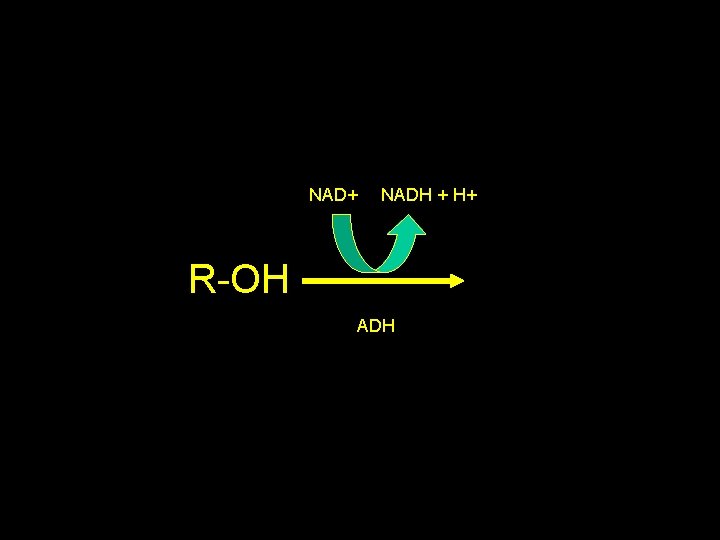
NAD+ NADH + H+ R-OH ADH

NADH H+ Pyruvate NAD+ Lactate NAD+ NADH H+ CO 2 Acetyl-Co. A NADH NAD+

Clinical Manifestations • Clinical manifestations may be related to the parent compound or metabolites • There may be an initial asymptomatic period • Inebriation (unreliable) – Isopropyl>ethylene glycol>methanol

Clinical Manifestations • Vasodilation – hypotension and reflex tachycardia • Hypoglycemia • Anion gap acidosis – Methanol and ethylene glycol • Visual disturbances (”snow Field”) – Formic acid is a retinal toxin

Clinical Manifestations • ATN may develop secondary to calcium oxalate crystalluria • Cranial nerve deficits have been reported with ethylene glycol

Clinical Manifestations • Ispopropanol ingestion usually does not cause major toxicity unless a large amount is ingested – CNS depression, hemorrhagic gastritis and tracheobronchitis

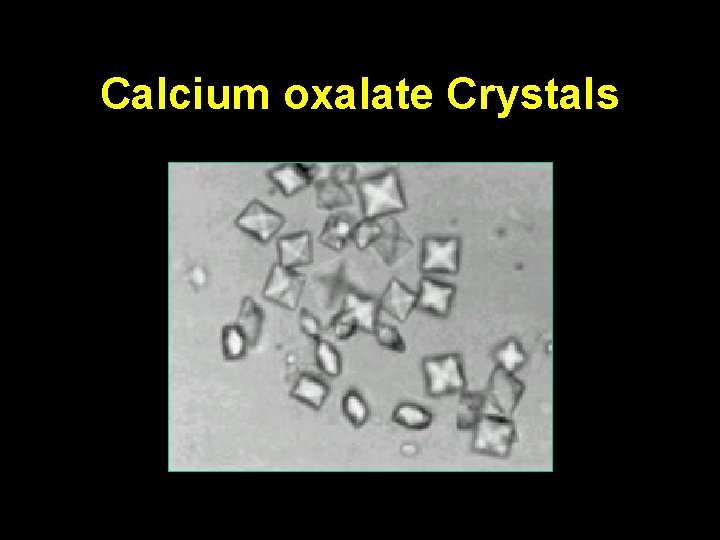
Diagnosis • Both ethylene glycol and methanol result in an anion gap acidosis • Isopropyl alcohol usually does not result in an anion gap acidosis • Hypocalcemia may be seen in ethylene glycol intoxication – Chelation of calcium by oxalate – calcium oxalate crystals

Diagnosis • The absence of crystals is an unreliable finding • The urine of a patient with ethylene glycol ingestion may fluoresce – Short lived, unreliable

Calcium oxalate Crystals

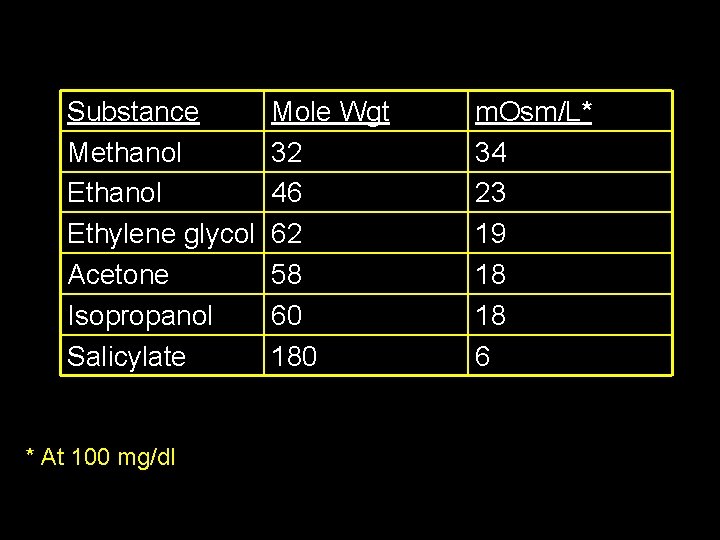
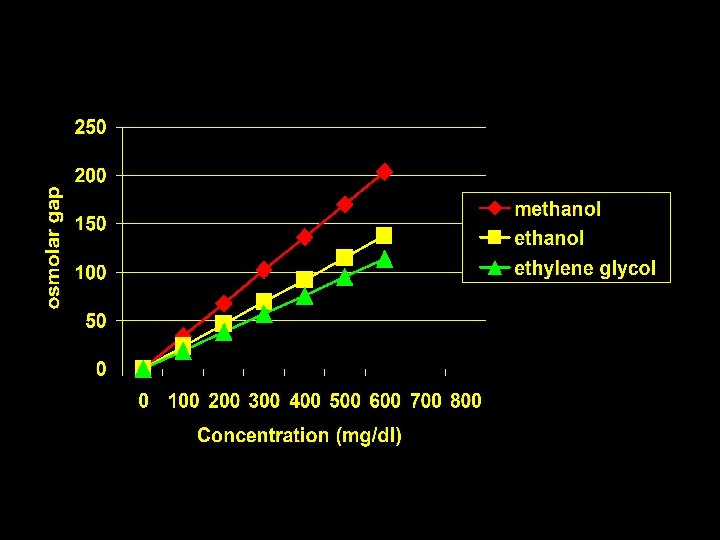
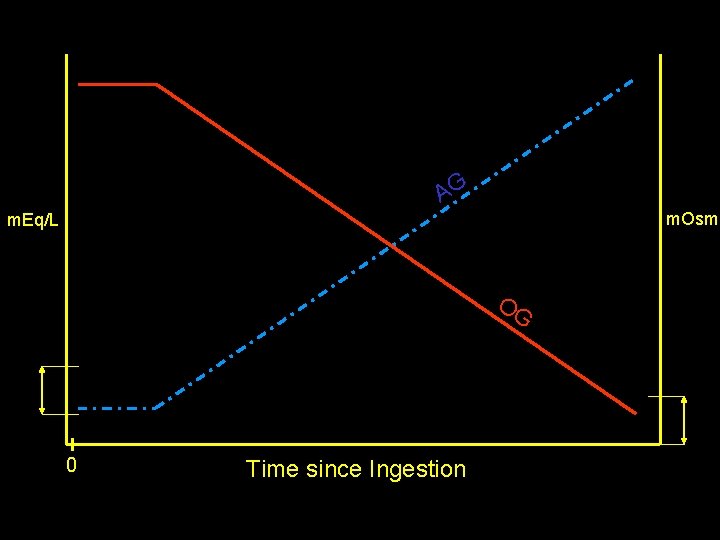
The “Osmolar Gap” Measured Serum Osmolarity Minus Calculated Serum Osmolarity [ 2(NA) + BUN/2. 8 + Glucose/18+Etoh/4. 6]

Substance Methanol Ethylene glycol Acetone Isopropanol Salicylate * At 100 mg/dl Mole Wgt 32 46 62 58 60 180 m. Osm/L* 34 23 19 18 18 6


AG m. Osm m. Eq/L OG 0 Time since Ingestion

Quantitative testing • If quantitative levels are readily available they can be used to determine proper management • Best method is gas chromatography with flame ionization – Subject to false positives

Management • • ABC’s +/---- NGT aspiration AC/ipecac/lavage = Bad move Thiamine and pyridoxine in the setting of ethylene glycol toxicity • Folic acid in the setting of methanol toxicity

Management • Sodium bicarbonate as needed • Inhibition of Alcohol dehydrogenase – Ethanol – Fomepizole

Ethanol vs Fompepizole Ethanol: - Oral or IV - CNS depression - Difficult titration - Frequent levels - Hypoglycemia Fomepizole: - IV - No CNS depression - Easy dosing - No levels to monitor - More predictable pharmacokinetcs - No Hypoglycemia - Cost

Fomepizole…because shit happens

(CH 2 OH)2 X Ethylene glycol ADH CH 2 OHCHO Glycoaldehyde ADH CH 2 OHCOOH Glycolic Acid ADH Thiamine 100 mg IV/day Mg++ Alpha-hydroxy-betaketoadipic acid CHOCOOH Glyoxylic Acid B 6 100 mg/day Oxalic Acid Glycine + Benzoic Acid Hippuric Acid

CH 3 OH X Methanol ADH CH 2 O Formaldehyde ADH CHOOH Folate CO 2 + H 2 O Formic Acid

Case • An 18 year old male is brought into the ED by his mother when he was difficult to wake up in the AM • Apparently he was partying the night before, he is not able to provide a history • He becomes progressively more obtunded while in the ED

Case • A 22 year old frustrated medical student drinks a bottle of formaldehyde he stole from gross anatomy lab • He complains of throat and esophageal irritation and has had multiple episodes of emesis

Case • A 65 year old man is found comatosed • His wife states that he has been depressed recently and has been drinking heavily • An empty bottle of antifreeze was found in his kitchen garbage can

Case • A 17 year old female ingests a bottle of rubbing alcohol • She appears drunk, has multiple episodes of emesis and complain of abdominal pain

Case • A 25 year old man presents to the ED with blurry vision • For the past few days he has been feeling “cruddy” • He admits to the ingestion of homemade everclear 3 days prior

Toxic alcohol Pearls • Calcium oxalate crystals, renal failure = ethylene glycol • “Snow field vision” = methanol • Methanol has a slower metabolism and there may be a significant lag until the onset of symptoms • A “normal” osmolar gap does not rule out the diagnosis

Toxic alcohol Pearls • “ketosis without acidosis” = isopropyl alcohol • Inhibition of alcohol dehydrogenase with fomepizole

The End