Alcohols Alcohols l l l Methanol CH 3











- Slides: 20

Alcohols

Alcohols l l l Methanol CH 3 OH Ethanol C 2 H 5 OH Propan-1 -ol C 3 H 7 OH Propan-2 -ol C 3 H 7 OH Butan-1 -ol C 4 H 9 OH Butan-2 -ol C 4 H 9 OH

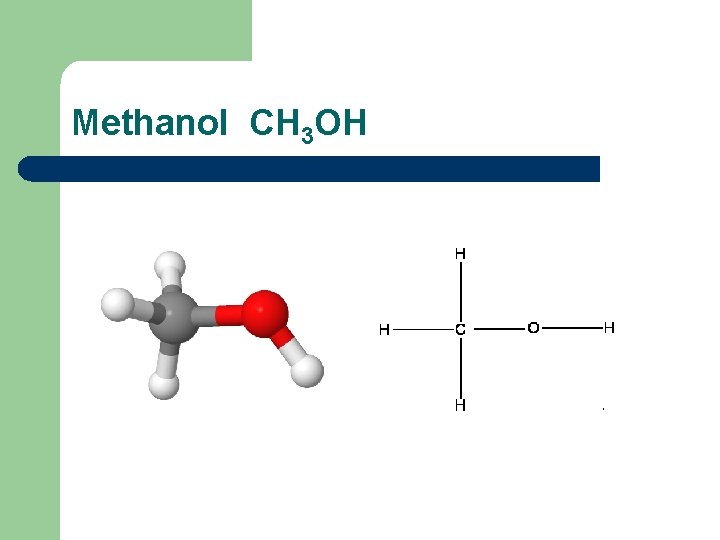
Methanol CH 3 OH

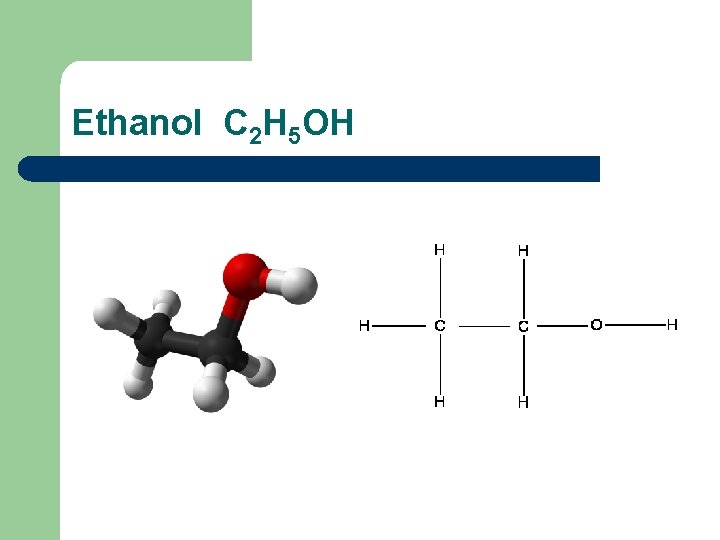
Ethanol C 2 H 5 OH

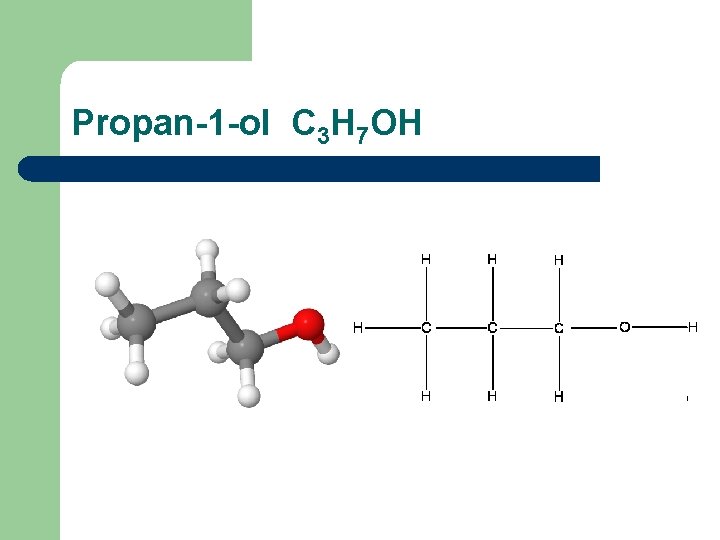
Propan-1 -ol C 3 H 7 OH

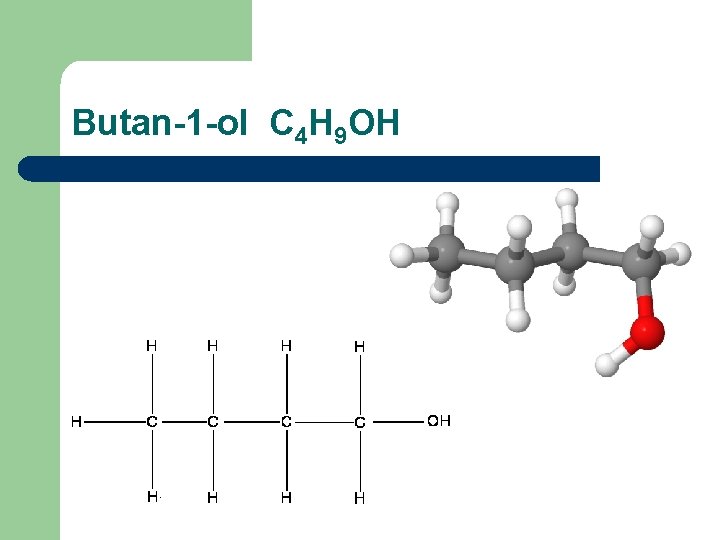
Butan-1 -ol C 4 H 9 OH

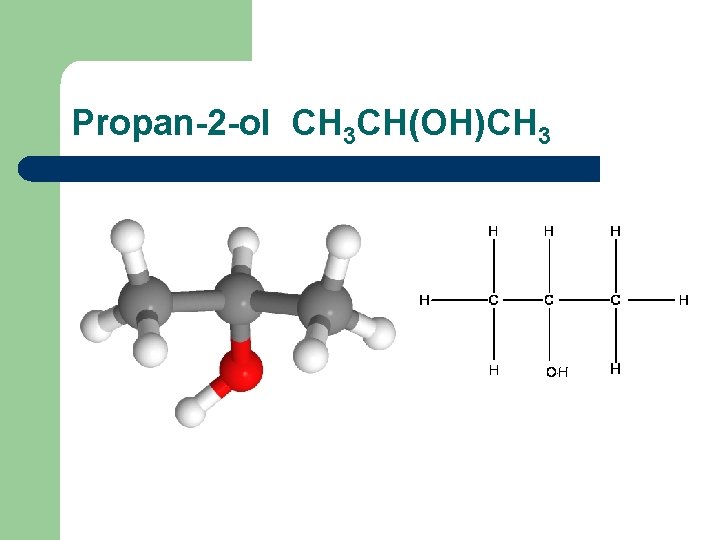
Propan-2 -ol CH 3 CH(OH)CH 3

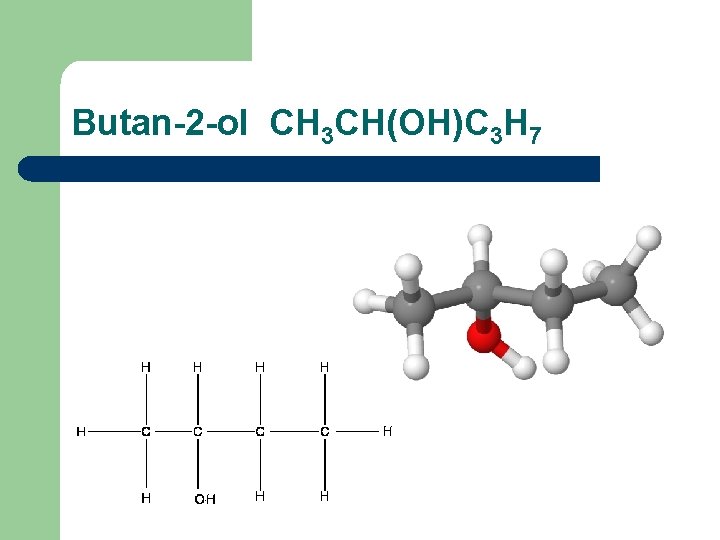
Butan-2 -ol CH 3 CH(OH)C 3 H 7

Classification of alcohols 1. Primary alcohol: contains one carbon atom directly attached to the carbon that contains the hydroxyl group, e. g. propan -1 -ol

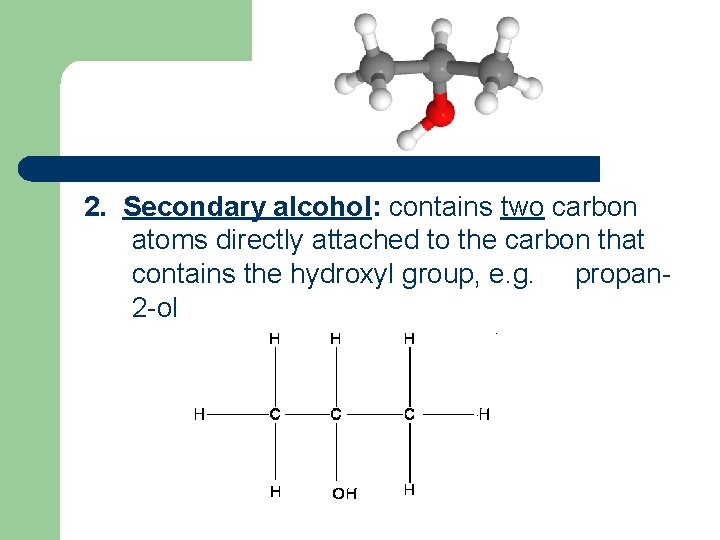
2. Secondary alcohol: contains two carbon atoms directly attached to the carbon that contains the hydroxyl group, e. g. propan 2 -ol

Physical properties l l Physical state: Liquid Boiling points much higher than the corresponding alkanes, due to polar OH group

Physical properties Solubility of methanol in l (i) cyclohexane – not very soluble methanol is polar cyclohexane is not l (ii) water - completely soluble because it is polar. l As alcohol molecule gets bigger the polar part becomes less significant so the alcohol becomes less soluble in water and more soluble in cyclohexane

Butan-1 -ol is – (i) soluble in cyclohexane – (ii) not very soluble in water l The polar OH group is becoming less significant as the molecule gets bigger

Comparison with water l l Both have polar OH groups Alcohols have a non-polar part Both form hydrogen bonds between their molecules Water is more polar and has a greater capacity to form hydrogen bonds and so has a higher boiling point than methanol or ethanol

Methanol l Methanol: is toxic (can cause blindness, insanity and death) l It is added to industrial alcohol to prevent people drinking it. This mixture is called methylated spirits. l The methanol acts as a denaturing agent – it renders a substance unfit for purpose without destroying the usefulness or applications of the substance. A purple dye is often added as a warning.

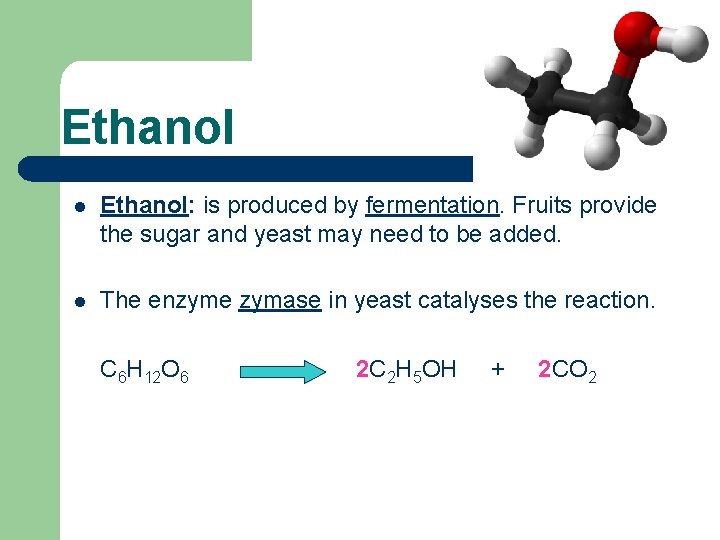
Ethanol l Ethanol: is produced by fermentation. Fruits provide the sugar and yeast may need to be added. l The enzyme zymase in yeast catalyses the reaction. C 6 H 12 O 6 2 C 2 H 5 OH + 2 CO 2

Alcoholic Drinks Ingredient Drink Grapes Wine % (v/v) alcohol 12 Apples Cider 4. 5 Malted grain Beer 5

Ethanol l To produce drinks of higher alcohol concentration the fermented liquids must be distilled. l Spirits (whiskey, brandy, gin, vodka) contain 40% alcohol.

Gasohol l Ethanol obtained from sugar cane is used for making gasohol in Brazil. This is then used as a fuel.

Uses of ethanol 1. Alcoholic drinks 2. Fuel 3. Solvent (can dissolve both polar and nonpolar solutes)