Stoichiometry Moles Unit of measurement that specifies the


















- Slides: 21

Stoichiometry

Moles • Unit of measurement that specifies the amount of particles being used • 1 mole (mol) is known as 6. 023 x 1023 representative particles of the substance being measured • This number, 6. 023 x 1023 is known as Avogadro’s number

Representative Particle • The term representative particle refers to the species present in a substance, usually • atoms • molecules • formula units The representative particle of most elements is the atom.

Representative Particle (cont. ) • The representative particle is the molecule for • diatomic molecules (H 2, N 2, O 2, F 2, Cl 2, Br 2, and I 2). • The molecular compounds water (H 2 O) and sulfur dioxide (SO 2) are composed of H 2 O and SO 2 molecules, respectively. • For ionic compounds, such as calcium chloride, the representative particle is the formula unit Ca. Cl 2.

Converting to and from Moles • The mole allows chemists to count the number of representative particles in a substance. • A mole of any substance contains Avogadro’s number of representative particles, or 6. 023 × 1023 representative particles.

Converting to and from Moles 1 mol 6. 023 × 1023 representative particles 1 mol

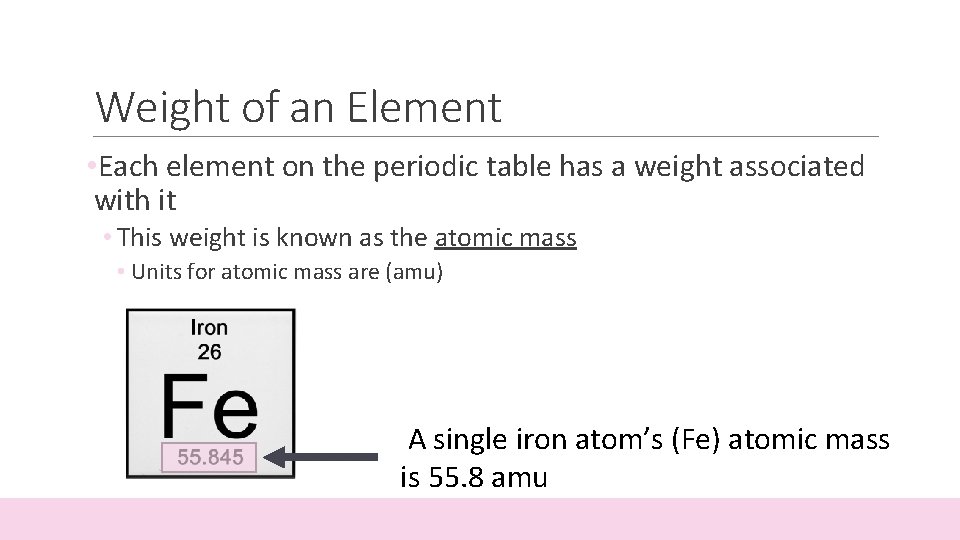
Weight of an Element • Each element on the periodic table has a weight associated with it • This weight is known as the atomic mass • Units for atomic mass are (amu) A single iron atom’s (Fe) atomic mass is 55. 8 amu

Molar Mass

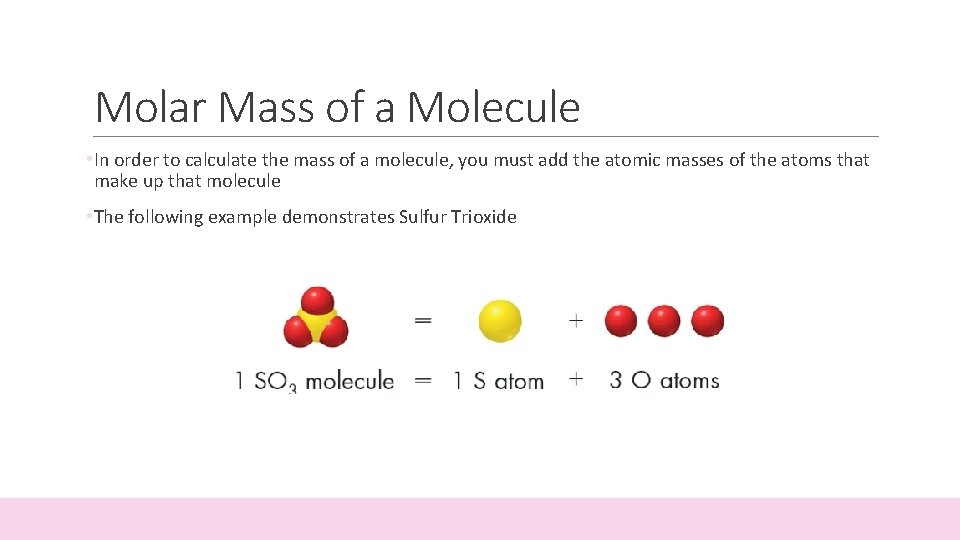
Molar Mass of a Molecule • In order to calculate the mass of a molecule, you must add the atomic masses of the atoms that make up that molecule • The following example demonstrates Sulfur Trioxide

Molar Mass of a Molecule (cont. )

Converting From Grams to Moles

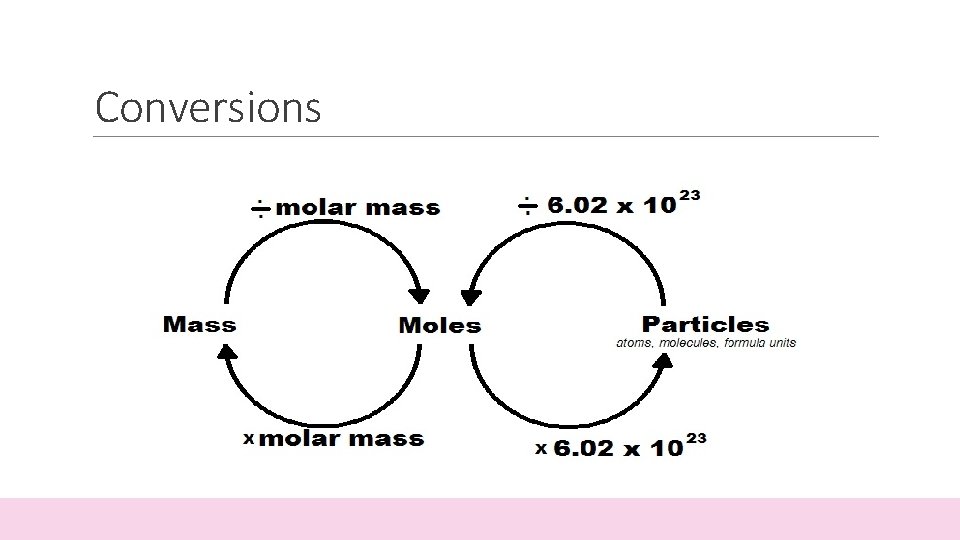
Conversions

Predicting Amount of Products Formed • The coefficients in a balanced equation give the relative amounts (in moles) of reactants and products • Mole Ratio: the ratio between the amounts (in moles) between one starting material and one product 2 H 2 O 2 H 2 + 1 O 2 • For every 2 moles of water (H 2 O) put into the reaction, you get 2 moles of hydrogen (H 2) and 1 mole of oxygen (O 2)

Predicting Amount of Products Formed (cont. )

Predicting Amount of Products Formed (cont. )

Predicting Amount of Products Formed (cont. )

Limiting Reagents • A limiting reagent (or limiting reactant), is the reactant that is first used up in a chemical reaction • Once this reactant is used up, the reaction stops • This reactant limits the amount of product that can form • The excess reagent (or excess reactant), is the unused reactant that is left over once the limiting reagent is used Passage I stole from the AP Chem book:

Limiting Reagents (cont. ) Assume you are given the following reaction: H 2 + Cl 2 2 HCl When given 4. 0 mol of hydrogen, and 2. 0 mol of chlorine, how many moles of hydrochloric acid (HCl) can form? • In order to solve this problem: • You must calculate the amount of product formed from each reactant, H 2 and Cl 2 • The limiting reagent will be the reactant (H 2 or Cl 2) that produces the smaller amount of product

Limiting Reagents (cont. ) Larger number (not possible) Smaller number (Maximum amount)

Reaction Yield

Reaction Yield (cont. )