Moles MOLES Moles Atoms Moles Grams Copyright Sautter














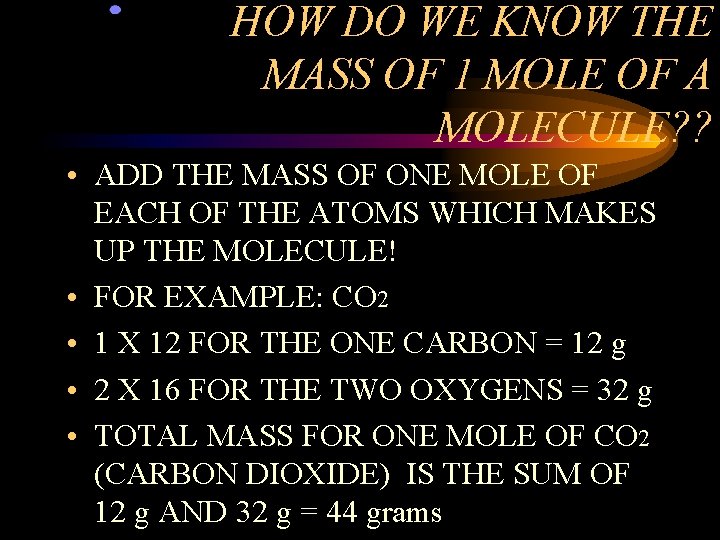




- Slides: 22

Moles MOLES Moles Atoms Moles Grams Copyright Sautter 2003

What is a mole? • A mole is a name for a number just like the word dozen is a name for a number. • As you know, a dozen means the number 12. • A mole is a number too but a much, much bigger number. • A mole is 602, 000, 000, 000. In words it is six hundred and two hexillion ! • Why is a mole so big? Because it is used to count atoms and molecules which are very, very small, It takes lots of them just to be seen much less work with them. A speck of dusk contains trillions of atoms or more!



Counting Atoms and Molecules • If atoms and molecules are so small and numerous, how can they be counted? ? • If atoms and molecules could be seen and all the people on the earth began counting the water molecules in our five teaspoons of water and if everyone counted day and night for his entire life, and if we added all their results together we would still not count them all !! • Counting atoms and molecules is difficult but we have an easy way to count them.

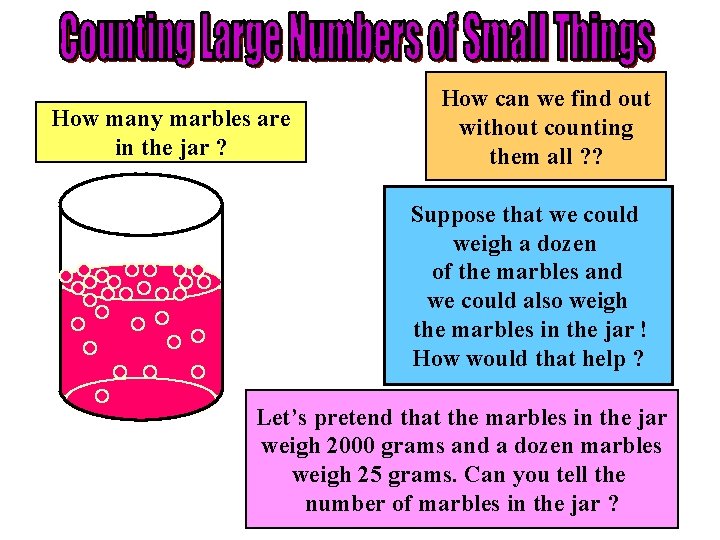
How many marbles are in the jar ? How can we find out without counting them all ? ? Suppose that we could weigh a dozen of the marbles and we could also weigh the marbles in the jar ! How would that help ? Let’s pretend that the marbles in the jar weigh 2000 grams and a dozen marbles weigh 25 grams. Can you tell the number of marbles in the jar ?

COUNTING LARGE NUMBERS OF SMALL THINGS • In our marble example, the marbles weighed 2000 grams and one dozen of the marbles weighed 25 grams. • Dividing 2000 grams (all the marbles) by 25 grams (the weight of one dozen) gives 80 dozen marbles. • Multiplying 80 times 12 we get 960 marbles in the jar without actually counting all of the marble ! • In the case of atoms or molecules the same idea can be used. If we know the weight of a number of a certain type of atom or molecule and the weight of the sample that we have we can then determine the number of atoms or molecules in the sample by division and multiplication.

COUNTING LARGE NUMBERS OF SMALL THINGS • In counting atoms and molecules we will not use a dozen as our counted quantity. A dozen atoms would be so small that they could never even be seen with traditional microscopes. • Our counted quantity will be one mole (6. 02 x 1023 *) atoms or molecules. Unlike our marble example we will not count out and weigh the reference sample. Luckily, this work has been done for us. • The Periodic Table of the Elements lists the weight (mass) of one mole of every element. We can find the weight of one mole of each element in grams. • * 6. 02 x 1023 = 602, 000, 000, 000 = 1 mole

MOLES • MOLES GRAMS • MULTIPLE BY GRAM MOLECULAR / ATOMIC WEIGHT* • GRAMS MOLES • DIVIDE BY GRAM MOLECULAR / ATOMIC WEIGHT • * weight of one mole from the periodic table How are the number of moles determined from the number of grams ?

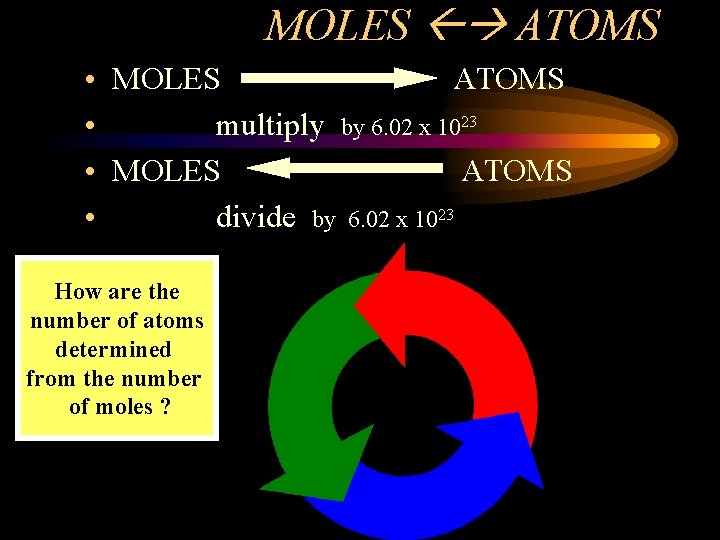
MOLES ATOMS • MOLES ATOMS • multiply by 6. 02 x 1023 • MOLES ATOMS • divide by 6. 02 x 1023 How are the number of atoms determined from the number of moles ?

FINDING GRAM ATOMIC WEIGHTS (WT OF 1 MOLE) Oxygen • FIND THE ATOMIC MASS OF THE ELEMENT ON THE PERIODIC TABLE. • IT IS THE LARGER OF THE TWO NUMBERS SHOWN IN THE BLOCK! O Atomic number 8 Atomic mass 16. 0 Mass of one mole 16. 0 grams 6. 02 x 1023 atoms of O

Let’s try some problems with moles ! How many moles are contained in 36. 0 grams of oxygen? Solution: From the Periodic Table 36. 0 grams / 16. 0 grams per mole = 2. 25 moles 36. 0 grams contains 2. 25 moles of Oxygen Atoms

How many atoms are contained in 36. 0 grams of oxygen? Solution: 36. 0 grams / 16. 0 grams per mole = 2. 25 moles of O 2. 25 mole x (6. 02 x 1023 atoms per mole) = 1. 35 x 1023 atoms of oxygen

How many moles are contained in 3. 6 x 1024 atoms? • Solution: • (3. 6 x 1024 atoms) / (6. 02 x 1023) = 6. 0 moles • OF ANY KIND OF ATOMS !!

How many grams does 3. 6 x 1024 atoms of S “weigh” (mass)? • SOLUTION: • From the Periodic Table, Gram Atomic Mass of S is 32. 1 grams • (3. 6 x 1024) / (6. 02 x 1023) = 6. 0 moles of S • 6. 0 x 32. 1 = 192. 6 grams of Sulfur is the “weight” (mass) of 3. 6 x 1024 atoms of S!

AVOGADRO’S NUMBER! WHAT IS IT? ? • ONE MOLE IS 6. 02 x 1023 of ANYTHING just as ONE DOZEN 12 of ANYTHING! • This NUMBER (6. 02 x 1023) is called Avogadro’s Number!!

WHAT ABOUT MOLECULES? ? • A MOLECULE IS A GROUP OF CHEMICALLY BONDED (LINKED) ATOMS. • FOR EXAMPLE: CARBON DIOXIDE • FORMULA: CO 2 • IT CONSISTS OF 1 ATOM OF CARBON C AND TWO ATOMS OF OXYGEN O
HOW DO WE KNOW THE MASS OF 1 MOLE OF A MOLECULE? ? • ADD THE MASS OF ONE MOLE OF EACH OF THE ATOMS WHICH MAKES UP THE MOLECULE! • FOR EXAMPLE: CO 2 • 1 X 12 FOR THE ONE CARBON = 12 g • 2 X 16 FOR THE TWO OXYGENS = 32 g • TOTAL MASS FOR ONE MOLE OF CO 2 (CARBON DIOXIDE) IS THE SUM OF 12 g AND 32 g = 44 grams

HOW DO WE FIND THE NUMBER OF MOLES, MASSES AND NUMBER OF MOLECULES THEN? ? • USE THE SAME STEPS AS WHEN DEALING WITH ATOMS EXCEPT WE NOW USE THE GRAM MOLECULAR WEIGHT (MASS) AS WE SHOWED IN THE PREVIOUS SLIDE!

FINDING THE NUMBER OF MOLES OF MOLECULES • HOW MANY MOLES OF CARBON DIOXIDE ARE CONTAINED 88. 0 GRAMS OF CO 2 ? • AS WE SAW EARLIER, 1 MOLE OF CO 2 HAS A MASS OF 44. 0 GRAMS. • 88. 0 grams / 44. 0 grams per mole = 2. 0 moles of CO 2 • HOW MANY MOLECULES ARE PRESENT? • 1 Mole = 6. 02 x 1023 molecules • 2. 0 mole x (6. 02 x 1023) = 12. 04 x 1023 CO 2 molecules • HOW MANY ATOMS ARE PRESENT? • Each CO 2 contains 1 carbon and 2 oxygen atoms, a total of 3 atoms in each molecule. • 12. 04 x 1023 molecules x 3 atoms per molecule = 36. 1 x 10 24 atoms

NOW IT’S YOUR TURN!! • TRY SOME PROBLEMS USING THE STEPS SHOWN HERE. • THE MORE YOU DO THE EASIER THEY GET!!!!
