Gas Stoichiometry Gas Stoichiometry Moles Liters of a








- Slides: 11

Gas Stoichiometry

Gas Stoichiometry Moles Liters of a Gas: – STP - use 22. 4 L/mol – Non-STP - use ideal gas law

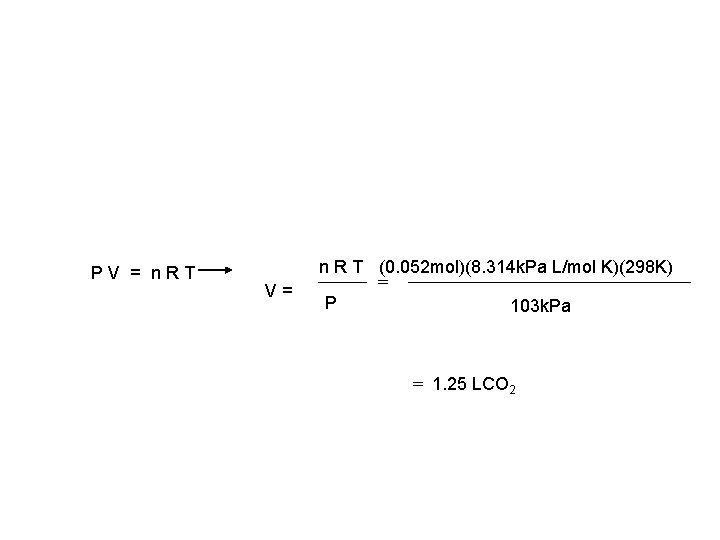
Gas Stoichiometry Problem What volume of CO 2 forms from 5. 25 g of Ca. CO 3 at 103 k. Pa & 25ºC? Ca. CO 3 5. 25 g ooking for liters: Start with stoich and calculate moles of CO 2. 5. 25 g 1 mol Ca. CO 3 Ca. O 1 mol CO 2 100. 09 g 1 mol Ca. CO 3 + CO 2 ? L non-STP 0. 052 mols CO 2 Plug this into the Ideal Gas Law to find liters.

PV = n. RT V= n R T (0. 052 mol)(8. 314 k. Pa L/mol K)(298 K) = P 103 k. Pa = 1. 25 LCO 2

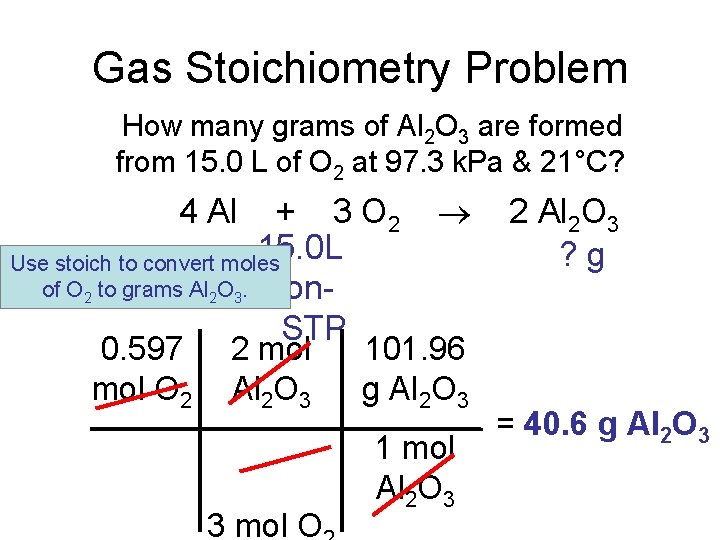
Gas Stoichiometry Problem How many grams of Al 2 O 3 are formed from 15. 0 L O 2 at 97. 3 k. Pa & 21°C? 4 Al + 3 O 2 15. 0 L non. STP of 2 Al 2 O 3 ? g GIVEN: WORK: P = 97. 3 k. Pa V = 15. 0 L n=? T = 21°C = 294 K R = 8. 315 L k. Pa/mol K PV = n. RT (97. 3 k. Pa) (15. 0 L) = n (8. 315 L k. Pa/mol K) NEXT (294 K) Given liters: Start with Ideal Gas Law and calculate moles of O 2. n = 0. 597 mol O

Gas Stoichiometry Problem How many grams of Al 2 O 3 are formed from 15. 0 L of O 2 at 97. 3 k. Pa & 21°C? + 3 O 2 15. 0 L Use stoich to convert moles of O 2 to grams Al 2 O 3. non. STP 0. 597 2 mol 101. 96 mol O 2 Al 2 O 3 g Al 2 O 3 4 Al 3 mol O 1 mol Al 2 O 3 2 Al 2 O 3 ? g = 40. 6 g Al 2 O 3

Gas Stoichiometry Find vol. hydrogen gas made when 38. 2 g zinc react w/excess hydrochloric acid. @ STP. Zn (s) + 2 HCl (aq) 38. 2 g excess Zn. Cl 2(aq) + H 2(g) XL

What mass solid magnesium is required to react w/250 m. L carbon dioxide at 1. 5 atm and 77 o. C to produce solid magnesium oxide and solid carbon? 2 Mg (s) + CO 2 (g) 250 m. L 0. 25 L X g Mg 0. 25 L V = 250 m. L T = 77 o. C + 273 = K P = 1. 5 atm PV = n. RT 2 Mg. O (s) + C (s) PV n= RT 350 K 151. 95 k. Pa atm (0. 250 L) 151. 951. 5 k. Pa = 0. 013 mol CO 2 n=. atm/ /mol 0. 0821 mol. K. K (350 K) 8. 314 LL. k. Pa x g Mg = 0. 013 mol CO 2 Mg 2 mol Mg 1 mol CO 2 24. 3 g Mg = 0. 63 g Mg 1 mol Mg

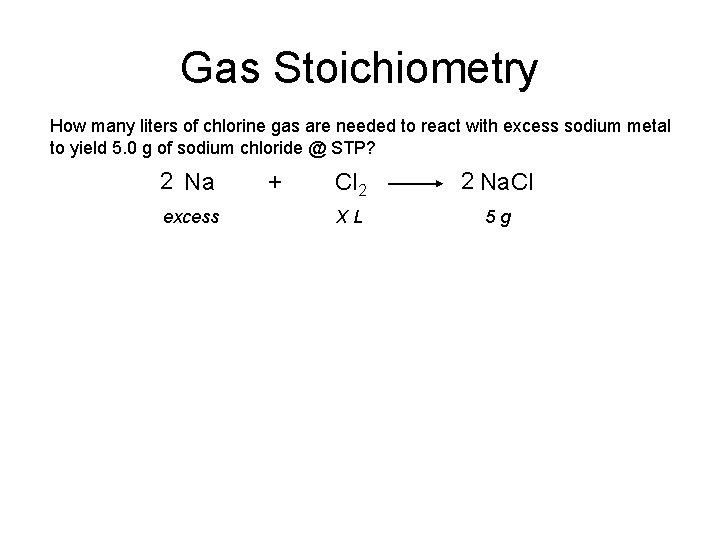
Gas Stoichiometry How many liters of chlorine gas are needed to react with excess sodium metal to yield 5. 0 g of sodium chloride @ STP? 2 Na excess + Cl 2 2 Na. Cl XL 5 g

Sample problem 2 Ammonia (NH 3) gas can be synthesized from nitrogen gas + hydrogen gas. What volume of ammonia at 450 k. Pa and 80°C can be obtained from the complete reaction of 7. 5 kg hydrogen? First we need a balanced equation: N 2(g) + 3 H 2(g) 2 NH 3(g) 1 mol H 2 2 mol NH 3= 2475 mol # mol NH 7500 = g H 3 x 2. 02 g H 2 3 mol H 2 PV = n. RT P = 450 k. Pa, n = 2475 mol, T = 353 K (2475 mol)(8. 31)(353 K) = V = 16 135 L (450 KPa) NH 3

Sample problem 3 Hydrogen gas (and Na. OH) is produced when sodium metal is added to water. What mass of Na is needed to produce 20. 0 L of H 2 at STP? First we need a balanced equation: 2 Na(s) + 2 H 2 O(l) H 2(g) + 2 Na. OH(aq) PV = n. RT P= 1 atm, V= 20. 0 L, T= 273 K (1 atm)(20. 0 L) = n = 0. 893 mol H 2 (0. 0821 atm • L/K • mol)(273 K) or # mol = 20. 0 L x 1 mol / 22. 4 L = 0. 893 mol 2 mol Na 22. 99 g Na # g Na= 0. 893 mol Hx x 2 = 41. 1 g Na 1 mol H 2 1 mol Na