Source waters flowpaths and solute flux in mountain



























- Slides: 68

Source waters, flowpaths, and solute flux in mountain catchments Mark Williams

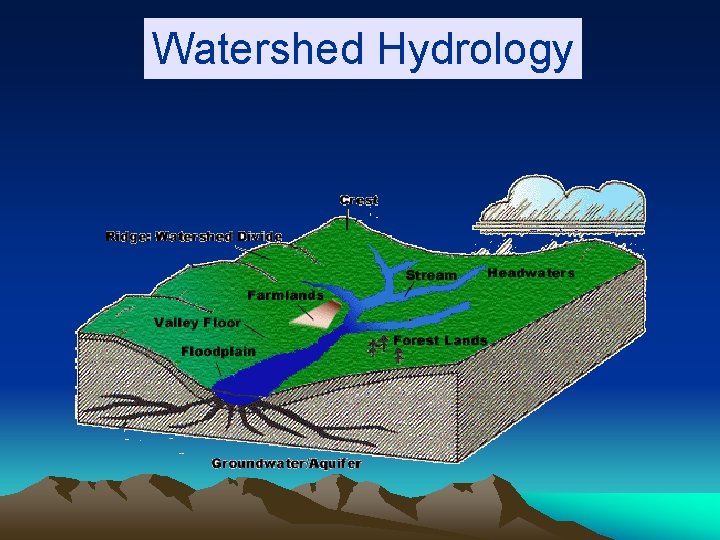
Watershed Hydrology

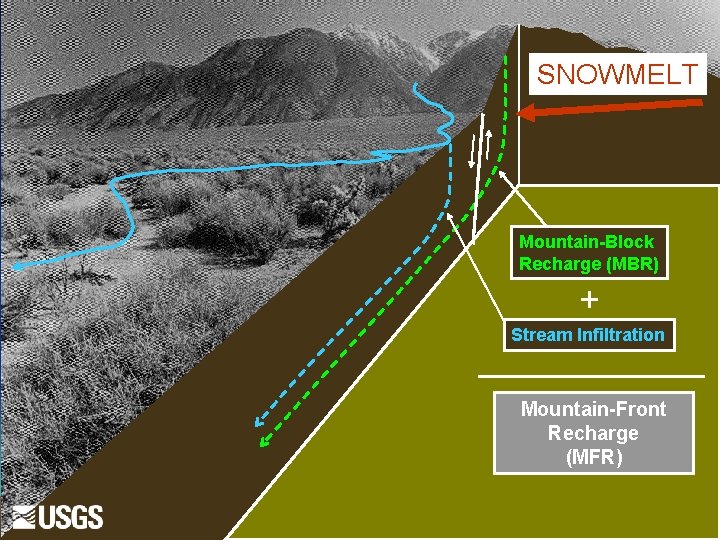
SNOWMELT Mountain-Block Recharge (MBR) + Stream Infiltration Mountain-Front Recharge (MFR)

HYDROLOGIC UNKNOWNS ¡ Flowpaths ¡ Residence time ¡ Circulation depth ¡ Reservoir sizes ¡ Groundwater fluxes Add snowmelt runoff Permafrost ¡ Fractured rock environment increases difficulty of understanding mountain plumbing

APPLICATION IN GREEN LAKES VALLEY: LTER RESEARCH SITE Green Lake 4 Sample Collection • Stream water weekly grab samples • Snowmelt - snow lysimeter • Soil water - zero tension lysimeter • Talus water – biweekly to monthly Sample Analysis • Delta 18 O and major solutes

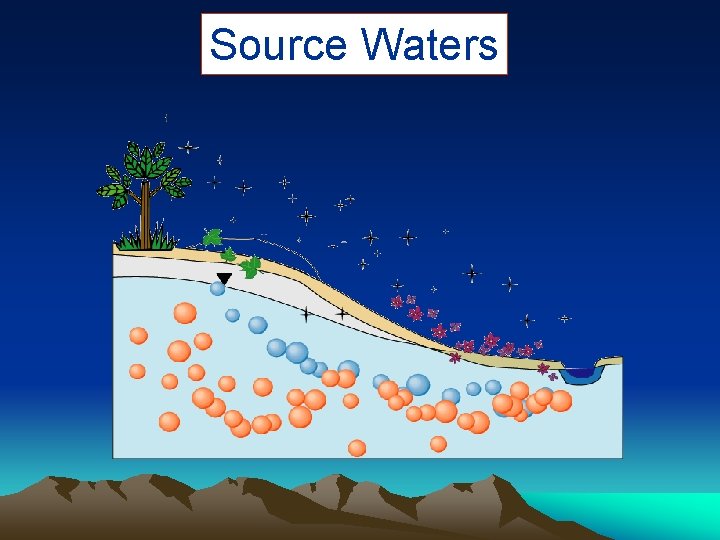
Source Waters

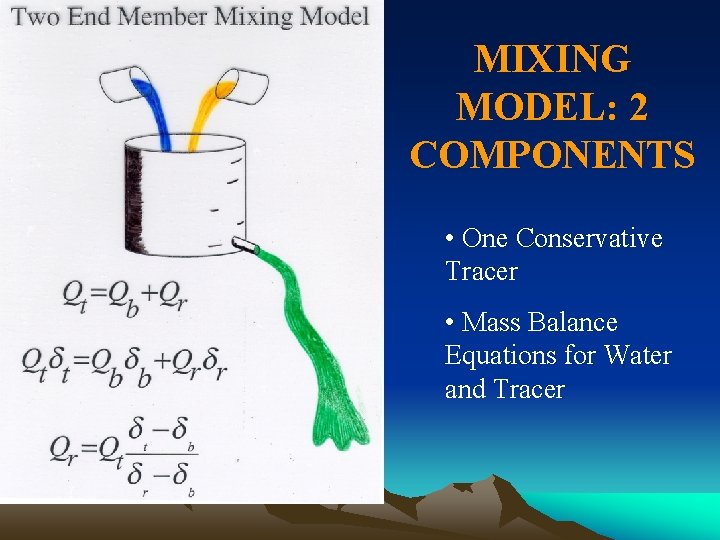
Mixing Models in Catchment Hydrology

MIXING MODEL: 2 COMPONENTS • One Conservative Tracer • Mass Balance Equations for Water and Tracer

ASSUMPTIONS FOR MIXING MODEL • Tracers are conservative (no chemical reactions); • All components have significantly different concentrations for at least one tracer; • Tracer concentrations in all components are temporally constant or their variations are known; • Tracer concentrations in all components are spatially constant or treated as different components; • Unmeasured components have same tracer concentrations or don’t contribute significantly.

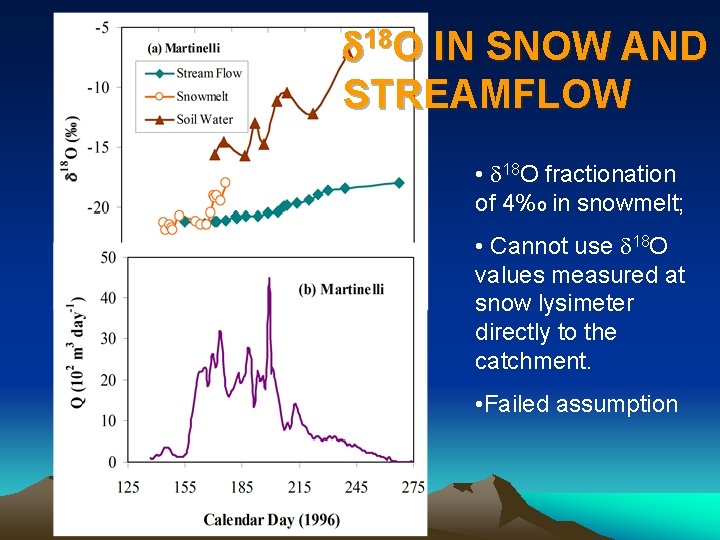
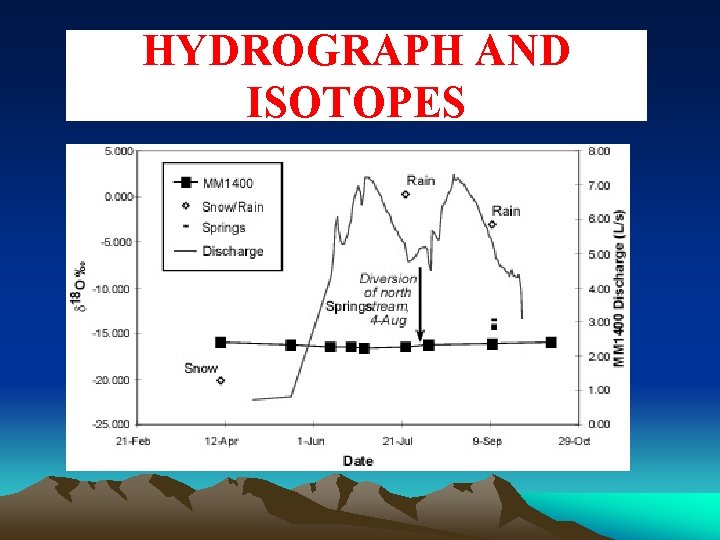
d 18 O IN SNOW AND STREAMFLOW • d 18 O fractionation of 4%o in snowmelt; • Cannot use d 18 O values measured at snow lysimeter directly to the catchment. • Failed assumption

Fractionation in Snowpack Snow surface d 18 O = -20‰ Ground d 18 O = -22‰ Difference between maximum 18 O values and Minimum 18 O values about 4 ‰

Isotopic Fractionation • Fractionation occurs as melt from surface percolates towards bottom of snowpack • Isotopic exchange between ice and percolating liquid water • d 18 Osolid > d 18 Oliquid > d 18 Ovapour • Molecules of surface meltwater are not the same as the ones at the base of the snowpack

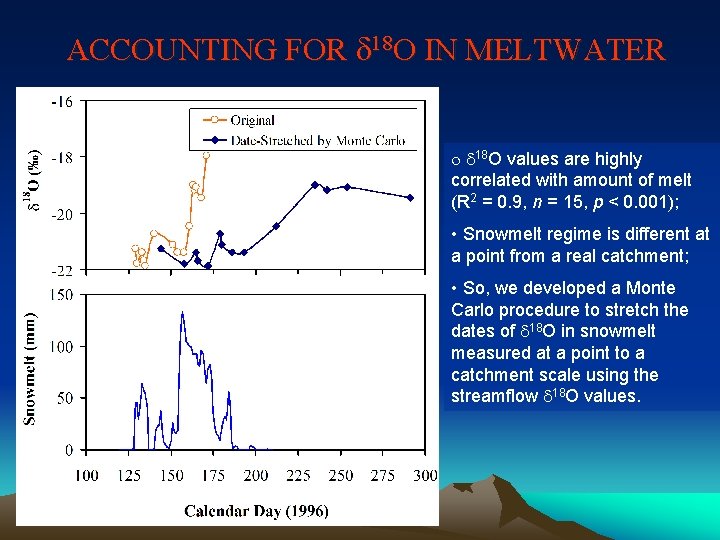
ACCOUNTING FOR d 18 O IN MELTWATER o d 18 O values are highly correlated with amount of melt (R 2 = 0. 9, n = 15, p < 0. 001); • Snowmelt regime is different at a point from a real catchment; • So, we developed a Monte Carlo procedure to stretch the dates of d 18 O in snowmelt measured at a point to a catchment scale using the streamflow d 18 O values.

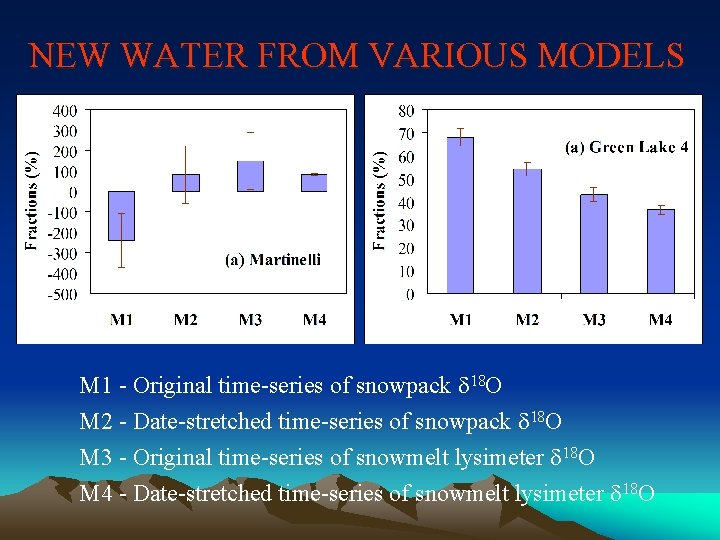
NEW WATER FROM VARIOUS MODELS M 1 - Original time-series of snowpack d 18 O M 2 - Date-stretched time-series of snowpack d 18 O M 3 - Original time-series of snowmelt lysimeter d 18 O M 4 - Date-stretched time-series of snowmelt lysimeter d 18 O

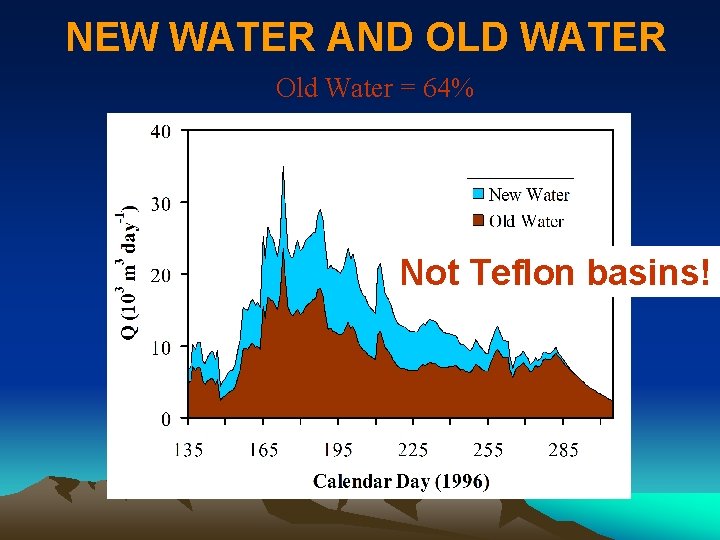
NEW WATER AND OLD WATER Old Water = 64% Not Teflon basins!

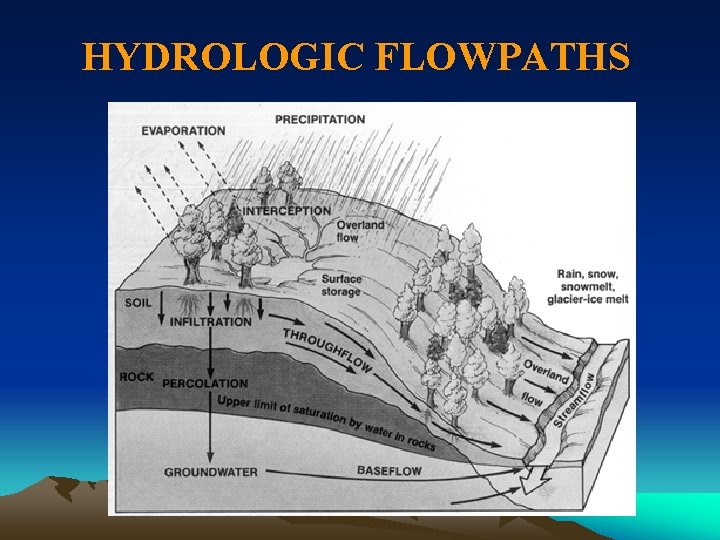
HYDROLOGIC FLOWPATHS

MIXING MODEL: 3 COMPONENTS Simultaneous Equations Solutions • Two Conservative Tracers • Mass Balance Equations for Water and Tracers Q - Discharge C - Tracer Concentration Subscripts - # Components Superscripts - # Tracers

MIXING MODEL: 3 COMPONENTS (Using Discharge Fractions) Simultaneous Equations Solutions • Two Conservative Tracers • Mass Balance Equations for Water and Tracers f - Discharge Fraction C - Tracer Concentration Subscripts - # Components Superscripts - # Tracers

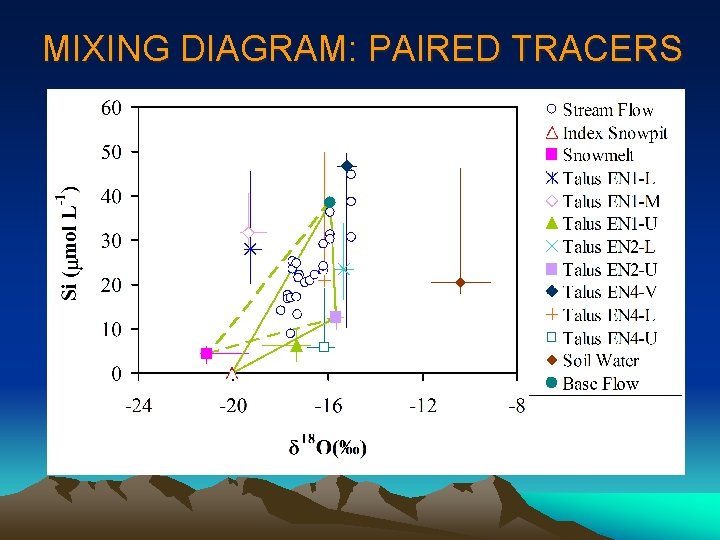
MIXING DIAGRAM: PAIRED TRACERS

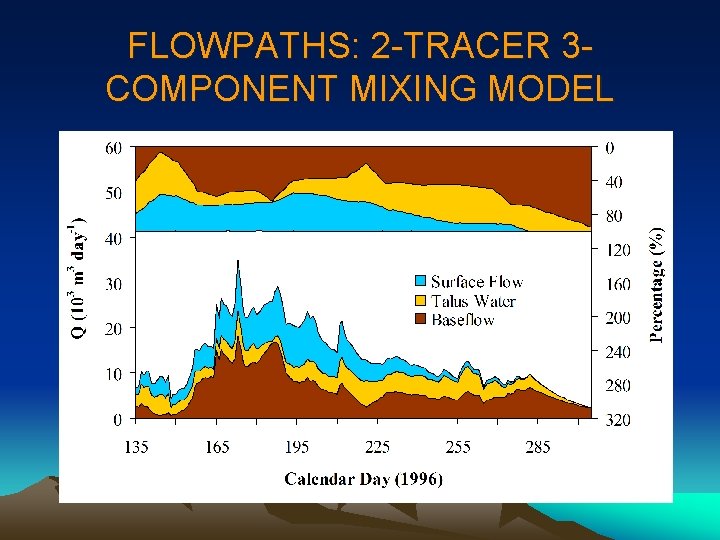
FLOWPATHS: 2 -TRACER 3 COMPONENT MIXING MODEL

FLOWPATHS: 2 -TRACER 3 -COMPONENT MIXING • Did we choose the right end-members? • Did we choose the right tracers? • Is there any way to quantitatively evaluate our results?

END-MEMBER MIXING ANALYSIS (EMMA) • Uses more tracers than components • Decides number of end-members • Quantitatively select end-members • Quantitatively evaluate results of the mixing model

MIXING MODEL: Generalization Using Matrices Simultaneous Equations Where • One tracer for 2 components and two tracers for 3 components • N tracers for N+1 components? -- Yes • However, solutions would be too difficult for more than 3 components • So, matrix operation is necessary Solutions Note: • Cx-1 is the inverse matrix of Cx • This procedure can be generalized to N tracers for N+1 components

This slide is from Hooper, 2001

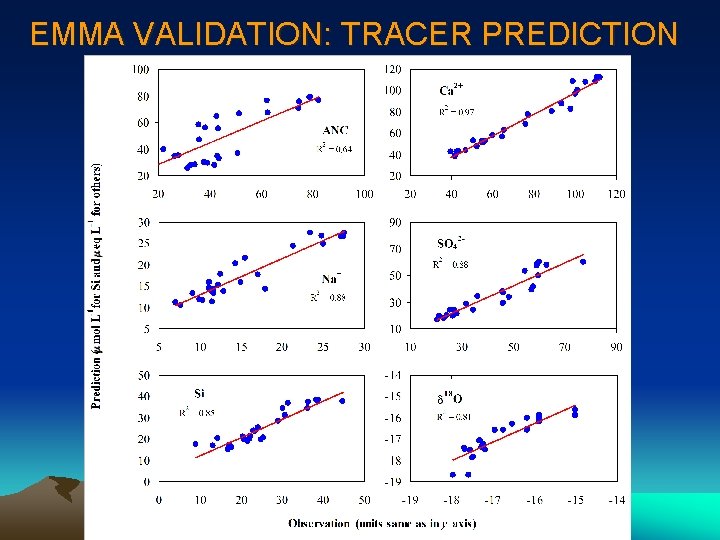
EMMA PROCEDURES • Identification of Conservative Tracers - Bivariate solute-solute plots to screen data; • PCA Performance - Derive eigenvalues and eigenvectors; • Orthogonal Projection - Use eigenvectors to project chemistry of streamflow and end-members; • Screen End-Members - Calculate Euclidean distance of endmembers between their original values and S-space projections; • Hydrograph Separation - Use orthogonal projections and generalized equations for mixing model to get solutions! • Validation of Mixing Model - Predict streamflow chemistry using results of hydrograph separation and original end-member concentrations.

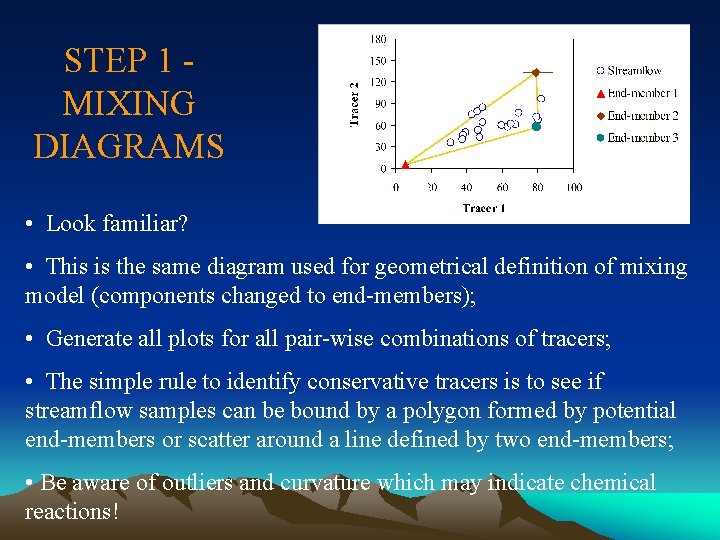
STEP 1 MIXING DIAGRAMS • Look familiar? • This is the same diagram used for geometrical definition of mixing model (components changed to end-members); • Generate all plots for all pair-wise combinations of tracers; • The simple rule to identify conservative tracers is to see if streamflow samples can be bound by a polygon formed by potential end-members or scatter around a line defined by two end-members; • Be aware of outliers and curvature which may indicate chemical reactions!

STEP 2 - PCA PERFORMANCE • For most cases, if not all, we should use correlation matrix rather than covariance matrix of conservative solutes in streamflow to derive eigenvalues and eigenvectors; • Why? This treats each variable equally important and unitless; • How? Standardize the original data set using a routine software or minus mean and then divided by standard deviation; • To make sure if you are doing right, the mean should be zero and variance should be 1 after standardized!

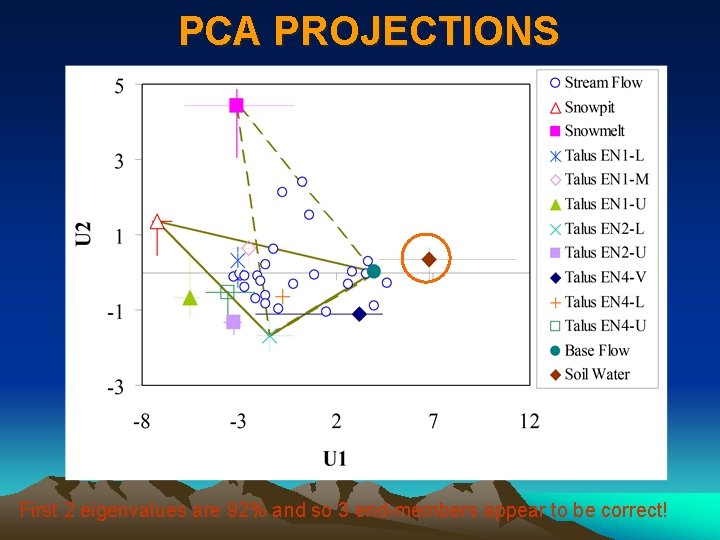
APPLICATION OF EIGENVALUES • Eigenvalues can be used to infer the number of end-members that should be used in EMMA. How? • Sum up all eigenvalues; • Calculate percentage of each eigenvalue in the total eigenvalue; • The percentage should decrease from PCA component 1 to p (remember p is the number of solutes used in PCA); • How many eigenvalues can be added up to 90% (somewhat subjective! No objective criteria for this!)? Let this number be m, which means the number of PCA components should be retained (sometimes called # of mixing spaces); • (m +1) is equal to # of end-members we use in EMMA.

PCA PROJECTIONS First 2 eigenvalues are 92% and so 3 end-members appear to be correct!

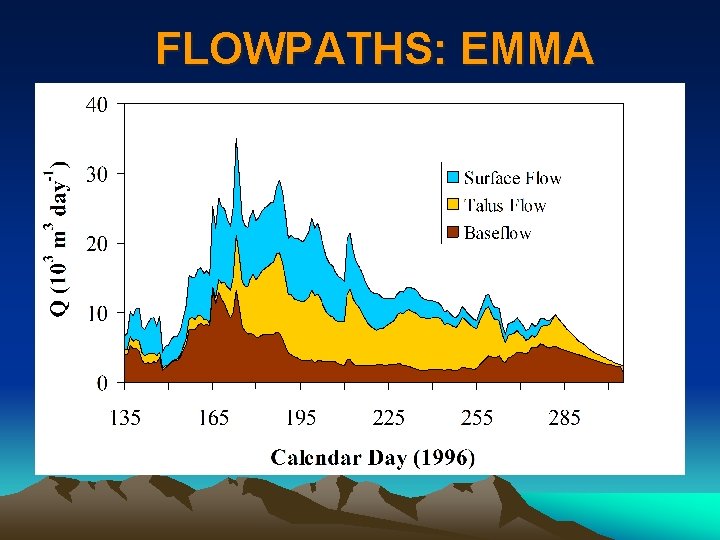
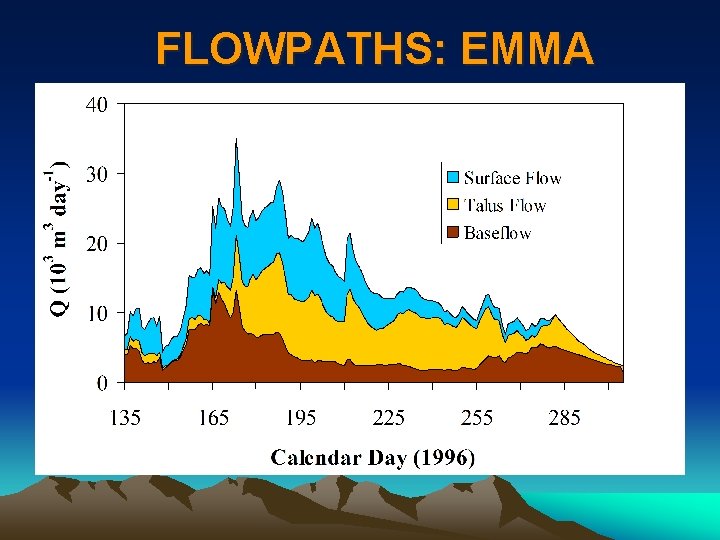
FLOWPATHS: EMMA

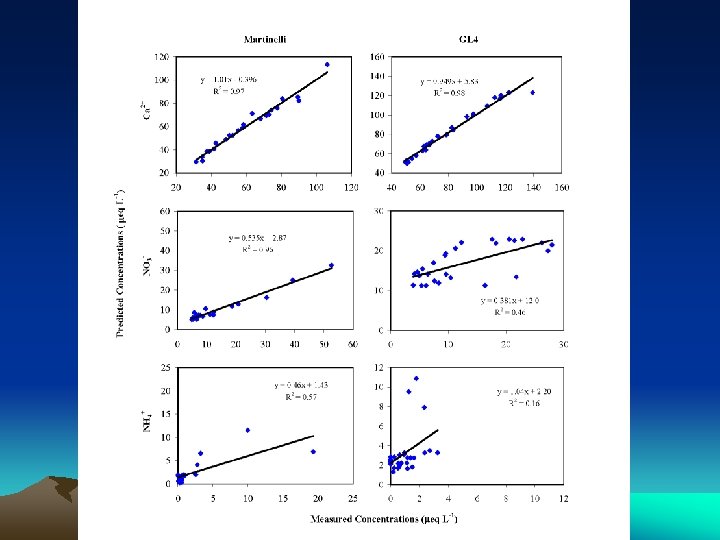
EMMA VALIDATION: TRACER PREDICTION

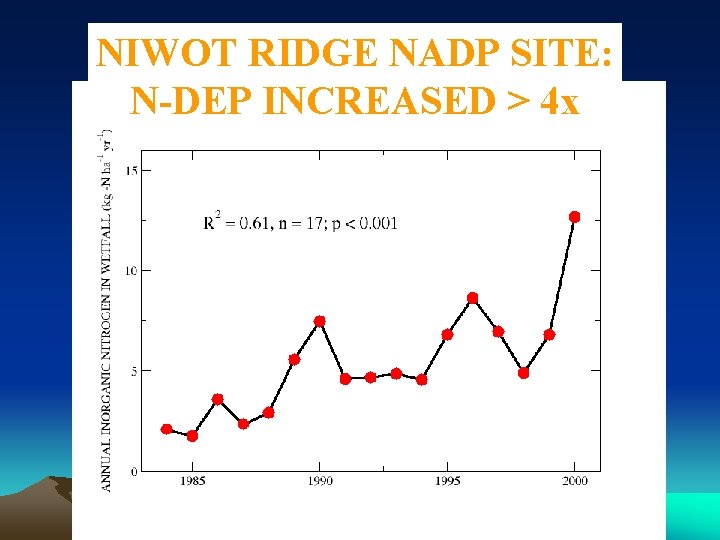
NITROGEN DEPOSITION

NIWOT RIDGE NADP SITE: N-DEP INCREASED > 4 x

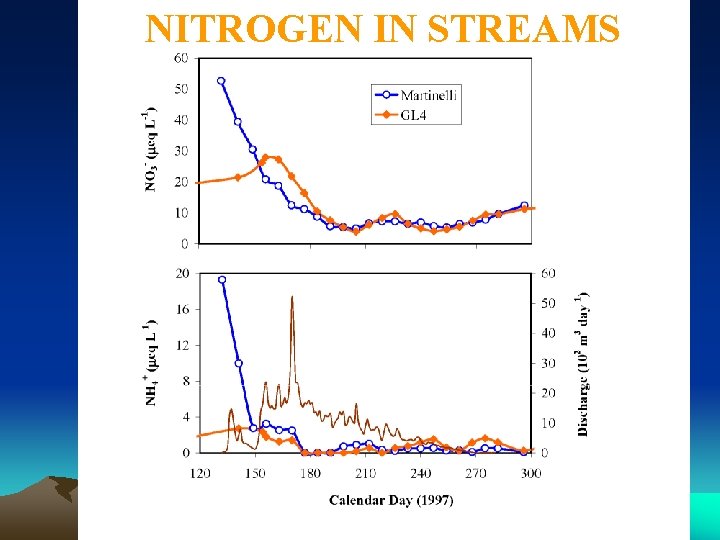
NITROGEN IN STREAMS

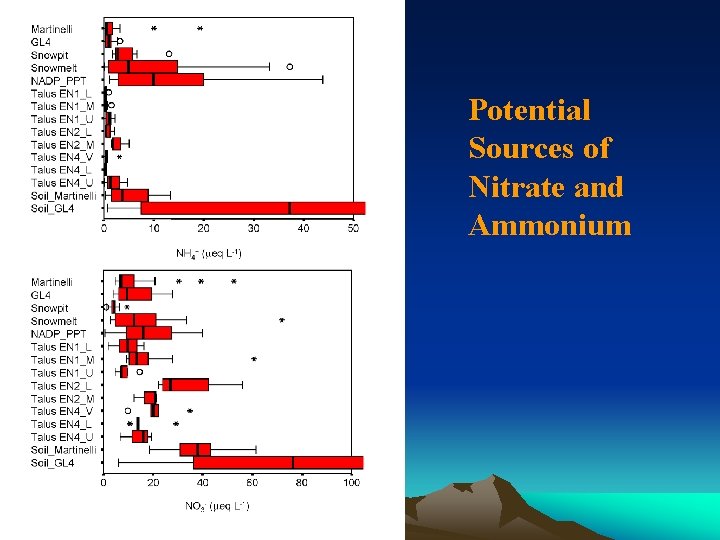
Potential Sources of Nitrate and Ammonium

FLOWPATHS: EMMA



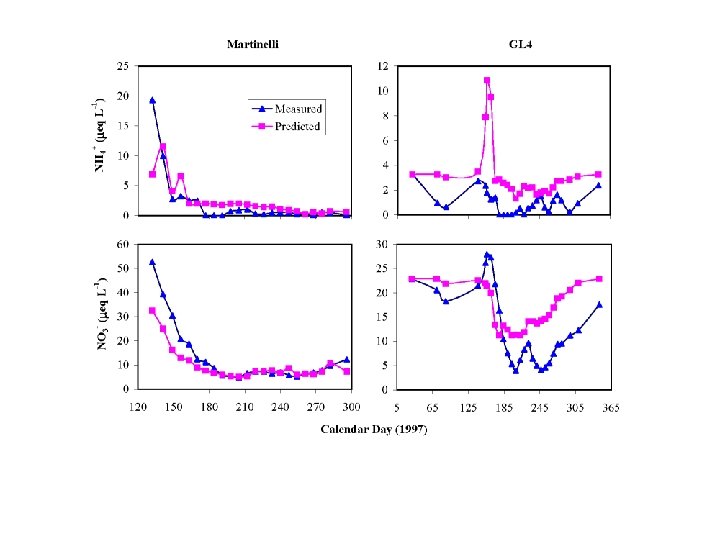
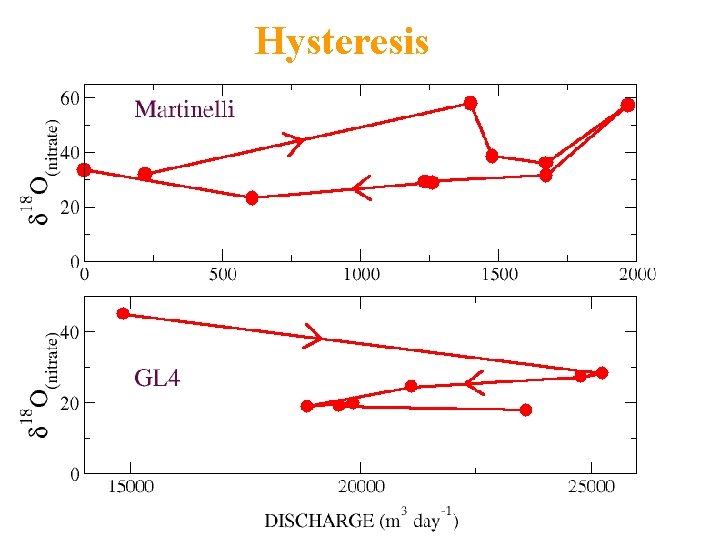
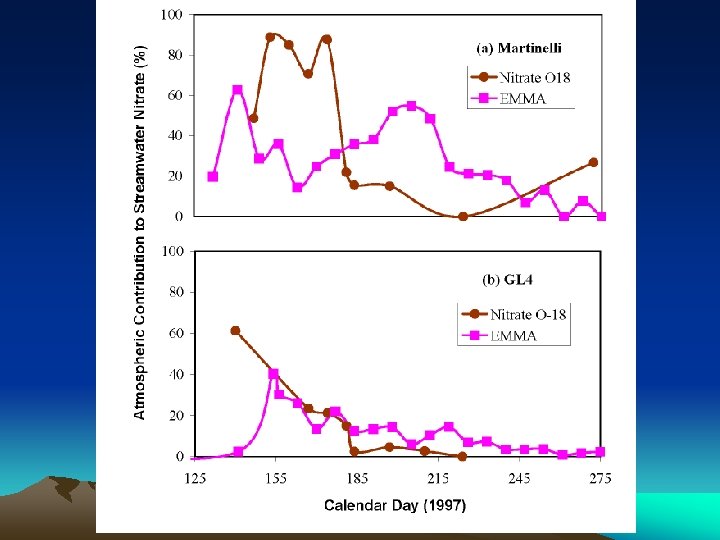
EMMA: NITRATE SOURCES • Under-predicts nitrate during snowmelt – Ionic pulse important • Overpredicts nitrate during summer – Denitrification? • 8 -ha Martinelli: 30% stream nitrate atmos • 225 -ha GL 4: 20% stream nitrate atmos

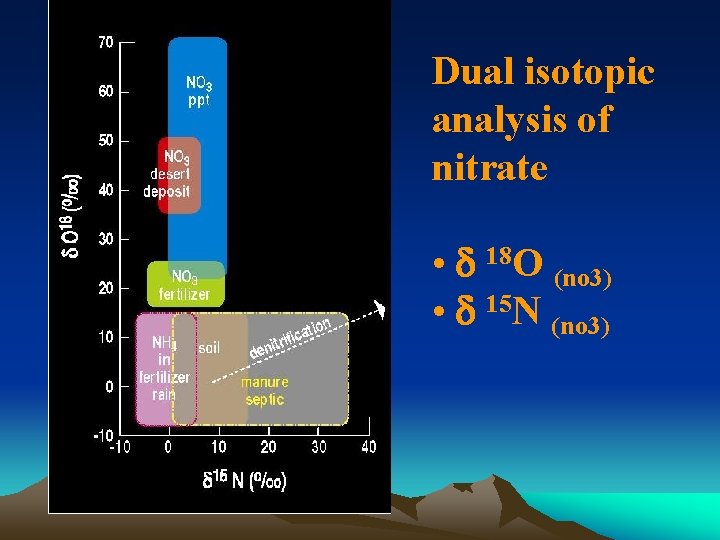
Dual isotopic analysis of nitrate • d 18 O (no 3) • d 15 N (no 3)

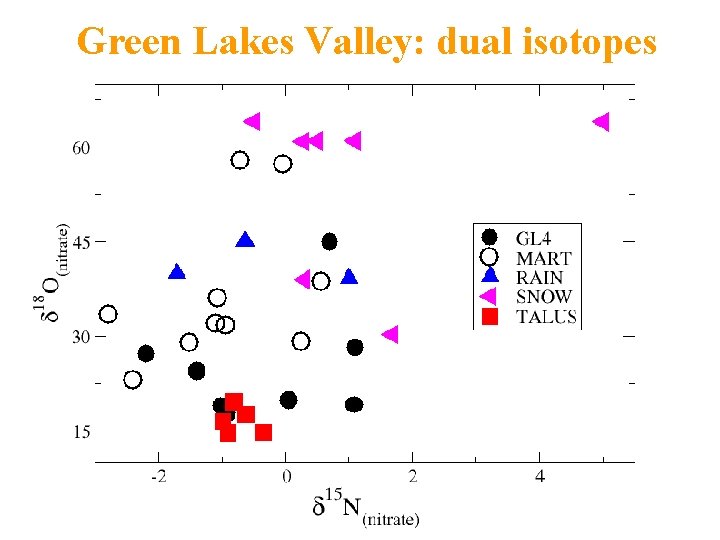
Green Lakes Valley: dual isotopes

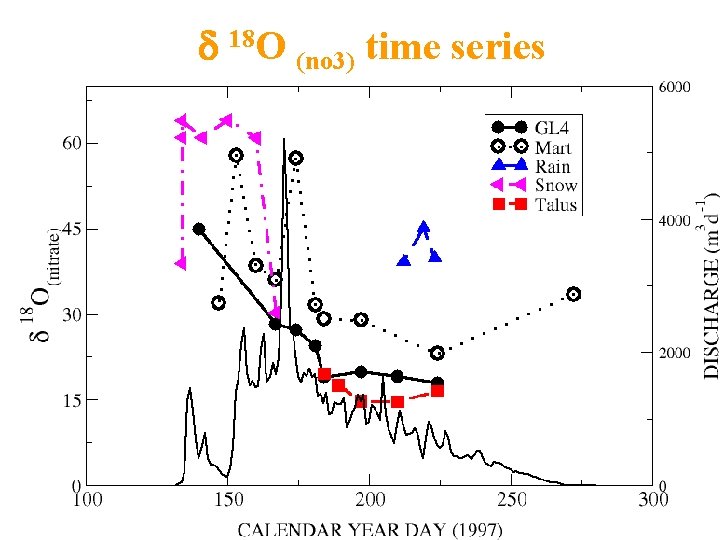
d 18 O (no 3) time series

Hysteresis

DUAL ISOTOPE: NITRATE SOURCES • 8 -ha Martinelli: 60% stream nitrate atmos • Twice EMMA • 225 -ha GL 4: 20% stream nitrate atmos • Same as EMMA


EMMA and NITRATE ISOTOPES • First time used together • 20% atmospheric nitrate in 220 -ha stream – EMMA, dual isotopes similar results • 60% atmospheric nitrate in 8 -ha stream – EMMA underestimates: unsampled flowpath • Talus nitrate microbial, not atmospheric • Denitrification probably very important

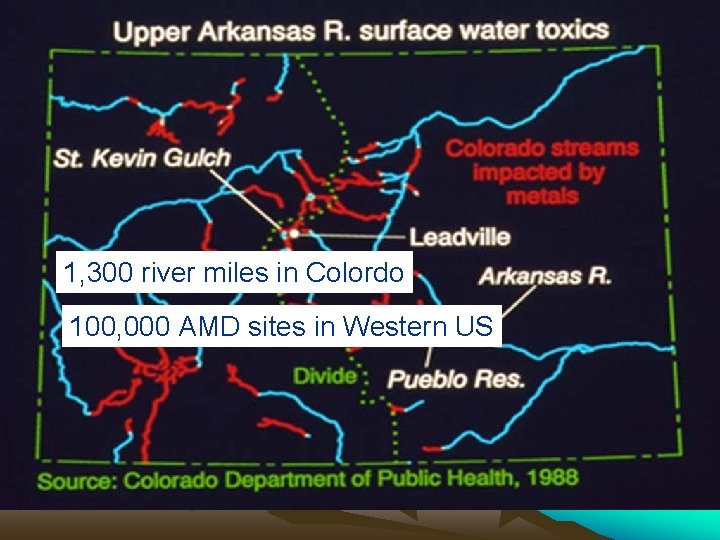
1 1, 300 river miles in Colordo 100, 000 AMD sites in Western US

END OF PIPE TREATMENT STRATEGY ¡ Millions of dollars to install ¡ Expensive to operate ¡ Operate for longterm ¡ Need low-cost alternatives


Watershed approach, CHALK CREEKhydrometric, MINE: isotopic, and chemical measurements GROUNDWATER SOURCE CONTROLS DEMONSTRATION PROJECT EPA VIII 104(b)3 Program SUPPLEMENTAL FUNDING REQUEST Assistance Agreement MM 998404 -02


HYDROGRAPH AND ISOTOPES

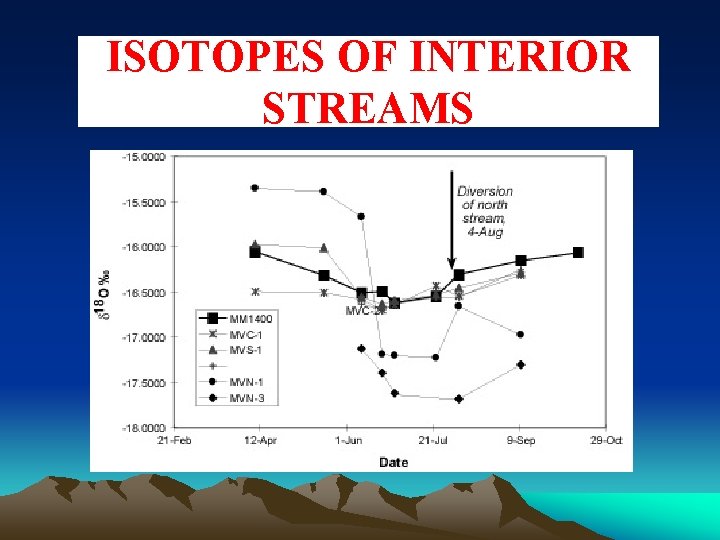
ISOTOPES OF INTERIOR STREAMS

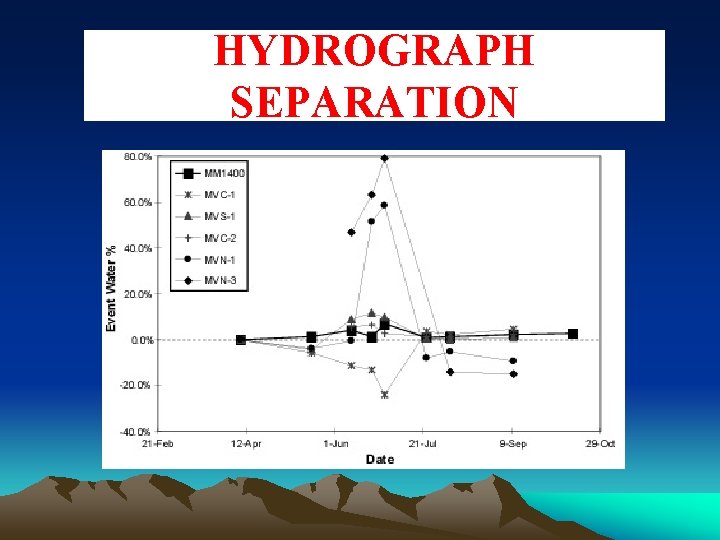
HYDROGRAPH SEPARATION

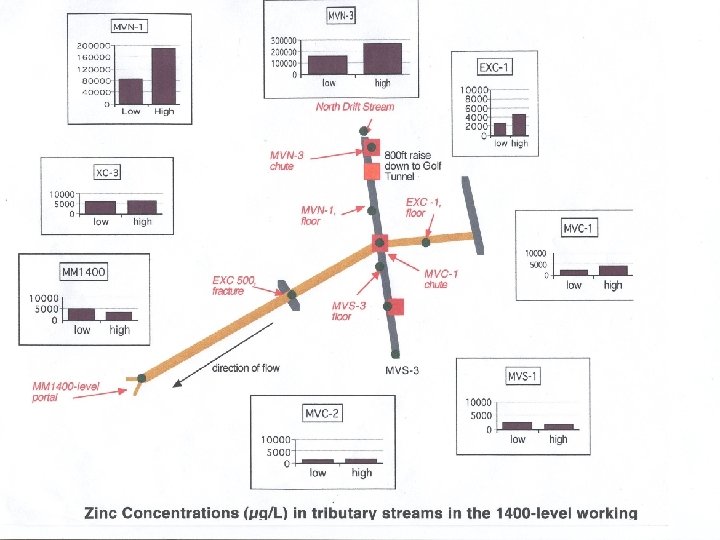
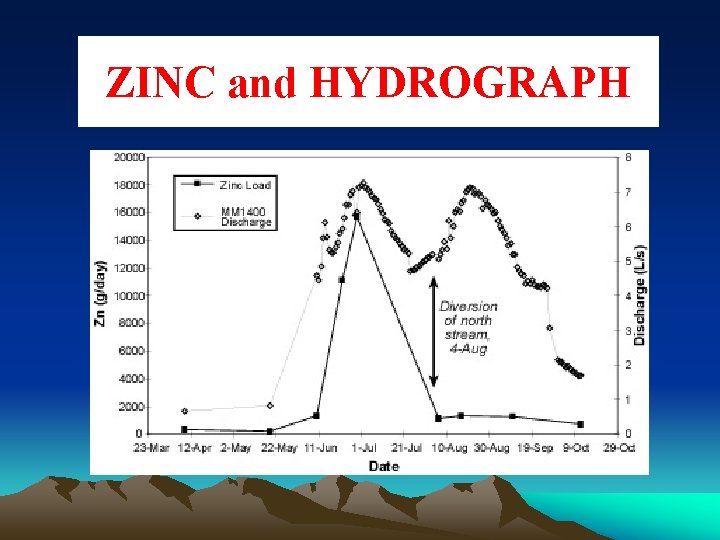
ZINC and HYDROGRAPH

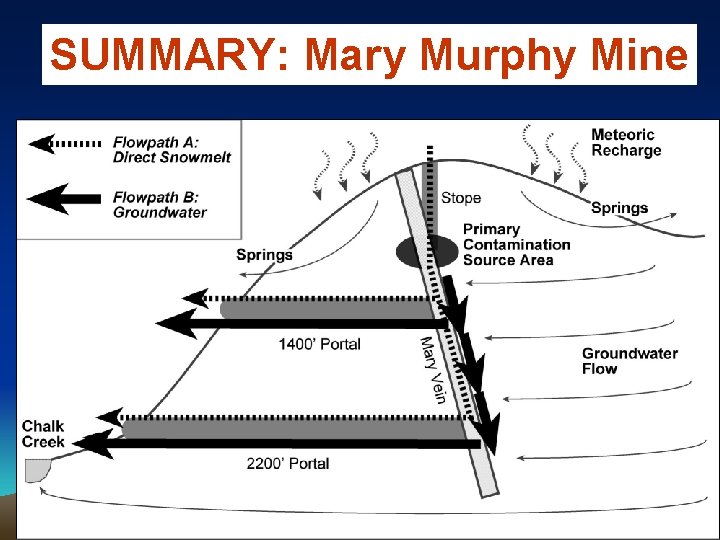
SUMMARY: Mary Murphy Mine

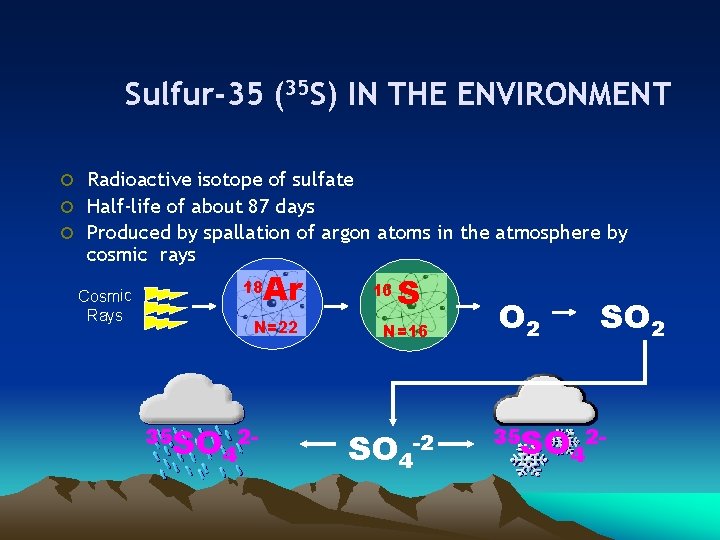
Sulfur-35 (35 S) IN THE ENVIRONMENT ¡ Radioactive isotope of sulfate ¡ Half-life of about 87 days ¡ Produced by spallation of argon atoms in the atmosphere by cosmic rays Cosmic Rays 18 Ar N=22 35 SO 24 16 S N=16 SO 4 -2 O 2 SO 2 35 SO 24



Tritium/3 He Dating • Tritium = 3 H • Radioactive isotope: • Half life of about 12 years • Naturally occurs in precip at about 6 -8 Tritium Units (TU) • Nuclear bomb testing in 1960’s created “bomb peak” (1000+ TU) • Can date waters younger than about 50 years:

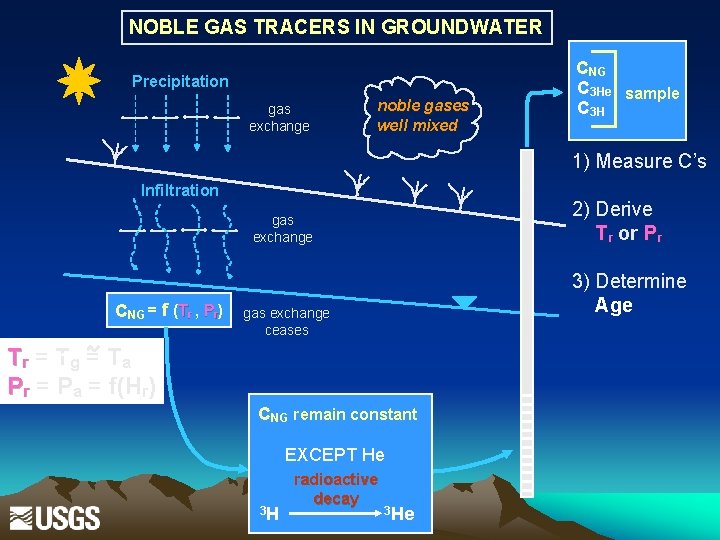
NOBLE GAS TRACERS IN GROUNDWATER Precipitation gas exchange noble gases well mixed CNG C 3 He sample C 3 H 1) Measure C’s Infiltration 2) Derive Tr or Pr gas exchange CNG = f (Tr , Pr) 3) Determine Age gas exchange ceases Tr = Tg = T a Pr = Pa = f(Hr) CNG remain constant EXCEPT He 3 H radioactive decay 3 He

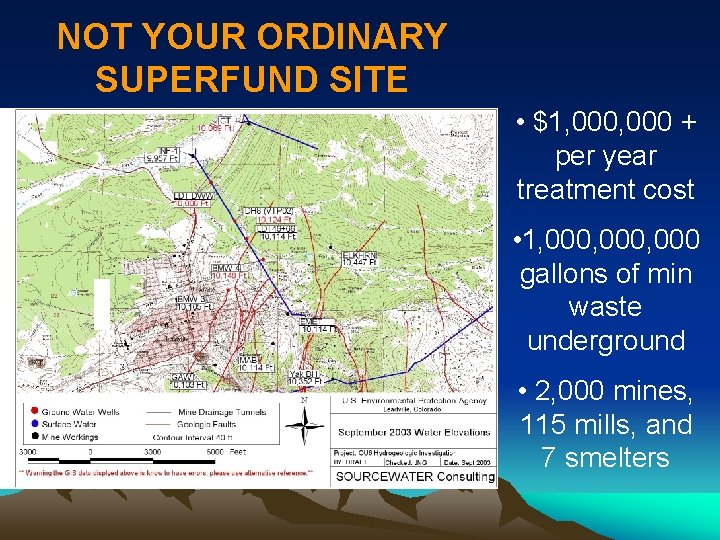
LEADVILLE EPA superfund site: $100, 000

NOT YOUR ORDINARY SUPERFUND SITE • $1, 000 + per year treatment cost • 1, 000, 000 gallons of min waste underground • 2, 000 mines, 115 mills, and 7 smelters

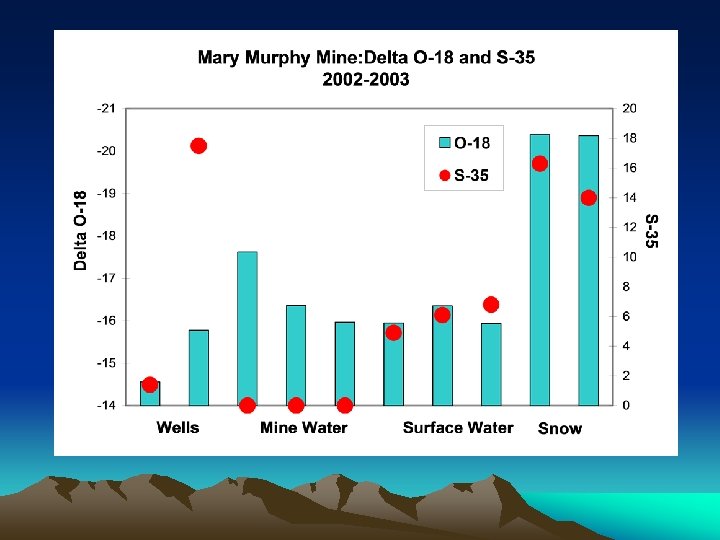
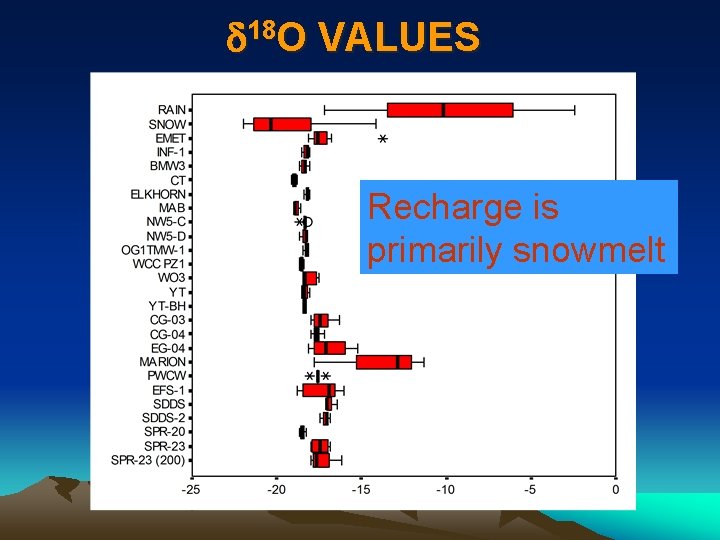
d 18 O VALUES Recharge is primarily snowmelt

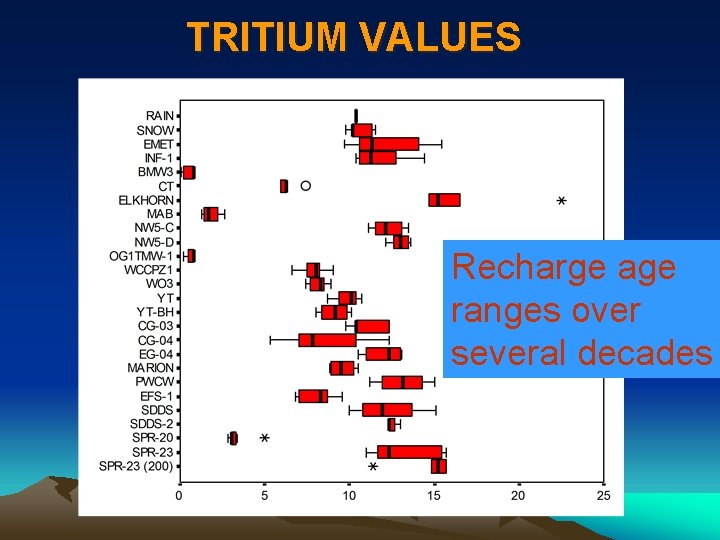
TRITIUM VALUES Recharge age ranges over several decades

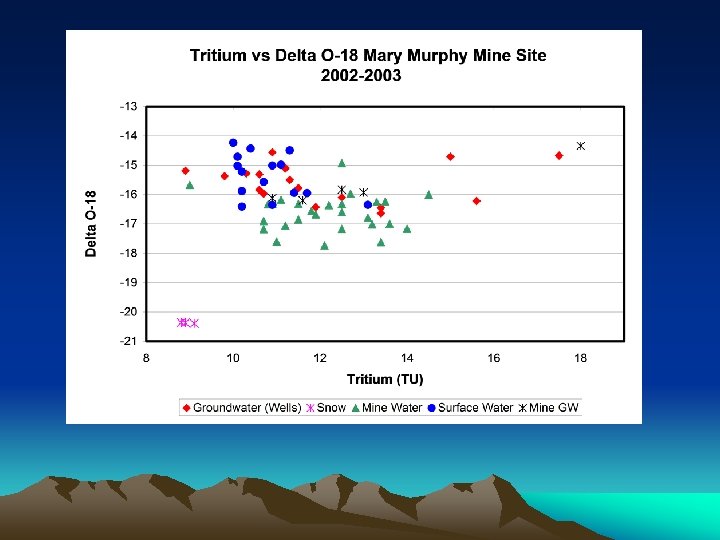
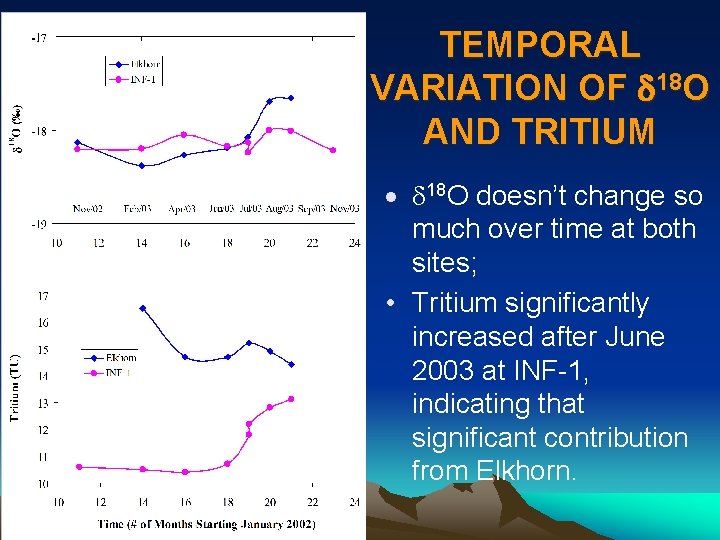
TEMPORAL VARIATION OF d 18 O AND TRITIUM · d 18 O doesn’t change so much over time at both sites; • Tritium significantly increased after June 2003 at INF-1, indicating that significant contribution from Elkhorn.

MIXING DIAGRAMS • Potential endmembers are clustered; • The bigger the circle, the higher the uncertainty in identifying a unique endmember; • But Elkhorn is unanimous as an end-member, which is natural groundwater well

Most snowmelt infiltrates into subsurface However, recharge rate is not known Subsurface storage is much larger than we thought and may be enough to support groundwater harvesting for Front Range Mountain block-piedmont connection unknown