General Organic and Biological Chemistry Fourth Edition Karen

General, Organic, and Biological Chemistry Fourth Edition Karen Timberlake Chapter 8 Solutions 8. 1 Solutions © 2013 Pearson Education, Inc. Lectures

Solutions: Solute and Solvent Solutions § are homogeneous mixtures of two or more substances. § contain a solute that is uniformly dispersed in another substance called the solvent. © 2013 Pearson Education, Inc. Chapter 8, Section 1 2

Nature of Solutes in Solutions Solutes § are spread uniformly throughout the solvent. § are not visible, but can give a color to the solution. § are usually present in a smaller amount than the solvent. © 2013 Pearson Education, Inc. Chapter 8, Section 1 3

Examples of Solutions The solute and solvent in a solution can be a solid, liquid, and/or a gas. © 2013 Pearson Education, Inc. Chapter 8, Section 1 4

Learning Check Identify the solute in each of the following solutions. 1. 2 g sugar and 100 m. L water 2. 60. 0 m. L of ethyl alcohol and 30. 0 m. L of methyl alcohol 3. 55. 0 m. L water and 1. 50 g Na. Cl 4. air: 200 m. L O 2 and 800 m. L N 2 © 2013 Pearson Education, Inc. Chapter 8, Section 1 5

Solution Identify the solute in each of the following solutions. Solute 1. 2 g sugar and 100 m. L water sugar 2. 60. 0 m. L of ethyl alcohol and 30. 0 m. L of methyl alcohol 3. 55. 0 m. L water and 1. 50 g Na. Cl 4. air: 200 m. L O 2 and 800 m. L N 2 O 2 © 2013 Pearson Education, Inc. Chapter 8, Section 1 6

Water § is the most common solvent in nature. § is a polar molecule. § forms hydrogen bonds between the partially positive hydrogen atom in one molecule and the partially negatively charged oxygen atom in a different water molecule. © 2013 Pearson Education, Inc. Chapter 8, Section 1 7

Water in the body § The average adult body contains 60% water by mass. § Dehydration occurs when as little as 10% of body fluids are lost. § Water lost can be replaced by liquids and foods in the diet. © 2013 Pearson Education, Inc. Chapter 8, Section 1 8

Formation of a Solution § To form a solution, the solute and solvent must have like polarities. § The energy between the solute and solvent particles must be strong enough to overcome the initial attractive forces between the solvent molecules and solute particles. © 2013 Pearson Education, Inc. Chapter 8, Section 1 9

Solutions with Ionic and Polar Solutes Na+ and Cl– ions § on the surface of a Na. Cl crystal are attracted to polar water molecules. § are hydrated in solution by many H 2 O molecules surrounding each ion. © 2013 Pearson Education, Inc. Chapter 8, Section 1 10

Equations for Solution Formation When Na. Cl(s) dissolves in water, the reaction can be written as: © 2013 Pearson Education, Inc. Chapter 8, Section 1 11

Learning Check Solid Li. Cl dissolves in water because: A. The Li+ ions are attracted to the oxygen atom ( ) of water. B. The Li+ ions are attracted to the hydrogen atom ( +) of water. C. The Cl– ions are attracted to the oxygen atom ( ) of water. D. The Cl– ions are attracted to the hydrogen atom ( +) of water. E. both A and D F. both B and C © 2013 Pearson Education, Inc. Chapter 8, Section 1 12

Solution Solid Li. Cl dissolves in water because: A. The Li+ ions are attracted to the oxygen atom ( ) of water. D. The Cl– ions are attracted to the hydrogen atom ( +) of water. E. both A and D © 2013 Pearson Education, Inc. Chapter 8, Section 1 13

Water and a Polar Solute Molecules of polar covalent compound methanol, CH 3 OH, form hydrogen bonds with polar water molecules to form a methanol–water solution. © 2013 Pearson Education, Inc. Chapter 8, Section 1 14

Like Dissolves Like Two substances form a solution when § there is an attraction between the particles of the solute and solvent. § a polar solvent, such as water, dissolves polar solutes, such as sugar and/or ionic solutes, such as Na. Cl. § a nonpolar solvent, such as hexane (C 6 H 14) dissolves nonpolar solutes, such as oil or grease. © 2013 Pearson Education, Inc. Chapter 8, Section 1 15
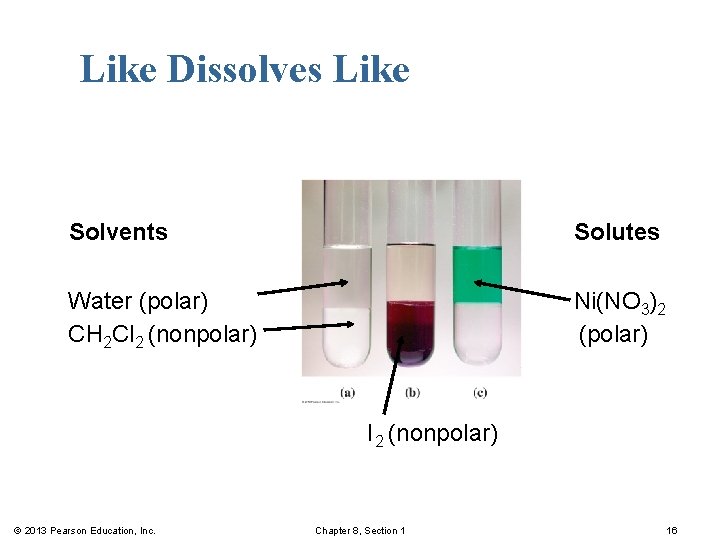
Like Dissolves Like Solvents Solutes Water (polar) CH 2 Cl 2 (nonpolar) Ni(NO 3)2 (polar) I 2 (nonpolar) © 2013 Pearson Education, Inc. Chapter 8, Section 1 16

Learning Check Will each of the following solutes dissolve in water? Why or why not? A. B. C. D. Na 2 SO 4 gasoline (nonpolar) I 2 HCl © 2013 Pearson Education, Inc. Chapter 8, Section 1 17

Solution Will each of the following solutes dissolve in water? Why or why not? A. B. C. D. Na 2 SO 4 gasoline I 2 HCl Yes, the solute is ionic. No, the solute is nonpolar. Yes, the solute is polar. Most polar and ionic solutes dissolve in water because water is a polar solvent. © 2013 Pearson Education, Inc. Chapter 8, Section 1 18
- Slides: 18