Solutions Edward Wen Learning Outcomes Solutions applying solubility

Solutions Edward Wen

Learning Outcomes • Solutions (applying solubility rules, calculations with concentrations and dilutions) • Definition and description of the properties of Colloids • Definition and description of the properties of Suspensions • Osmotic pressure effects on cells – defining isotonic, hypertonic 2

Solution • Homogeneous mixtures ü composition may vary from one sample to another ü appears to be one substance, though contains multiple materials • most Homogeneous materials are actually Solutions ü Gas state: common air ü Liquid: Gasoline (dozens of compounds), Soda water (sugar or asparatame, CO 2, citric acid, fructose) ü Solid: Alloy such as brass 3

Solutions: Solute + Solvent • Solute: the dissolved substance. Sugar in Soda üseems to “disappear” ü“takes on the state” of the solvent • Solvent: the substance solute dissolves in. Water in Soda üdoes not appear to change state • Aqueous solutions: solutions in which the solvent is water. 4

Common Types of Solution Solute Solvent Solution Phase Example Gaseous solutions gas air (mostly N 2 & O 2) gas liquid soda (CO 2 in H 2 O) Liquid solutions liquid vodka (C 2 H 5 OH in H 2 O) solid liquid seawater (Na. Cl in H 2 O) Solid solutions solid brass (Zn in Cu) • Alloys: solutions that contain Metal solutes and a Metal solvent, such as Nickel (5 cents of 1$), Brass, Stainless steel 5

How Soluble? Solubility • Soluble: when one substance (solute) dissolves in another (solvent) Homogeneous üSalt and Sugar are soluble in water: Saline and Soda üAcetic acid (HC 2 H 3 O 2) in water: Vinegar üOxygen gas in Nitrogen gas: Air • Insoluble: when one substance does not dissolve in another Heterogeneous üOil is insoluble in water: Italian salad dressing 6

Will It Dissolve? Chemist’s Rule of Thumb – Like Dissolves Like • a chemical will dissolve in a solvent if it has a similar structure to the solvent • when the solvent and solute structures are similar, the solvent molecules will attract the solute particles at least as well as the solute particles to each other 7
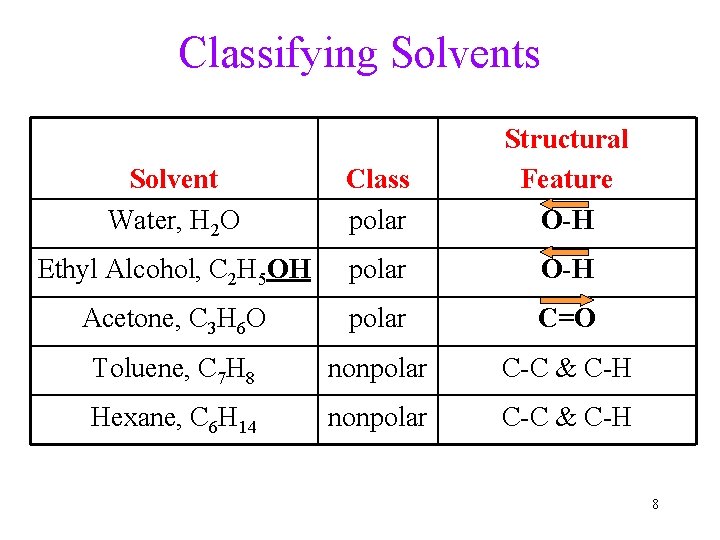
Classifying Solvents Solvent Water, H 2 O Class polar Structural Feature O-H Ethyl Alcohol, C 2 H 5 OH polar O-H Acetone, C 3 H 6 O polar C=O Toluene, C 7 H 8 nonpolar C-C & C-H Hexane, C 6 H 14 nonpolar C-C & C-H 8

Solubility in Water, A Polar Solvent? • Ionic compound (Yes): Ions are attracted to polar Water. Salt Na. Cl dissolve in water • Polar molecules (Yes): attracted to polar solvents ü table Sugar, Alcohol, glucose • Nonpolar molecules are NOT attracted to Water ü b-carotene, (C 40 H 56), is not water soluble; it dissolves in fatty (nonpolar) tissues Those molecules with both polar and nonpolar structures: depends on structural features in the molecule 9
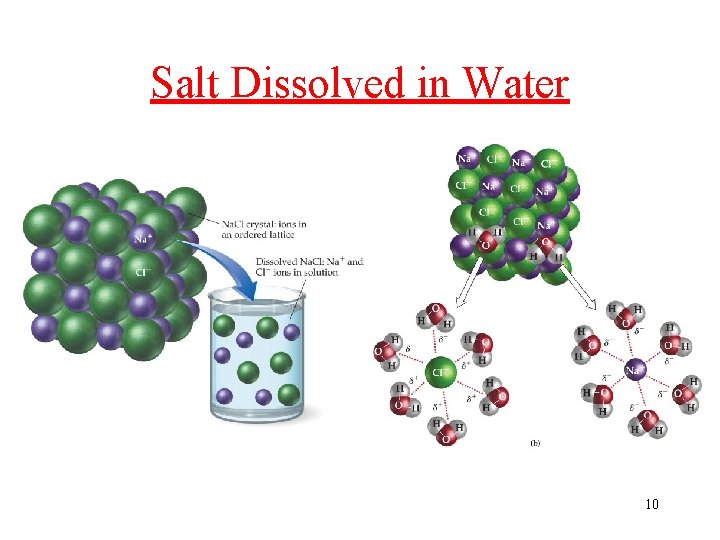
Salt Dissolved in Water 10

Solubility • Definition: the maximum amount of solute that can be dissolved in a given amount of solvent • Usually a limit to the solubility of one substance in another üExceptions: gases are always soluble in each other ütwo liquids that are mutually soluble are said to be miscible Øalcohol and water are miscible Øoil and water are immiscible 11

Descriptions of Solubility • Saturated solutions have the maximum amount of solute that will dissolve in that solvent at that temperature • Unsaturated solutions can dissolve more solute • Supersaturated solutions are holding more solute than they should be able to at that temperature üunstable 12
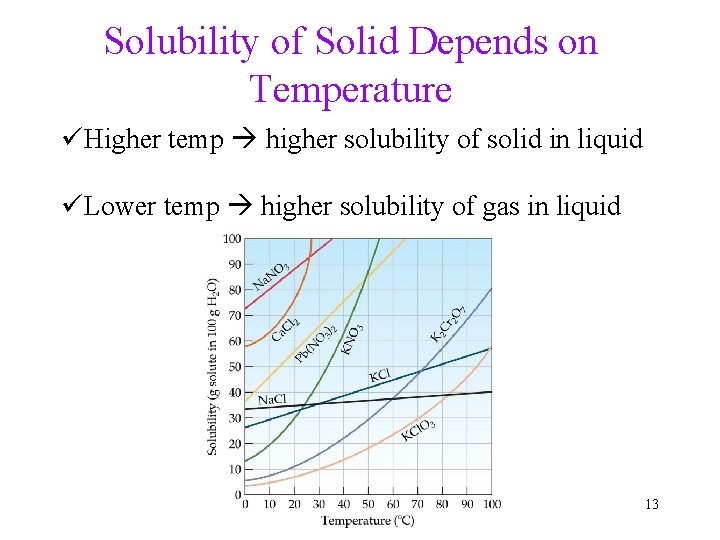
Solubility of Solid Depends on Temperature üHigher temp higher solubility of solid in liquid üLower temp higher solubility of gas in liquid 13

Solubility of Gases Decreases at higher Temperature Observation 1: Warm soda pop fizzes more than cold soda pop Cause: Solubility of CO 2 in water decreases as temperature increases. Observation 2: When water is heated up, gas bubbles appear even before boiling occurs. Cause: Solubility of air in water decreases as temperature increases. 14

Solubility of Gas depends on Pressure Henry’s Law: • higher pressure = higher solubility • CO 2 is dissolved under Pressure into bottled/canned soda 15

Under which conditions does sugar have the best solubility? A. B. C. D. high temperature and high pressure high temperature and low pressure low temperature and high pressure

Under which conditions does oxygen gas have the best solubility? A. high temperature and high partial pressure (PO ) B. high temperature and low partial pressure C. low temperature and low partial pressure D. low temperature and high partial pressure 2

Solution Concentration Descriptions • Concentrated solutions have high solute concentrations. Example: Soda syrup before mixed with carbonated water. • Diluted solutions have low solute concentrations. Example: Soda drink from adding carbonated water to syrup 18

Concentrations – Quantitative Descriptions of Solutions • Solutions have variable composition. Salt vs. Water in Seawater • To describe a solution accurately, you need to describe the components and their relative amounts • Concentration = amount of solute in a given amount of solution ü Seawater: Salt concentration 3. 4% ü Dead Sea: Salt concentration 30% ü Vinegar: Acetic acid concentration 5% 19

Weight/Volume Percent (w/v)% • mass of solute (gram) in every 100 m. L of solution ü Vinegar is 5% (w/v) solution of acetic acid by mass, then there are 5 grams of acetic acid in every 100 m. L of vinegar 20

Example: Chloraseptic sore throat spray contains 0. 35 g of phenol in 25 m. L of solution. Find the weight/volume percent concentration of phenol. Information Given: 0. 35 g phenol. 25 m. L solution Find: (w/v)% 21

Volume/Volume Percent (v/v)% • Volume of solute (m. L) in every 100 m. L of solution ü Common rubbing alcohol is 50~91% (v/v) solution of isopropanol (or 2 propanol) in every 100 m. L of solution. 22

Example: A 750 -m. L bottle of wine contains 101 m. L of ethanol. Find the volume/volume percent concentration of ethanol. Information Given: 101 m. L ethanol, 750 m. L solution (wine) Find: (v/v)% 23

Mass Percent (%) • mass of solute (gram) in every 100 gram of solution ü if a solution is 0. 9% by mass, then there are 0. 9 grams of solute in every 100 grams of solution • Mass of solution = Mass of solute + Mass of solvent 24

Example: Calculate the mass percent of a solution containing 27. 5 g of ethanol (C 2 H 6 O) and 175 g of H 2 O Information Given: 27. 5 g C 2 H 6 O; 175 g H 2 O Find: % by mass Eq’n: = 13. 5802% = 13. 6% 25

Parts per million (ppm) concentration • mass of solute (gram) in every 1, 000 gram of solution • Or volume of solute (m. L) in every 1, 000 milliliter of solution 26

Using Concentrations as Conversion Factors • concentrations show the relationship between the amount of solute and the amount of solvent ü 12% by mass sugar(aq) means 12 g sugar 100 g solution • The concentration can then be used to convert the amount of solute into the amount of solution, or visa versa 27

Example: A soft drink contains 11. 5% (w/v) sucrose (C 12 H 22 O 11). What volume of soft drink in milliliters contains 85. 2 g of sucrose? Information Given: 85. 2 g C 12 H 22 O 11 Find: m. L sol’n CF: 11. 5 g C 12 H 22 O 11 100 m. L sol’n SM: g sucrose → g sol’n → m. L sol’n 741 m. L 28

Example: Rubbing alcohol contains 85% (v/v) isopropanol. What volume of isopropanol is contained in 500. m. L rubbing alcohol? Information Given: 500. m. L rubbing alcohol Find: m. L sol’n CF: 85 g isopropanol 100 m. L rubbing alcohol 4. 3 x 102 m. L 29

Molarity Concentration READ the label in the reagent bottle! bottle “What does ‘ 6 M HCl’ mean? ” • Definition: Moles of solute per 1 liter of solution • Purpose: describing how many molecules of solute in each liter of solution • Unit: mole/L, abbreviated as “M”. • If a sugar solution concentration is 2. 0 M , 1 liter of solution contains 2. 0 moles of sugar, 2 liters = 4. 0 moles sugar, 0. 5 liters = 1. 0 mole sugar moles of solute molarity = liters of solution 30

Why Molarity? Many reagents used in chemistry, even many biology labs, are in the form of solution. Molarity concentration of solution is particularly important and useful because • Easy to prepare a solution to a given molarity • Easy to use: To obtain given amount (mole) of reagent, just calculate the volume of solution to be used: Volume (L) = mole molarity 31

Example: Calculating Molarity

Example: Calculate the molarity of a solution made from 15. 5 g of Na. Cl in 250 m. L solution. Information Given: 250 m. L solution; 15. 5 g Na. Cl; 1. 06 M Na. Cl 33

Calculations involving Molarity = mole Volume (L) • Solve for mole: Mole = Molarity Volume (L) • Solve for volume of solution in liters: Volume (L) = mole molarity 34

Example: How many liters of a 0. 114 M Na. OH solution contains 1. 24 mol of Na. OH? Information Given: 1. 24 mol Na. OH Find: L solution CF: 0. 114 mol = 1 L SM: mol → L 35

Example: How many moles of (NH 4)2 S are in 11. 25 m. L 0. 2334 M (NH 4)2 S solution? Information Given: 0. 2334 M (NH 4)2 S solution V = 11. 25 m. L Find: mol (NH 4)2 S CF: 0. 2334 mol = 1 L 0. 002626 mol 36

Example: How many grams of Na. Cl are in 10. 00 m. L 0. 2000 M Na. Cl solution? Information Given: 0. 2000 M Na. Cl solution V = 10. 00 m. L Find: gram Na. Cl CF: 1 L = 0. 2000 mol 1 mol Na. Cl = 58. 44 g Na. Cl 0. 1169 g 37

Dilution: More Solvent Added • When mixing more solvent into a solution, the volume of final solution is greater than the original solution • The mole of solute remains the same before and after mixing more solvent • The final concentration of solution is lower than in the beginning (diluted)

Making a Solution by Dilution: mole solute is the same = M 1 x V 1 = M 2 x V 2 Example: A student added 1. 00 L water to 2. 00 L 1. 00 M HCl. The final volume became 3. 00 L. What is the final concentration? 0. 667 M.

Example: Making solution from concentrate: What Volume of 12. 0 M KCl Is Needed to Make 5. 00 L of 1. 50 M KCl Solution? Given: Concentration Volume Initial solution 12. 0 M ? L Final solution 1. 50 M 5. 00 L 40

Example—Determine the Concentration of the Solution after dilution • A solution made by diluting 125 m. L of 0. 80 M HCl to 500 m. L. M 2 = 0. 20 M 41

Physical Properties Affected by Concentration Change in Concentration affects the physical properties of solution. • Vapor pressure of solvent • Freezing point of solution • Osmotic pressure
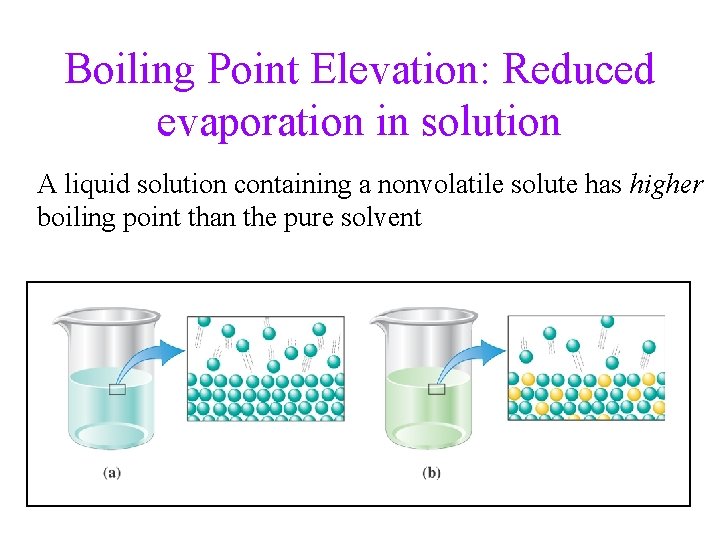
Boiling Point Elevation: Reduced evaporation in solution A liquid solution containing a nonvolatile solute has higher boiling point than the pure solvent

Freezing point Depression A liquid solution containing a nonvolatile solute has lower freezing point than the pure solvent. • A water-antifreeze mixture has a higher boiling point and lower freezing point than pure water. This will reduce the chance of engine coolant being frozen or being evaporated too quickly. • Spray salt on icy road help preventing ice on the road.

Osmosis Higher concentration of solution has stronger tendency to uptake solvent The liquid level in the tube rises until equilibrium is reached.

Osmosis Semi-permeable membrane separating (a) pure water and a salt-water solution, and (b) a dilute salt-water solution. Worm cruelty: salt on earthworm or snail too.

Osmotic Pressure and Reverse Osmosis (RO) Osmotic pressure is the amount of pressure needed to prevent the solution in the tube from rising as a result of the process of osmosis. RO: When the added pressure exceeds the osmotic pressure, pure water leaves the concentrated solution.
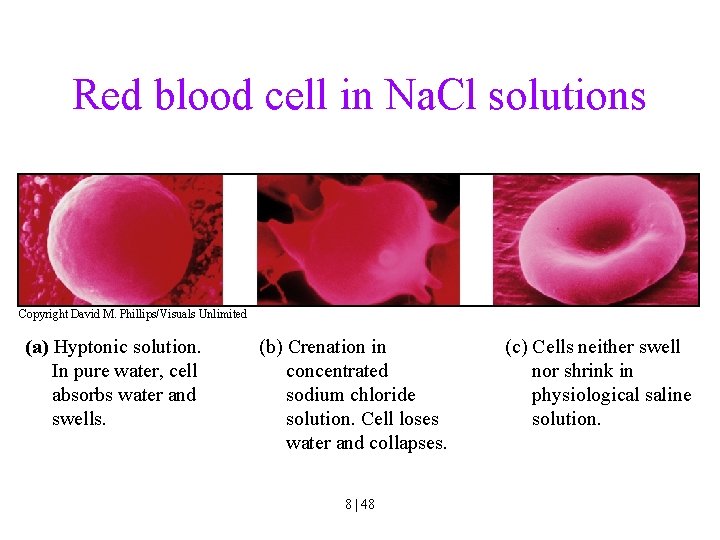
Red blood cell in Na. Cl solutions Copyright David M. Phillips/Visuals Unlimited (a) Hyptonic solution. In pure water, cell absorbs water and swells. (b) Crenation in concentrated sodium chloride solution. Cell loses water and collapses. 8 | 48 (c) Cells neither swell nor shrink in physiological saline solution.
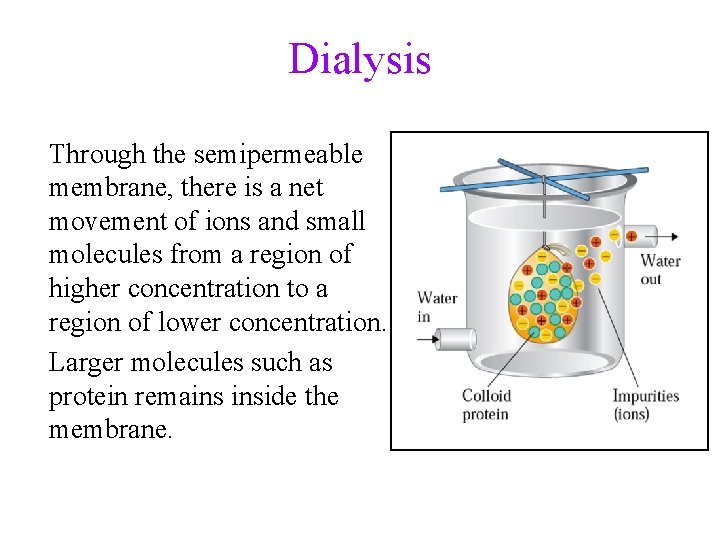
Dialysis Through the semipermeable membrane, there is a net movement of ions and small molecules from a region of higher concentration to a region of lower concentration. Larger molecules such as protein remains inside the membrane.
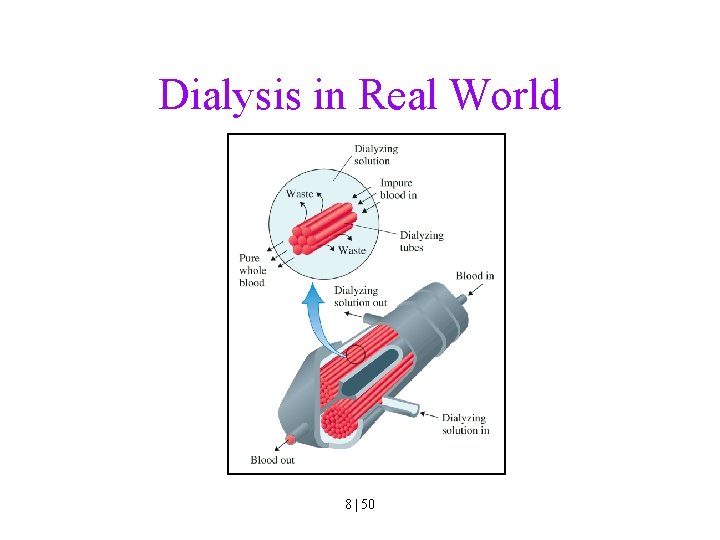
Dialysis in Real World 8 | 50
- Slides: 50