SATURATION Solutions Review Solute being dissolved Solvent doing

SATURATION

Solutions Review • Solute – being dissolved • Solvent – doing the dissolving • Soluble – can be dissolved by a solvent • Insoluble – can not be dissolved by a solvent • Solvation – the process of surrounding a solute particle by a solvent particle making a solution

Draw a solvated solution of potassium nitrate.

How to identify a solution: • Does is conduct electricity? – yes: electrolyte – no: non electrolyte • Does it scatter light? – Yes: not a solution but rather a colloid – No: can be a solution or a suspension (visible particles)

Solubility • Maximum amount of solute that can dissolve in a given amount of solvent at a specific temperature and pressure. • Defined as g solute/100 g solvent

A solution can be defined as: • Unsaturated • Super Saturated

Unsaturated Solution • Unsaturated – contains less dissolved solute for given amount of solvent (capable of dissolving more) – Kool-aid with not enough sugar capable of dissolving more Sponge can absorb more liquid
Saturated Solution • Contains maximum amount of dissolved solute (will not dissolve anymore and can often see undissolved solute) – Kool-aid with too much sugar or just the perfect amount Sponge can not absorb any more liquid – A saturated solution is in equilibrium (solute is dissolving and reforming at same rate

Super. Saturated Solution • Contains more dissolved solute than a saturated solution at a given temperature • Made by changing the conditions of the solution (temperature or pressure) Created from a super saturated solution of sugar water. • Solution is unstable and can often suddenly release excess solute
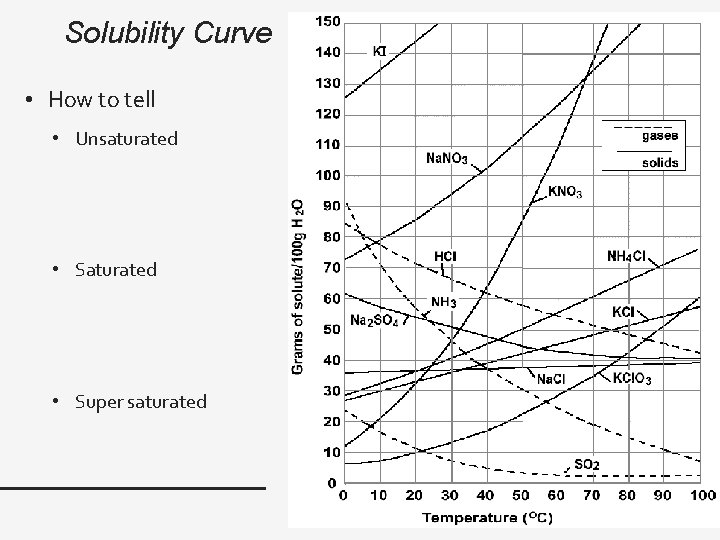
Solubility Curve • How to tell • Unsaturated • Super saturated
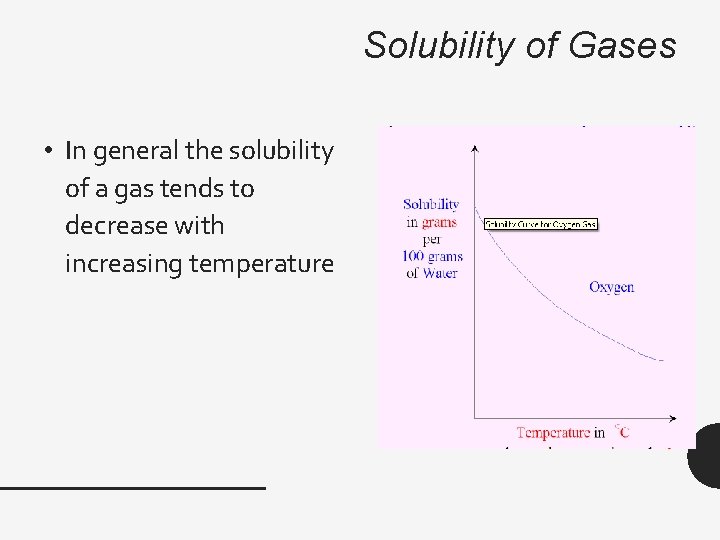
Solubility of Gases • In general the solubility of a gas tends to decrease with increasing temperature

How can you tell? • Add a seed crystal. • A saturated, unsaturated and super saturated solution can all look exactly the same.
• Practice pp 19 & 20

Saturation of Sodium Acetate Lab • Starting with 2 ml of water, you will add varying amounts of Na. C 2 H 3 O 2 to create a solution of varying saturation. • Very important to follow directions • Do not shake vigorously • Due Monday 4/24
- Slides: 14