Solubility DemoPacking Peanuts Solvent Solute Spacking peanuts Solute
Solubility Demo-Packing Peanuts Solvent Solute S-packing peanuts Solute 8 -packing peanuts Water Acetone 1. 2. Write your observations in each of the boxes. What can you conclude from your observations and why?

Solubility Why was the sleep-deprived chemistry student staring at the orange juice carton? Because it said “CONCENTRATE”

Units

Factors that Affect Solubility i. Temperature 1. Solids/liquids As the liquid’s temperature ↑, solubility ↑ 2. Gases As the liquid’s temperature ↑, solubility ↓ ii. Pressure (gases only) As the pressure↑, solubility ↑ As the pressure ↓, solubility ↓

Solid Solubility & Temp Salt Sugar
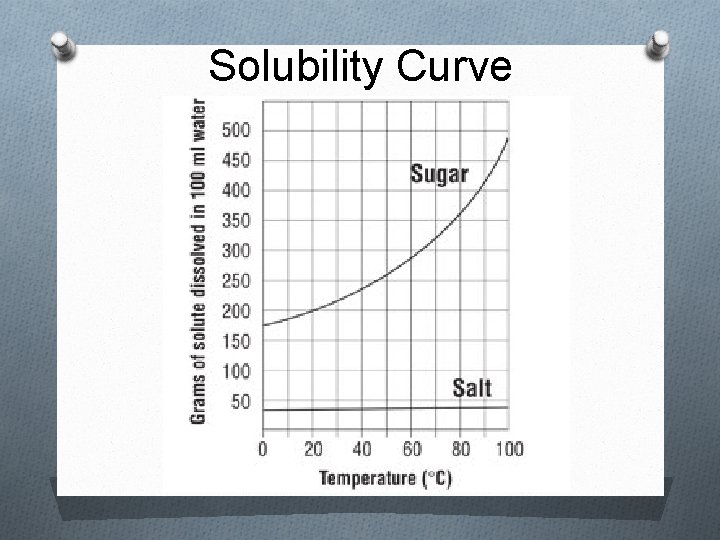
Solubility Curve
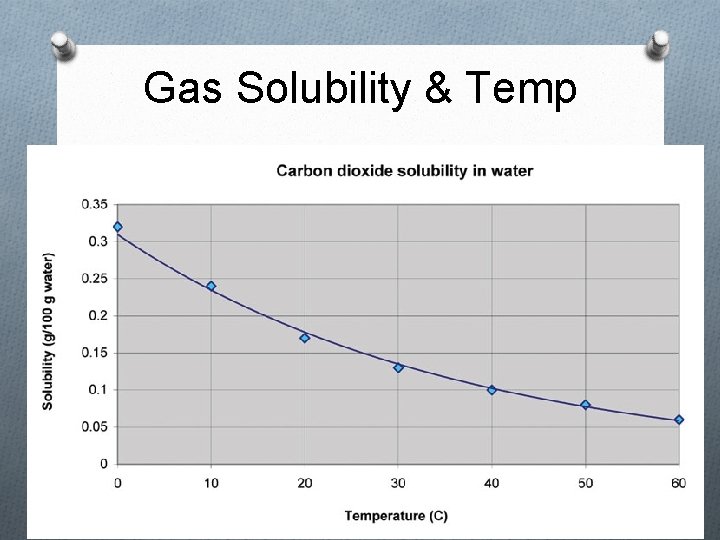
Gas Solubility & Temp
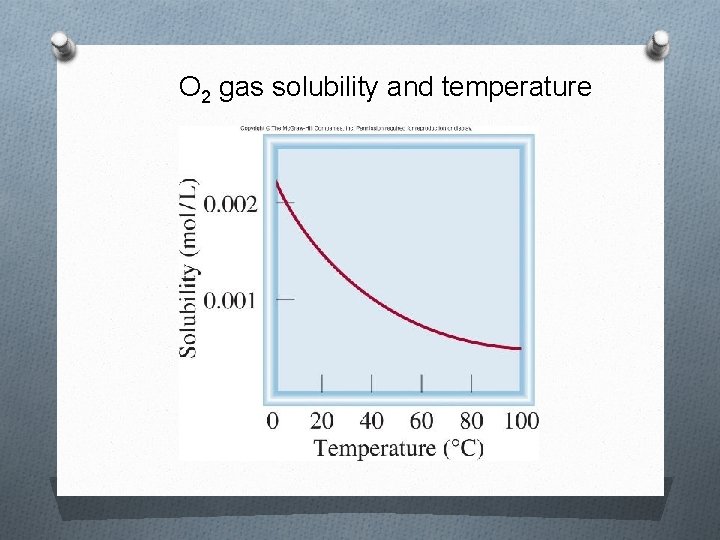
O 2 gas solubility and temperature

Example 1. If the solubility of sodium chloride at 25°C is 36. 2 g/100. g water, what is the maximum mass of sodium chloride that can be dissolved in 200. 0 g of water at the same temperature? 2. If the solubility of potassium nitrate at 20°C is 79 grams in 250 grams of water, what is the solubility in grams per liter?
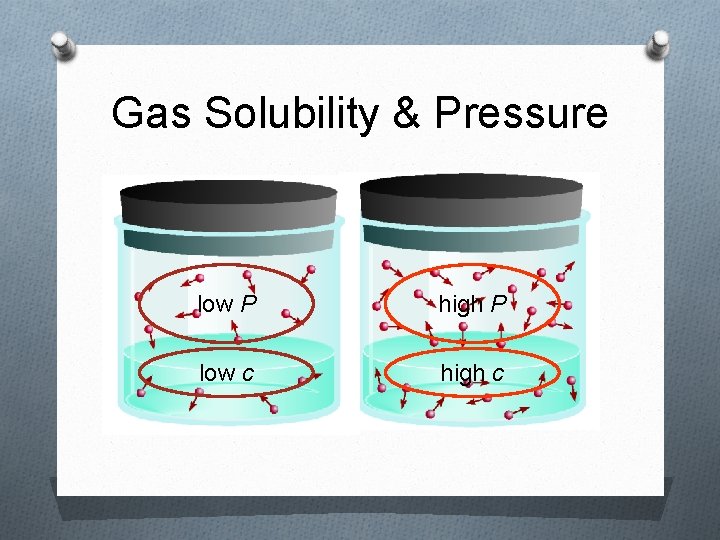
Gas Solubility & Pressure low P high P low c high c
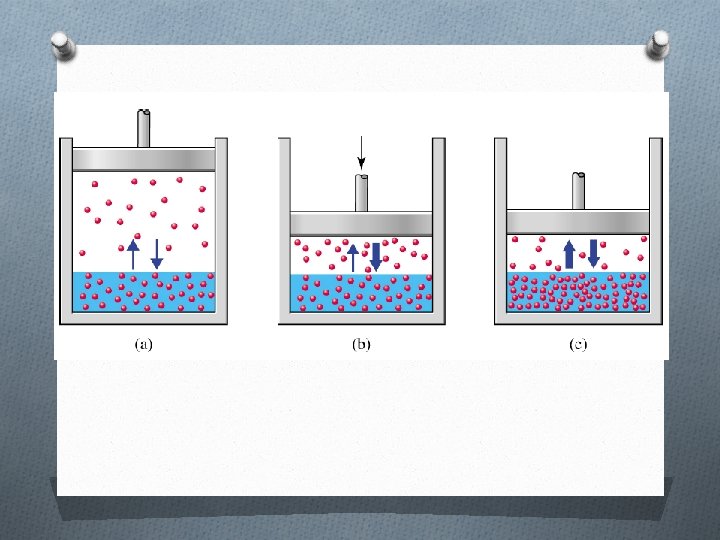

Henry’s Law The solubility of a gas in a liquid is proportional to the pressure of the gas over the solution S ∝ P S = k. P S P = k

Example The solubility of a gas is 0. 35 g/L at 25. 0 k. Pa of pressure. What is the solubility when the pressure is increased to 115 k. Pa?

Stop

Solubility Pogil Solubility of Gases-grape juice Quick Quiz
Solubility Demo-Gases Solvent Temp Hot Temp Room Temp Cold Soda 1. 2. Write your observations in each of the boxes. What can you conclude from your observations and why?

Stop

Concentration O Percent by mass (m/m) O Percent by volume (v/v) O Parts per million (ppm) O Molarity (mol/L)

Percent by Mass 1. An aquarium must contain 13. 6 g of sodium chloride per 100. g of water. What is the percent by mass of sodium chloride in the solution? 2. You have a 650. 0 g aqueous potassium chloride solution. If the percent by mass of the solute is 1. 25% how many grams of solute are in the solution? Grams of solvent?

Percent by Mass 3. Calculate the percent by mass of the solution in 5. 50 g of sodium bromide in 78. 2 g of solution.

Percent by Volume 1. What is the percent by volume of ethanol in a solution that contains 45 m. L of ethanol dissolved in 145 m. L of water? 2. If you have 200. 0 m. L of a 35. 0% aqueous solution of ethanol, what volume of ethanol and water are in the solution?

Parts per million 1. 150 m. L of an aqueous sodium chloride solution contains 0. 0045 g of sodium chloride. Calculate the concentration of sodium chloride in parts per million. 2. If 25 grams of a chemical is dissolved in 75 grams of water, what is the concentration of the chemical in parts per million?

3. Suppose 17 grams of sucrose is dissolved in 183 grams of water. What is the concentration of sucrose in parts per million? 4. Convert 0. 0035% sodium chloride by mass into parts per million.

Stop

Molarity

1. Calculate the molarity of a 1. 75 L solution containing 3. 45 moles of calcium chloride. 2. Calculate the molarity of an 855 m. L solution containing 2. 75 moles of sodium nitrate. 3. A 100. 5 m. L IV solution contains 5. 10 g of glucose. What is the molarity of this solution?

4. How many moles of sodium nitrate are in 655 m. L of a 3. 45 M solution? Now putting %Concentration, Molarity and mass all together: 5. Calculate the percent by mass of the solute in 5. 50 g of Na. Br in 78. 2 g of solution.

6. A solution has a density of 1. 08 g/m. L. Find the % by mass of a 2. 50 M Na. Cl solution. 7. Find the molarity of a 3. 35% by mass Na. OH solution with a density of solution = 1. 04 g/m. L.
- Slides: 29