Scientific Measurements SI International System Units SI International











































































































- Slides: 110

Scientific Measurements


SI (International System) Units:

SI (International System) Units: Mass:

SI (International System) Units: Mass: the quantity of matter Unit:

SI (International System) Units: Mass: the quantity of matter Unit: gram (g) or kg (kilogram)

SI (International System) Units: Mass: the quantity of matter Weight: Unit: gram (g) or kg (kilogram)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Unit: gram (g) or kg (kilogram) Unit:

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Unit: gram (g) or kg (kilogram) Unit: Newton (N)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: Unit: gram (g) or kg (kilogram) Unit: Newton (N)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit:

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit:

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Temperature: Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Temperature: average kinetic energy Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec) Unit:

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Temperature: average kinetic energy Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec) Unit: Kelvin (K)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Temperature: average kinetic energy Quantity: Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec) Unit: Kelvin (K)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Temperature: average kinetic energy Quantity: number of things Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec) Unit: Kelvin (K) Unit:

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Temperature: average kinetic energy Quantity: number of things Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec) Unit: Kelvin (K) Unit: mole (mol)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Temperature: average kinetic energy Quantity: number of things Volume: Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec) Unit: Kelvin (K) Unit: mole (mol)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Temperature: average kinetic energy Quantity: number of things Volume: space occupied Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec) Unit: Kelvin (K) Unit: mole (mol) Unit:

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Temperature: average kinetic energy Quantity: number of things Volume: space occupied Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec) Unit: Kelvin (K) Unit: mole (mol) Unit: liter (L) or milliliter (ml)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Temperature: average kinetic energy Quantity: number of things Volume: space occupied Density: Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec) Unit: Kelvin (K) Unit: mole (mol) Unit: liter (L) or milliliter (ml)

SI (International System) Units: Mass: the quantity of matter Weight: measure of gravity Length: distance between 2 points Time: interval between 2 occurrences Temperature: average kinetic energy Quantity: number of things Volume: space occupied Density: ratio of mass to volume Unit: gram (g) or kg (kilogram) Unit: Newton (N) Unit: meter (m) Unit: second (s or sec) Unit: Kelvin (K) Unit: mole (mol) Unit: liter (L) or milliliter (ml)

SI (International System) Units: Mass: the quantity of matter Unit: gram (g) or kg (kilogram) Weight: measure of gravity Unit: Newton (N) Length: distance between 2 points Unit: meter (m) Time: interval between 2 occurrences Unit: second (s or sec) Temperature: average kinetic energy Unit: Kelvin (K) Quantity: number of things Unit: mole (mol) Volume: space occupied Unit: liter (L) or milliliter (ml) Density: ratio of mass to volume Unit: g/cm 3 or g/ml

SI (International System) Units: Mass: Unit: gram (g) or kg (kilogram) Weight: Unit: Newton (N) Length: Unit: meter (m) Time: Unit: second (s or sec) Temperature: Unit: Kelvin (K) Quantity: Unit: mole (mol) Volume: Unit: liter (L) or milliliter (ml) Density: Unit: g/cm 3 or g/ml



Metric Prefixes

Metric Prefixes 1 m = length from nose to finger tip

Metric Prefixes 1 m = length from nose to finger tip Kilo =

Metric Prefixes 1 m = length from nose to finger tip Kilo = 1000 x

Metric Prefixes 1 m = length from nose to finger tip Kilo = 1000 x 1 km = distance to Schnucks

Metric Prefixes 1 m = length from nose to finger tip Kilo = 1000 x Deci = 1 km = distance to Schnucks

Metric Prefixes 1 m = length from nose to finger tip Kilo = 1000 x Deci = 1/10 th or 0. 1 x 1 km = distance to Schnucks

Metric Prefixes 1 m = length from nose to finger tip Kilo = 1000 x Deci = 1/10 th or 0. 1 x 1 km = distance to Schnucks 1 dm = width of your hand

Metric Prefixes 1 m = length from nose to finger tip Kilo = 1000 x Deci = 1/10 th or 0. 1 x Centi = 1 km = distance to Schnucks 1 dm = width of your hand

Metric Prefixes 1 m = length from nose to finger tip Kilo = 1000 x Deci = 1/10 th or 0. 1 x Centi = 1/100 th or 0. 01 x 1 km = distance to Schnucks 1 dm = width of your hand

Metric Prefixes 1 m = length from nose to finger tip Kilo = 1000 x Deci = 1/10 th or 0. 1 x Centi = 1/100 th or 0. 01 x 1 km = distance to Schnucks 1 dm = width of your hand 1 cm = width of little finger

Metric Prefixes 1 m = length from nose to finger tip Kilo = 1000 x Deci = 1/10 th or 0. 1 x Centi = 1/100 th or 0. 01 x Milli = 1 km = distance to Schnucks 1 dm = width of your hand 1 cm = width of little finger

Metric Prefixes 1 m = length from nose to finger tip Kilo = 1000 x Deci = 1/10 th or 0. 1 x Centi = 1/100 th or 0. 01 x Milli = 1/1000 th or 0. 001 x 1 km = distance to Schnucks 1 dm = width of your hand 1 cm = width of little finger

Metric Prefixes 1 m = length from nose to finger tip Kilo = 1000 x Deci = 1/10 th or 0. 1 x Centi = 1/100 th or 0. 01 x Milli = 1/1000 th or 0. 001 x 1 km = distance to Schnucks 1 dm = width of your hand 1 cm = width of little finger 1 mm = width of finger nail

Powers of Ten™ (1977). mp 4

millimeter liter milliliter gram kilogram milligram


Definition: temperature is a measure of the average kinetic energy of a substance.




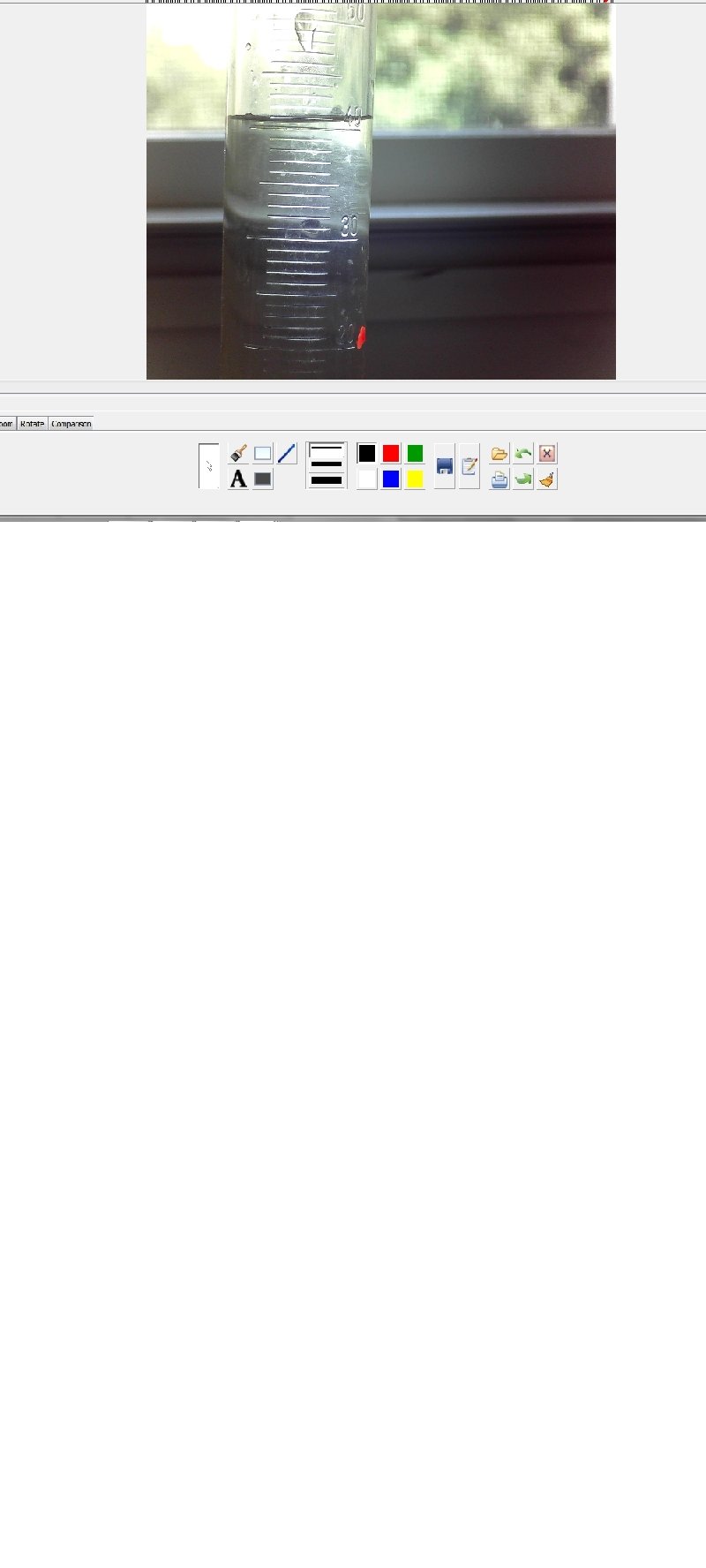
How precise is your meter stick? Determine the length, estimating between the smallest tic marks. Length with meter stick #1: Length with meter stick #2: Length with meter stick #3: Length with meter stick #4: Which is most precise? Why? Is that measurement necessarily accurate?







Last










Last






Last







Last

Mr. Wieland's dog, Tempo, weighs 29. 5 lbs. He brings Tempo to class and five students weigh him, resulting in the following measurements: 25. 8 lb, 25. 6 lb, 25. 9 lb, 25. 6 lb, and 25. 7 lb.

Mr. Wieland's dog, Tempo, weighs 29. 5 lbs. He brings Tempo to class and five students weigh him, resulting in the following measurements: 25. 8 lb, 25. 6 lb, 25. 9 lb, 25. 6 lb, and 25. 7 lb. Are these measurements accurate?

Mr. Wieland's dog, Tempo, weighs 29. 5 lbs. He brings Tempo to class and five students weigh him, resulting in the following measurements: 25. 8 lb, 25. 6 lb, 25. 9 lb, 25. 6 lb, and 25. 7 lb. Are these measurements accurate? Are these measurements precise?

Mr. Wieland's dog, Tempo, weighs 29. 5 lbs. He brings Tempo to class and five students weigh him, resulting in the following measurements: 25. 8 lb, 25. 6 lb, 25. 9 lb, 25. 6 lb, and 25. 7 lb. Are these measurements accurate? Are these measurements precise? What could you conclude about the scale?

Mr. Wieland's dog, Tempo, weighs 29. 5 lbs. He brings Tempo to class and five students weigh him, resulting in the following measurements: 25. 8 lb, 25. 6 lb, 25. 9 lb, 25. 6 lb, and 25. 7 lb. Are these measurements accurate? Are these measurements precise? What could you conclude about the scale? What can you conclude about the students' abilities to read a scale?

Mr. Wieland's dog, Tempo, weighs 29. 5 lbs. Five other students weighed the pup on a different scale, resulting in the following measurements: 32. 5 lb, 26. 5 lb, 25. 9 lb, 33. 0 lb, and 25. 8 lb.

Mr. Wieland's dog, Tempo, weighs 29. 5 lbs. Five other students weighed the pup on a different scale, resulting in the following measurements: 32. 5 lb, 26. 5 lb, 25. 9 lb, 33. 0 lb, and 25. 8 lb. Are these measurements accurate?

Mr. Wieland's dog, Tempo, weighs 29. 5 lbs. Five other students weighed the pup on a different scale, resulting in the following measurements: 32. 5 lb, 26. 5 lb, 25. 9 lb, 33. 0 lb, and 25. 8 lb. Are these measurements accurate? Are these measurements precise?

Mr. Wieland's dog, Tempo, weighs 29. 5 lbs. Five other students weighed the pup on a different scale, resulting in the following measurements: 32. 5 lb, 26. 5 lb, 25. 9 lb, 33. 0 lb, and 25. 8 lb. Are these measurements accurate? Are these measurements precise? What can you conclude about the scale?

Mr. Wieland's dog, Tempo, weighs 29. 5 lbs. Five other students weighed the pup on a different scale, resulting in the following measurements: 32. 5 lb, 26. 5 lb, 25. 9 lb, 33. 0 lb, and 25. 8 lb. Are these measurements accurate? Are these measurements precise? What can you conclude about the scale? What can you conclude about the students' abilities to read a scale?

Mini Quiz If the correct length of a pencil is 13. 0 cm and students measure as 12. 4 cm, 12. 5 cm, and 12. 6 cm, were they accurate, precise, both or neither? Are precise measurements always accurate? Are accurate measurements always precise?




100 200 300 400 500 600 700 800 900 10 20 30 40 50 60 70 80 90 2 3 4 5 6 7 8 9 1 . .



Density definition: Amount of mass (in grams - g) per a unit of volume (in liters - L - or milliliters - ml). Formula: Density = mass/volume Density unit: g/ml

Density How do you determine mass? How do you determine volume of something odd shaped?

Density is a "fingerprint property, " meaning it can be used to identify substances. Each substance has a characteristic density.

Density Suppose a king wanted you to make sure his crown were pure gold. How would you do it?

Density volume of crown


Mass vs Weight I weigh 180 lbs (800 N) Earth Moon I have a mass of 81 kg Earth Moon

Mass vs Weight I weigh 180 lbs (800 N) I weigh 30 lbs (133 N) Earth Moon I have a mass of 81 kg Earth Moon

Mass vs Weight Balance Scales


Attachments Powers of Ten™ (1977). mp 4