Scientific Measurements Units of Measurements 2 Conversion Problems

Scientific Measurements Units of Measurements 2. Conversion Problems 3. Density Measurements and their Uncertainty 1. 4.

Do any of these signs list a measurement? - Measurements contain BOTH a number and a unit.

1. Measuring with SI Units � All measurements depend on units that serve as reference standards. The standards of measurement used in science are those of the metric system. � Based on multiples of 10 � The International System of Units (abbreviated SI, after the French name, Le Système International d’Unités) is a revised version of the metric system.

do the SI prefixes deci, centi, and milli mean? � Think about the word decimal, century, and millennium. � What


The five SI base units commonly used by chemists are the meter, the kilogram, the kelvin, the second, and the mole.

Units & Quantities What metric units are commonly used to measure length, volume, mass, temperature and energy?

Units of Length � In SI, the basic unit of length, or linear measure, is the meter (m). � For very large or and very small lengths, it may be more convenient to use a unit of length that has a prefix.

Units of Volume - space occupied by any sample of matter How do you calculate the volume of a cube? The SI unit of volume is the amount of space occupied by a cube that is 1 m along each edge. This volume is the cubic meter (m)3

Units of Volume “I’ll have a liter of cola” What unit of volume are you most familiar with? � The liter is a NON SI unit of measurement! � A liter (L) is the volume of a cube that is 10 centimeters (10 cm) along each edge (10 cm = 1000 cm 3 = 1 L).

Units of Volume � Common quantities The volume of 20 drops of liquid from a medicine dropper is approximately 1 m. L. A sugar cube has a volume of 1 cm 3. 1 m. L is the same as 1 cm 3.

Unit of Mass � The mass of an object is measured in comparison to a standard mass of 1 kilogram (kg), which is the basic SI unit of mass. Common metric units of mass include kilogram, milligram, and microgram.

Unit of Mass � How does weight differ from mass? Weight is a force that measures the pull on a given mass by gravity. Mass is a measure of the quantity of matter. (the space it occupies)

Units of Temperature - is a measure of how hot or cold an object is. Thermometers are used to measure temperature. • Substances expand with an increase in temperature.
Heat of Transfer When 2 objects at different temperatures are in contact, heat travels from: Lower temperature Higher temperature
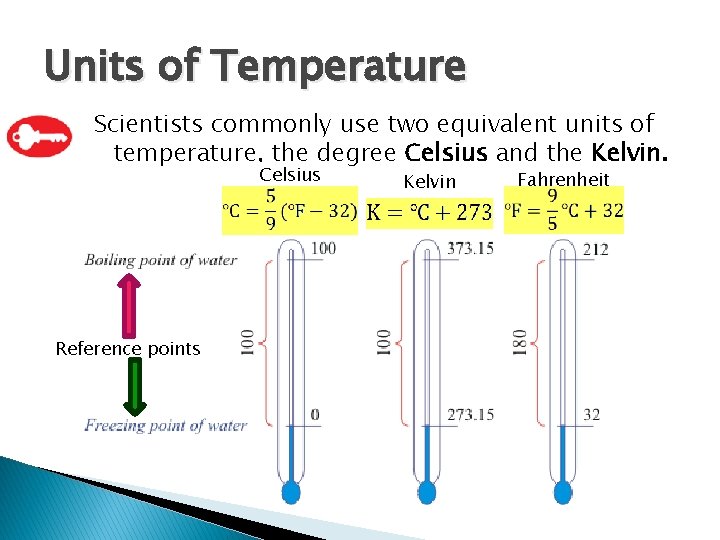
Units of Temperature Scientists commonly use two equivalent units of temperature, the degree Celsius and the Kelvin. Celsius Reference points Kelvin Fahrenheit

Units of Temperature � The zero point on the Kelvin scale, 0 K, or absolute zero, is equal to 273. 15 °C.

Units of Temperature � Because one degree on the Celsius scale is equivalent to one Kelvin on the Kelvin scale, converting from one temperature to another is easy. You simply add or subtract 273, as shown in the following equations.

Practice – Converting Between Temperature Scales Normal human body temperature is 37 C. What is that temperatures in kelvins? Analyze: List the known and the unknown. Known ◦ Temperature in C = 37 C Unknown ◦ Temperature in K = ? K Equation: K= C + 273 Calculate: Solve for the unknown. K= C + 273 37 + 273 = 310 K

Practice – Converting Between Temperature Scales Liquid nitrogen boils at 77. 2 K. What is this temperature in degrees Celsius? Analyze: List the known and the unknown. Calculate: Solve for the unknown. -196 C

Units of Energy 1 J = 0. 2390 cal 1 cal= 4. 184 J Energy - the capacity to do work or to produce heat. The joule and the calorie are common units of energy. � Joule (J) is the SI unit of energy. � 1 calorie (cal) is the quantity of heat that raises the temperature of 1 g of pure H 2 O by 1 C.

Were you paying attention? 1. Which of the following is not a base SI unit? A. meter B. gram C. second D. mole �

Were you paying attention? 2. If you measured both the mass and weight of an object on Earth and on the moon, you would find that A. both the mass and the weight do not change. B. both the mass and the weight change. C. the mass remains the same, but the weight changes. D. the mass changes, but the weight remains the same.

Were you paying attention? 3. A temperature of 30 degrees Celsius is equivalent to A. 303 K. B. 300 K. C. 243 K. D. 247 K. K = C + 273

� Can you think of any other examples in which quantities can be expressed in several different ways?

� Consider the conversion units of distance: 1 meter = 10 decimeters = 100 centimeters = 1000 millimeters When 2 measurements are equivalent, a ratio of the 2 measurements equals 1 or Conversion factor Remember: even though the numbers in the measurements 1 m and 100 cm differ, both measurements represent the same length

2. Conversion Problems What happens when a measurement is multiplied by a conversion factor? � When a measurement is multiplied by a conversion factor, the numerical value is generally changed, but the actual size of the quantity measured remains the same.

3. 3 Conversion �A Factors conversion factor is a ratio of equivalent measurements. � The ratios 100 cm/1 m and 1 m/100 cm are examples of conversion factors. Fig. 3. 11

3. 3 Conversion � The Factors scale of the micrograph is in nanometers. Using the relationship 109 nm = 1 m, you can write the following conversion factors.

Dimensional Analysis Dimensional analysis is a way to analyze and solve problems using the units, or dimensions, of the measurements. � Why � An is dimensional analysis useful? alterative way to problem solving

Example of Using Dimensional Analysis Sample Problem 3. 5 How many seconds are in a workday that lasts exactly eight hours? Analyze: List the knowns and the unknown. Known ◦ Time worked = 8 h ◦ 1 hour = 60 min ◦ 1 minute = 60 s Unknown ◦ Seconds worked = ? s Calculate: Solve for the unknown.

1. ) How many minutes are there in exactly one week? Analyze: List the knowns and the unknown. Known Unknown 1. 0080 x 104 min Calculate: Solve for the unknown. 2. ) How many seconds are in exactly a 40 -hour work week? Analyze: List the knowns and the unknown. Known Unknown Calculate: Solve for the unknown. 1. 44000 x 105 s

1. ) The directions for an experiment ask each student to measure 1. 84 g of copper (Cu) wire. The only copper wire available is a spool with a mass of 50. 0 g. How many students can do the experiment before the copper runs out? 27 students 2. ) A 1. 00 -degree increase on the Celsius scale is equivalent to a 1. 80 -degree increase on the Fahrenheit scale. If a temperature increase by 48. 0 C, what is the corresponding temperature increase on the Fahrenheit scale? 86. 4 F

Converting Between units What types of problems are easily solved by using dimensional analysis? � Problems in which a measurement with one unit is converted to an equivalent measurement with another unit are easily solved using dimensional analysis.

Converting Between Metric Units Express 750 dg in grams. Analyze: List the knowns and the unknown. Known Mass = 750 dg 1 g = 10 dg Unknown Mass = ? g Calculate: Solve for the unknown. Conversion unit

Practice Problems Convert the following. a) 15 cm 3 to liters 1. 5 x 10 -2 L b) 7. 38 g to kilograms 7. 38 x 10 -3 kg c) 6. 7 s to milliseconds 6. 7 x 103 ms d) 94. 5 g to micrograms 9. 45 x 107 g

Sports Stats ECLN Entertainment & Chemistry Learning Network Purpose: to use dimensional analysis to convert between English and metric units. Procedure: Using the player stats for the New England Patriots, convert heights and weights into heights and masses expressed in meters and kilograms, respectively. � You must document your approach: ◦ Identify the known, unknown, and conversion factor. ◦ Must show all calculations. 2. 54 cm = 1 inch 454 g = 1 lb

Multistep Problems What is 0. 073 cm in micrometers? Analyze: List the knowns and the unknown. Known Length = 0. 073 cm = 7. 3 x 10 -2 cm 102 cm = 1 m 1 m = 106 m Unknown Length = ? m Calculate: Solve for the unknown.

Practice Problems 1. The radius of a potassium atom is 0. 227 nm. Express the radius to the unit centimeters. 2. 27 x 10 -8 cm 2. The diameter of Earth is 1. 3 x 104 km. What is the diameter expressed in decimeters? 1. 3 x 108 dm

Converting Complex Units The mass per unit volume of a substance is a property called density. The density of manganese, a metallic element, is 7. 21 g/cm 3. What is the density of manganese expressed in units kg/m 3? Analyze: List the knowns and the unknown. Known Density of manganese = 7. 21 g/cm 3 103 g = 1 kg 106 cm 3 = 1 m 3 Unknown Density manganese = ? kg/m 3

Density of manganese = 7. 21 g/cm 3 103 g = 1 kg 106 cm 3 = 1 m 3 g/cm 3 kg/m 3 Calculate: Solve for the unknown. 7. 21 x 103 kg/m 3

Practice Problems 1. Gold has a density of 19. 3 g/cm 3. What is the density in kilograms per cubic meter? g/cm 3 103 g = 1 kg 106 cm 3 = 1 m 3 2. 1. 93 x 104 kg/m 3 There are 7. 0 x 106 red blood cells (RBC) in 1. 0 mm 3 of blood. How many red blood cells are in 1. 0 L of blood? mm 3 = 1 m 3 106 cm 3 = 1 m 3 1 cm 3 = 1 m. L 103 m. L = 1 L 7. 0 x 1012 RBC/L

Were you paying attention? 1 Mg = 1000 kg. Which of the following would be a correct conversion factor for this relationship? A. 1000. B. 1/1000. C. ÷ 1000. D. 1000 kg/1 Mg.

Were you paying attention? The conversion factor used to convert joules to calories changes A. the quantity of energy measured but not the numerical value of the measurement. B. neither the numerical value of the measurement nor the quantity of energy measured. C. the numerical value of the measurement but not the quantity of energy measured. D. both the numerical value of the measurement and the quantity of energy measured.

Were you paying attention? How many g are in 0. 0134 g? A. 1. 34 10– 4 B. 1. 34 10– 6 C. 1. 34 106 D. 1. 34 104

Were you paying attention? Express the density 5. 6 g/cm 3 in kg/m 3. A. 5. 6 106 kg/m 3 B. 5. 6 103 kg/m 3 C. 0. 56 kg/m 3 D. 0. 0056 kg/m 3

Which is heavier, a pound of lead or a pound of feathers?

� Why can boats made of steel float on water when a bar of steel sinks?

3. Determining Density What determines the density of a substance? Density is the ratio of the mass of an object to its volume.

Density � Each of these 10 -g samples has a different volume because the densities vary. Which substance has the highest ratio of mass to volume?

Date: 10/11/2012 Bell Ringer 1. 2. 3. Define what an intensive property is. Provide 3 examples of an intensive property. Provide 2 examples of an extensive property.
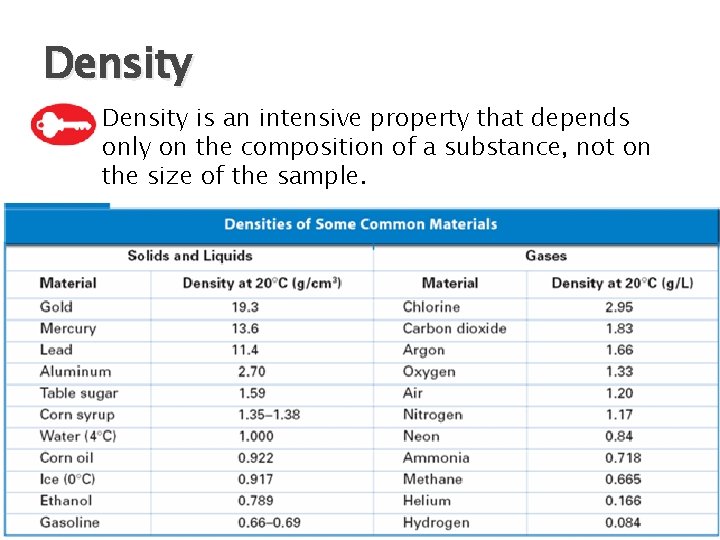
Density � Density is an intensive property that depends only on the composition of a substance, not on the size of the sample.

For example, a 10. 0 -cm 3 piece of lead has a mass of 114 g. What is the density of lead? Unit of density: g/cm 3

� What would happen if corn oil is poured into a glass containing corn syrup? OIL SYRUP

Determining Density � Simulation 1 ◦ Rank materials according to their densities. Chem. ASAP/dswmedia/rsc/asap 1_chem 05_cmsm 0301. html

Density and Temperature � Volume as temperature � The mass remains the same despite the temperature and volume changes. � Recall: � If volume changes with temperature and mass stays the same, then density must change.
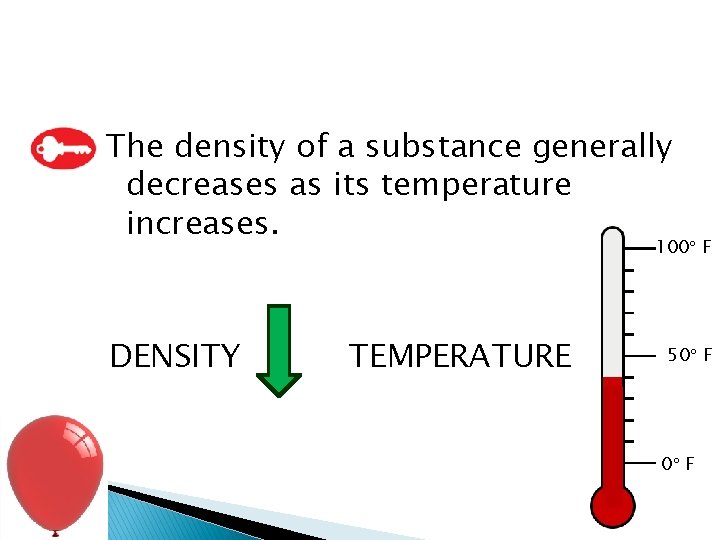
The density of a substance generally decreases as its temperature increases. 100 F DENSITY TEMPERATURE 50 F

Sample Problem A copper penny has a mass of 3. 1 g and a volume of 0. 35 cm 3. What is the density of copper? Analyze: List the knowns and the unknown. Known Mass = 3. 1 g Volume = 0. 35 cm 3 Unknown Density = ? g/cm 3 8. 8571 g/cm 3

Practice Problems 1. A student finds a shiny piece of metal that she thinks is aluminum. In the lab, she determines that the metal has a volume of 245 cm 3 and a mass of 612 g. Calculate the density. Is the metal aluminum? 2. 50 g/cm 3 - NO 2. A bar of silver has a mass of 68. 0 g and a volume 6. 48 cm 3. What is the density of silver? 10. 5 g/cm 3

Practice Problem Using Density to Calculate Volume What is the volume of a pure silver coin that has a mass of 14 g? The density of silver (Ag) is 10. 5 g/cm 3. Analyze: List the knowns and the unknown. Known Mass of coin = 14 g Density of silver = 10. 5 g/cm 3 Unknown volume of coin= ? cm 3 1. 3 cm 3 of Ag

Practice Problems 1. Use dimensional analysis and the given densities to make the following conversions. a. 14. 8 g of boron to cm 3 of boron. The density of boron is 2. 34 g/cm 3. Volume of B = 6. 32 cm 3 b. 4. 62 g of mercury to cm 3 of mercury. The density of mercury is 13. 5 g/cm 3. Volume of Hg = 0. 342 cm 3 2. Rework the preceding problems by applying the following equation.

Make the following conversions: 1. 2. 53 cm 3 of gold to grams (density of gold = 19. 3 g/cm 3) 48. 8 g 2. 1. 6 g of oxygen to liters (density of oxygen = 1. 33 g/L) 1. 2 L 3. 25. 0 g of ice to cubic centimeters (density of ice = 0. 917 g/cm 3) 27. 3 cm 3

Were You Paying Attention? If 50. 0 m. L of corn syrup have a mass of 68. 7 g, the density of the corn syrup is a. 0. 737 g/m. L. b. 0. 727 g/m. L. c. 1. 36 g/m. L. d. 1. 37 g/m. L.

Were You Paying Attention? What is the volume of a pure gold coin that has a mass of 38. 6 g? The density of gold is 19. 3 g/cm 3. a. 0. 500 cm 3 b. 2. 00 cm 3 c. 38. 6 cm 3 d. 745 cm 3

Were You Paying Attention? As the temperature increases, the density of most substances a. increases. b. decreases. c. remains the same. d. increases at first and then decreases.

� Today’s � The current weather in Eastville is Weather Channel projected forcast: � Eastern Shore News

� What everyday activities involve measuring? � Recall which units of measure are related to each of the examples you provide. � Estimate how tall you are in inches. � Have your lab partner estimate how tall you are with a yard stick. � Compare the estimates with the yard stick value.

4. Measurements and Their Uncertainty A measurement is a quantity that has both a number and a unit. Ex. ØHeight Ø Cooking Ø Speed ØWeight Measurements are fundamental to the experimental sciences. For that reason, it is important to be able to make measurements and to decide whether a measurement is correct.

Using and Expressing Measurements In scientific notation, a given number is written as the product of two numbers: a coefficient and 10 raised to a power. Example: 602, 000, 000, 000 Scientific notation = 6. 02 x 1023

3. What 1 is the difference between accuracy & precision?
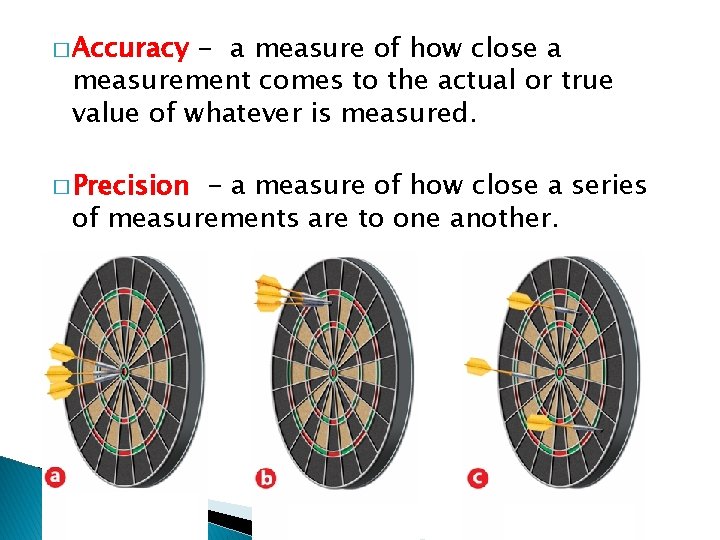
� Accuracy - a measure of how close a measurement comes to the actual or true value of whatever is measured. � Precision - a measure of how close a series of measurements are to one another.

� To evaluate the accuracy of a measurement, the measured value must be compared to the correct value. � To evaluate the precision of a measurement, you must compare the values of two or more repeated measurements.

3. 1 Accuracy, Precision, and Error Determining Error Suppose you are using a thermometer to measure the boiling point of pure H 2 O at STP. The thermometer reads 99. 1 C. The true, or accepted value of the boiling point of pure H 2 O under these conditions (STP) is actually 100 C. �Which is the accepted value? �Which is the experimental value? �What is the error?

� The accepted value is the correct value based on reliable references. � The experimental value is the value measured in the lab. � The difference between the experimental value and the accepted value is called the error. • Error can be positive or negative depending on whether the experimental value is greater than or less than the accepted value.

The percent error is the absolute value of the error divided by the accepted value, multiplied by 100%.

Suppose you are using a thermometer to measure the boiling point of pure H 2 O at STP. The thermometer reads 99. 1 C. The true, or accepted value of the boiling point of pure H 2 O under these conditions (STP) is actually 100 C.

� Just because a measuring device works, you cannot assume it is accurate. The scale below has not been properly zeroed, so the reading obtained for the person’s weight is inaccurate.

Significant Figures in Measurements Suppose you estimate a weight that is between 2. 4 lb and 2. 5 lb to be 2. 46 lb. The first two digits (2 and 4) are known. The last digit (6) is an estimate and involves some uncertainty. All three digits convey useful information, however, and are called significant figures. The significant figures in a measurement include all of the digits that are known, plus a last digit that is estimated.

Measurements MUST ALWAYS be reported to the correct number of significant figures because calculated answers often depend on the number of significant figures in the values used in the calculation. Animation See how the precision of a calculated result depends on the sensitivity of the measuring instruments.

3. Significant Figures in Measurements 1

Counting Significant Figures in Measurements How many significant figures are in each measurement? 1. 123 m 2. 40. 506 mm 3. 9. 8000 x 104 m 5 sig figs (rule 4) 4. 22 meter sticks Unlimited (rule 6) 5. 0. 07080 m 6. 98, 000 m 3 sig figs (rule 1) 5 sig figs (rule 2) 4 sig figs (rule 2, 3, 4 ) 2 sig figs (rule 5)

1. How many significant figures are in each length? a. 0. 05730 meters 4 sig figs b. 8765 meters 4 sig figs c. 0. 00073 meters 2 sig figs d. 40. 007 meters 5 sig figs 2. How many significant figures are in each measurement? a. 143 grams 3 sig figs b. 0. 074 meter 2 sig figs c. 8. 750 x 10 -2 gram 4 sig figs d. 1. 072 meter 4 sig figs

Significant Figures in Calculations � How does the precision of a calculated answer compare to the precision of the measurements used to obtain it?

3. 1 Significant Figures in Calculations In general, a calculated answer cannot be more precise than the least precise measurement from which it was calculated. � The calculated value must be rounded to make it consistent with the measurements from which it was calculated.

Rounding To round a number, you must first decide how many significant figures your answer should have. The answer depends on the given measurements and on the mathematical process used to arrive at the answer. Ø If the digit immediately to the right of the last significant digit is less than 5, it is simply dropped and the value of the last significant digit stays the same. 2. 691 m 2. 69 m Ø If the digit in question is 5 or greater, the value of the digit in the last significant place is increased by 1 294. 8 m. L 295 m. L

Sig. Fig. ’s - Rounding Measurements Round off each measurement to the number of significant figures shown in parentheses. Write the answer in scientific notation. 1. 314. 721 meters (4) 2. 0. 001775 meter (2) 3. 8792 meters (2)

Practice Problems Round each measurement to three significant figures. Write your answers in scientific notation. 8. 71 x 101 m 1. 87. 073 meters 2. 4. 3621 x 108 meters 3. 0. 01552 meter 4. 9009 meters 5. 1. 7777 x 10 -3 meter 6. 629. 55 meters 4. 36 x 108 m 1. 55 x 10 -2 m 9. 01 x 103 m 1. 78 x 10 -3 m 6. 30 x 102 m

Sig. Fig. ’s - Addition and Subtraction � The answer to an addition or subtraction calculation should be rounded to the same number of decimal places (not digits) as the measurement with the least number of decimal places. � Example: 12. 52 meters + 349. 0 meters + 8. 24 meters Step 1 – align the decimal points and identify least # of decimal places. 12. 52 meters - 2 decimal places 349. 0 meters - 1 decimal places + 8. 24 meters - 2 decimal places 369. 76 meters 369. 8 meters or 3. 698 x 102 meters

Practice Perform each operation. Express your answers to the correct number of significant figures. 1. 61. 2 meters + 9. 35 meters + 8. 6 meters 79. 2 m 2. 9. 44 meters – 2. 11 meters 7. 33 m 3. 1. 36 meters + 10. 17 meters 11. 53 m 4. 34. 61 meters – 17. 3 meters 17. 3 m

Sig. Fig. ’s - Multiplication and Division � In calculations involving multiplication and division, you need to round the answer to the same number of significant figures as the measurement with the least number of significant figures. � The position of the decimal point has nothing to do with the rounding process when multiplying and dividing measurements.

Practice Step 1 – determine # of sig figs Step 2 – calculate answer Step 3 – determine answer w/ correct # of sig figs. Perform the following operations. Give the answers to the correct number of significant figures. 1. 2. (2) (3) 7. 55 meters x 0. 34 meter = 2. 576 (meter)2 2. 6 m 2 (3) (2) 2. 10 meters x 0. 70 meter = 1. 47 (meter)2 1. 5 m 2 3. (5) (2) 2. 4526 meters ÷ 8. 4 = 0. 291976 meter 0. 29 m

Were You Paying Attention? 1. In which of the following expressions is the number on the left NOT equal to the number on the right? a. 0. 00456 10– 8 = 4. 56 10– 11 b. 454 10– 8 = 4. 54 10– 6 c. 842. 6 104 = 8. 426 106 d. 0. 00452 106 = 4. 52 109

Were You Paying Attention? 2. Which set of measurements of a 2. 00 -g standard is the most precise? a. 2. 00 g, 2. 01 g, 1. 98 g b. 2. 10 g, 2. 00 g, 2. 20 g c. 2. 02 g, 2. 03 g, 2. 04 g d. 1. 50 g, 2. 00 g, 2. 50 g

Were You Paying Attention? 3. A student reports the volume of a liquid as 0. 0130 L. How many significant figures are in this measurement? a. 2 b. 3 c. 4 d. 5
- Slides: 95