Minerals A mineral is Naturally formed Inorganic solid
















































- Slides: 53

Minerals

A mineral is Naturally formed Inorganic solid Crystalline structure Same chemical compound

Elements Are pure substances that cannot be broken down

Each element Is made up of one kind of atom

A compound Is a substance made up of two or more elements that have been joined or bonded together

Crystals Solid, geometric forms of minerals produced by a repeating pattern of atoms throughout the mineral.

Crystal Silver Halite Gold

Types of Minerals 1. Silicate minerals. Contains silicon and oxygen ( two most common elements in Earth’s crust)

Silicate minerals Feldspar-make up about half the earth’s crust Biotite mica- soft and shiny, break easy into sheets Quartz- Si. O 2

2. Nonsilicate minerals Does not contain silicon and oxygen

Native elements 20 minerals Gold, platinum diamond, copper sulfur, silver

Carbonates Combinations of carbon and oxygen Calcite- Ca. CO 3

Halides Elements combine: F, Cl, I, Br forms with Na, K, Ca Fluorite

Oxides Aluminum or iron combines with oxygen

Sulfates Contains sulfur and oxygen SO 4 Gypsum

Sulfides One or more element combines with sulfur Galena Pb. S

Science log What are the four characteristic of a mineral? What three minerals are in a granite rock?

A mineral is Naturally formed Inorganic solid Crystalline structure Same chemical compound

Quartz Mica Granite Feldspar

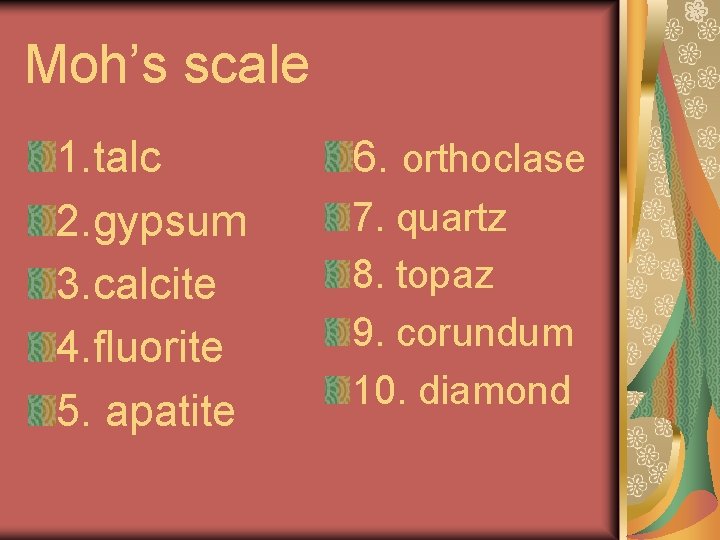
Moh’s scale 1. talc 2. gypsum 3. calcite 4. fluorite 5. apatite 6. orthoclase 7. quartz 8. topaz 9. corundum 10. diamond

1. Talc

2. Gypsum

3. Calcite

4. Fluorite Comes in white, green and purple

5. Apatite

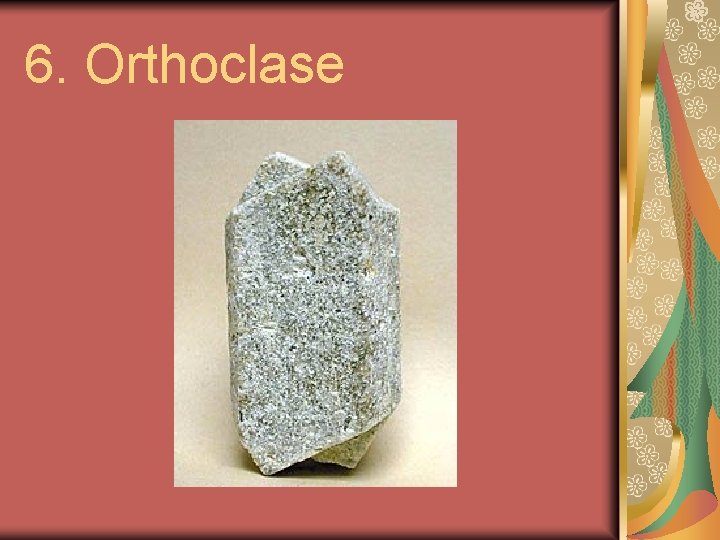
6. Orthoclase

7. Quartz

8. Topaz

9. Corundum

10 Diamond

Section 2 Identifying Minerals

Identifying Minerals Color- not always reliable Luster-surface reflects light -( shiny) Metallic luster, nonmetallic luster, submetallic luster.

Streak Color of the mineral in a powdered form.

Cleavage Minerals break along flat surfaces.

Fracture Minerals break unevenly along curved or irregular surfaces.

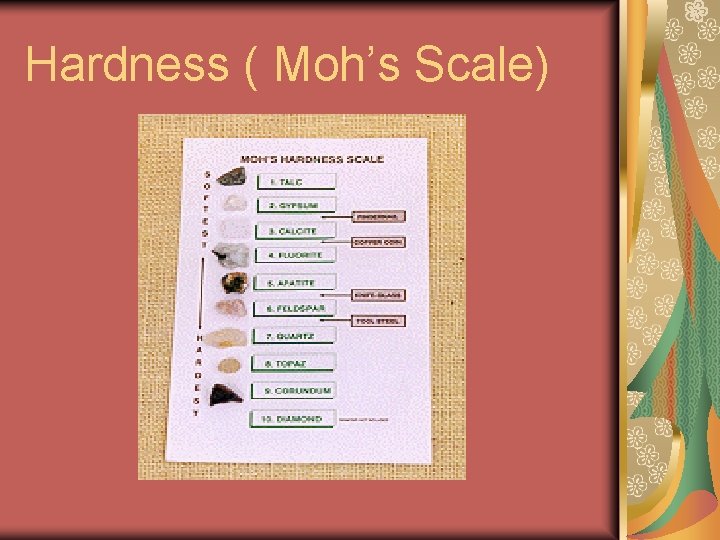
Hardness ( Moh’s Scale)

Density Measure of how much matter there is in a given amount of space.

Special properties Fluorescence Chemical reaction Optical properties Taste Magnetism Radioactivity- Geiger Counter

Science Log With your partner, finish page 343. You do not have to do #5 Start as soon as you come into class

Science Log List the ten minerals from least to most hardest in the Moh’s Scale. List the 7 ways to identify minerals. List the 6 special properties of minerals.

Science Log Jan 17, 08 Use your memory to put the minerals on the board in order of the Moh’s scale from 1 -10. Do not look at book or notes!!!! Use a new page in science log!!

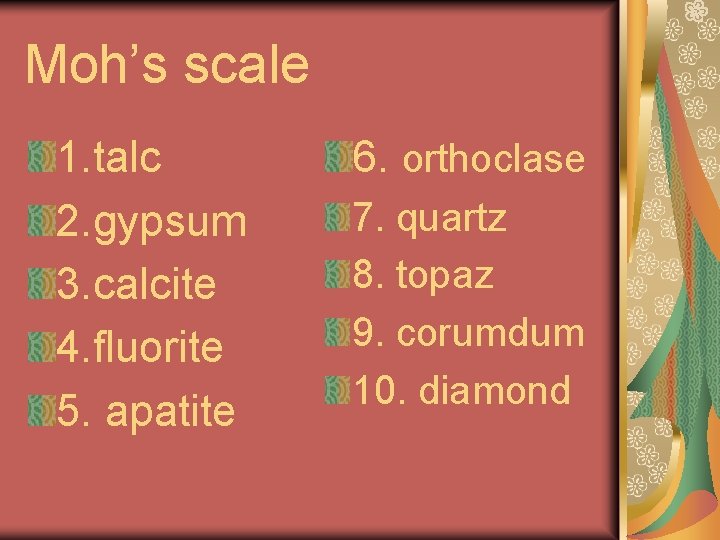
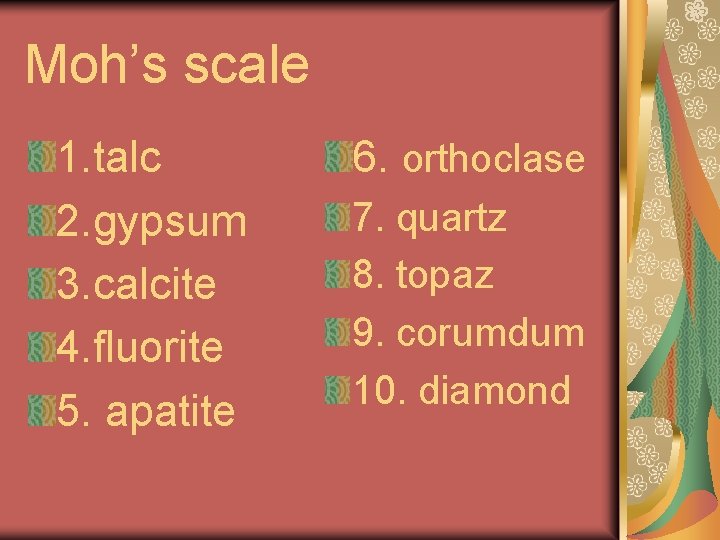
Moh’s scale 1. talc 2. gypsum 3. calcite 4. fluorite 5. apatite 6. orthoclase 7. quartz 8. topaz 9. corumdum 10. diamond

Name 7 ways to Identifying minerals Color Luster Streak Cleavage Fracture Hardness Density

Name 6 Special properties of minerals. Fluorescence Radioactivity Chemical reaction Optical properties Magnetism Taste

Section 3 Formation and mining of minerals.

Formation and mining Evaporation and Saltwater(gypsum and halite Limestone-Surface water and groundwater ( calcite and dolomite

Hot water solutions( gold, copper, sulfur, pyrite and galena Pegmatites- hot magma forms tear drop shape forming large crystals ( topaz, tourmaline)

Plutons- magma cools slowly forming millions of crystals. ( Mica, feldspar, magnetite, quartz

Metamorphic rockpressure and temperature, minerals form in rock. ( calcite, garnet, hematite and talc

Mining ore Mineral deposits large enough to mine Surface mining and deep mines

Reclamation Returning land to its original state after mining is complete.

Science Log 1 -18 -2008 Name five minerals that you have learned about.

Moh’s scale 1. talc 2. gypsum 3. calcite 4. fluorite 5. apatite 6. orthoclase 7. quartz 8. topaz 9. corumdum 10. diamond