Mineral a naturally occurring inorganic solid with a
































- Slides: 37

Mineral a naturally occurring inorganic solid with a distinct chemical composition and crystalline structure

Formation Magma Pressure Evaporation

Magma When minerals form out of molten rock, or magma. As the magma cools, the atoms move closer together and form chemical bonds, creating compounds. Many different minerals may form from one bed of magma.

Pressure When a rock is subjected to high temperature and pressure, the minerals in the rock break down chemically WITHOUT melting, creating new minerals.

Evaporation When minerals form as water containing dissolved ions slowly evaporates. For example, halite forms when water evaporates from a solution of salt and water.

Characteristics The characteristics of a mineral can help you figure out which mineral a sample is. For example: Cleavage/Fracture Luster Hardness Color Crystal structure Streak Specific Gravity Other properties

Crystal Structure Cubic 3 equal-length axes of symmetry l All 90° angles l Halite / Pyrite l

Crystal Structure Tetragonal 3 axes of symmetry: 2 same length, one different l All 90 ° angles l Zircon l

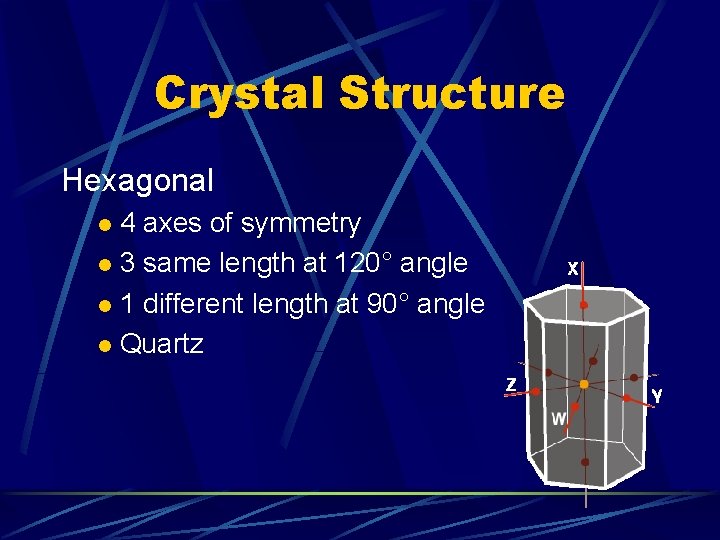
Crystal Structure Hexagonal 4 axes of symmetry l 3 same length at 120° angle l 1 different length at 90° angle l Quartz l

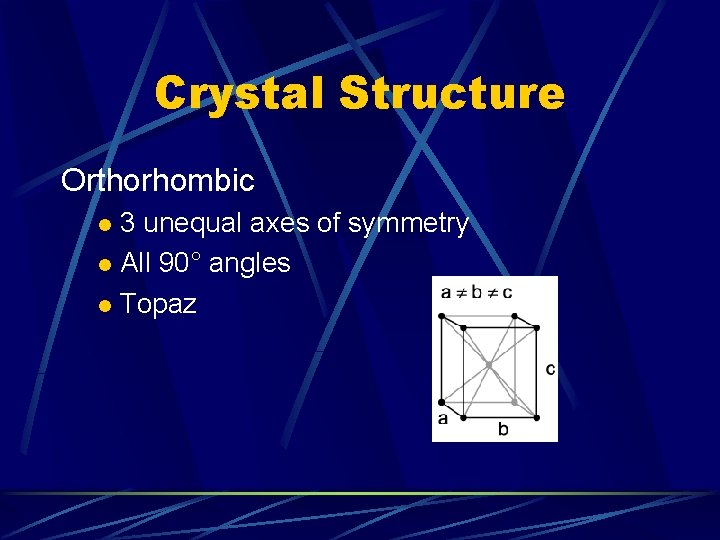
Crystal Structure Orthorhombic 3 unequal axes of symmetry l All 90° angles l Topaz l

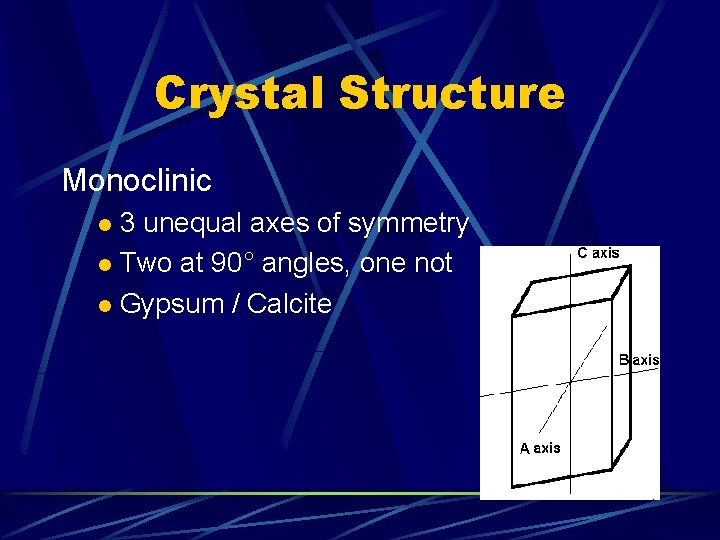
Crystal Structure Monoclinic 3 unequal axes of symmetry l Two at 90° angles, one not l Gypsum / Calcite l

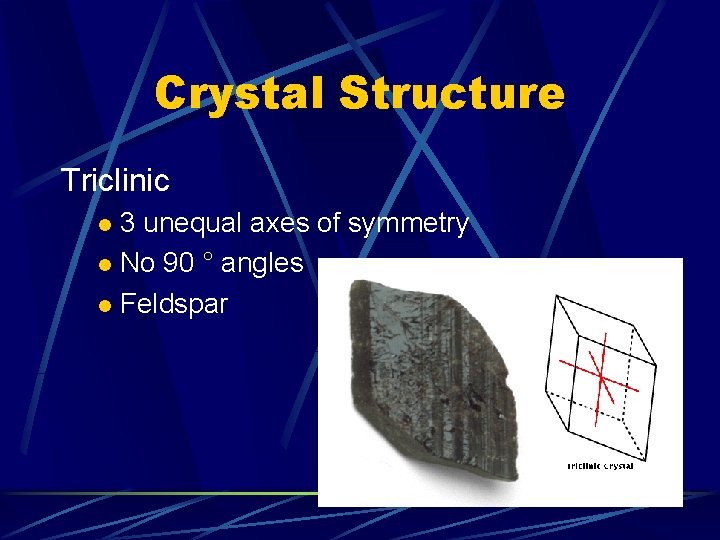
Crystal Structure Triclinic 3 unequal axes of symmetry l No 90 ° angles l Feldspar l

Cleavage the tendency of a mineral to split along planes of its crystalline structure where bonds are weakest; some minerals break only in one direction, others break in two or more directions

Cleavage Cubic: form cubes Pyrite Halite

Cleavage Rhombohedral: form six-sided prisms Calcite

Cleavage Basal: occur along a single plane parallel to the base (flat layers) Mica

Fracture the property of a mineral that describes an irregular pattern of breakage in a direction other than along cleavage planes; i. e. when a mineral does not break along flat planes

Fracture Conchoidal Fracture: smooth, curved fracture like the inside of a clam shell Obsidian Quartz

Fracture Fibrous/Splintery Fracture: looks like splinters Asbestos

Luster The property of a mineral that describes the appearance of light reflected from its surface Metallic (looks like a metal) Pyrite Hematite

Luster Vitreous (glassy) Emerald Quartz

Luster Adamantine (brilliant) Diamond

Luster Resinous (like resin or sap from a tree) Sphalerite

Luster Greasy or waxy Turquoise

Luster Pearly Talc

Luster Silky Asbestos

Luster Dull or earthy Limonite Bauxite

Color the least important property of a mineral, since chemical impurities can change the color of the same mineral Quartz

The Colors of Beryl beryllium aluminum silicate impurities of different elements or ions change the color of a mineral Iron (Fe) • Aquamarine = blue • Heliodor = yellow • Green beryl Manganese (Mn) • Morganite = pink • Red beryl = red Chromium (Cr) Emerald = emerald green

Corundum: Aluminum oxide Ruby corundum has both chromium and iron impurities, making it red. Sapphire corundum can be many colors, including purple, green, pink, orange, yellow or blue, depending on the amounts of iron and titanium.

Streak the property of a mineral that describes its color in powdered form

Streak When a mineral is rubbed firmly across an unglazed tile of white porcelain (a streak plate), it leaves a line of powder. This is called the streak. The color of the streak is always the same, whether or not the mineral has impurities. For example, quartz leaves a white streak, whether it's violet (amethyst), pink (rose quartz), or brown (smoky quartz).

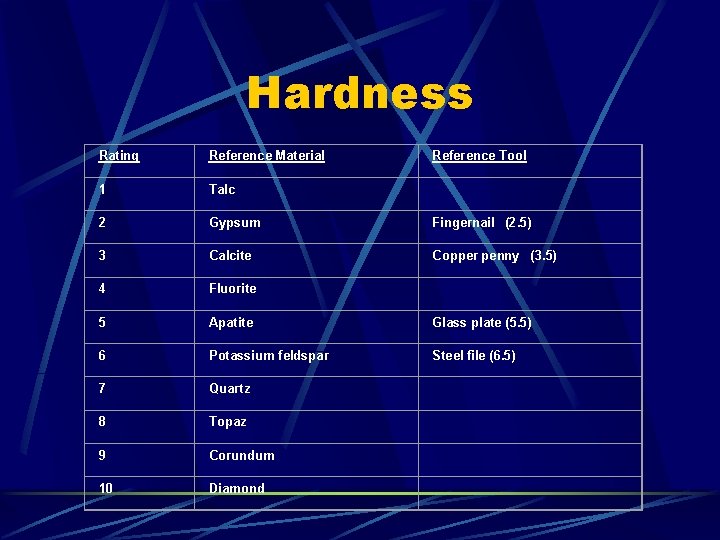
the resistance of a material to scratching

Hardness Rating Reference Material Reference Tool 1 Talc 2 Gypsum Fingernail (2. 5) 3 Calcite Copper penny (3. 5) 4 Fluorite 5 Apatite Glass plate (5. 5) 6 Potassium feldspar Steel file (6. 5) 7 Quartz 8 Topaz 9 Corundum 10 Diamond

Specific Gravity the ratio of the weight of a substance to the weight of an equal volume of water Specific Gravity = Weight in air – Weight in water

Other Properties any other special property of a mineral that distinguishes it from others Magnetic: affects a compass (example magnetite) Double refraction: when a mineral splits the light rays that pass through it, making a single object appear as two objects when you look through the mineral (example calcite)

Other Properties Fluorescent: when a mineral glows under an ultraviolet light (example fluorite) Salty taste: when a mineral tastes like salt (example halite) DO NOT TEST YOUR MINERALS FOR THIS!!! Radioactive: when a mineral gives off subatomic particles that are detected by a Geiger counter (example uraninite)