Chapter 12 Gravimetric Methods of Analysis Gravimetric methods




























- Slides: 35

Chapter 12 Gravimetric Methods of Analysis

Gravimetric methods of analysis 12 A Precipitation gravimetry 12 A-1 Properties of precipitates and precipitating reagents 12 A-2 Particle Size and Filterability of Precipitates 12 A-3 Colloidal Precipitates 12 A-4 Crystalline Precipitates 12 A-5 Coprecipitateion 12 A-6 Precipitation from Homogeneous Solution 12 A-7 Drying and Ignition of Precipitates 12 B Calculation of results from Gravimetric data Example 12 -1, 12 -2, 12 -3 12 C Applications of Gravimetric methods 12 c-1 inorganic Precipitating Agents 12 c-2 Reducing Agents 12 c-3 organic precipitating Agents 12 c-4 Organic functional group analysis 12 c-5 Volatilization gravimetry

There are Several analytical method on mass measurements 1. Precipitation Gravimetry methods The analyte is separated from a solution of the sample as a precipitate and is converted to a compound of known composition that can be weight. 2. Volatilization Gravimetry methods The analyte is separated from other constituents of a sample by conversion or a gas of known chemical composition. 3. Electrogravimetry 4. Gravimetry titrimetry 5. Atomic mass spectrometry

12 A PERCIPITAION GRAVIMETRY • • 2 HN 3+H 2 C 2 O 4→ 2 NH 4++C 2 O 42 Ca 2+(aq)+C 2 O 42 -(aq)→Ca. C 2 O 4(s)→Ca. O(s)+CO (g)+CO 2(g) Example 12 -1

Ex 12 -1:The calcium in a 200. 0 ML sample of a natural water was determined by precipitating the cation as Ca. C 2 O 4. The precipitate was filtered, washed, and ignited in a crucible with an empty mass of 26. 6002 g. the mass of the crucible plus Cao(56. 077 g/mol)was 26. 7134 g. Calcualte the concentration of Ca(40. 078 g/mol)in water in units of grams per 100 m. L of the water. Ans: The mass of Ca. O is 26. 7134 -26. 6002= 0. 1132 g The number of moles Ca in the sample is equal to the number of moles Ca. O amount of Ca = 0. 1132 g. Ca. O × × = 2. 0186× 10 -3 Conc. Ca= = 0. 04045 g/100 ML

12 A-1 Properties of precipitates and precipitating reagents • Specific reagents, which are rare, react only with a single chemical species. • Selective reagents, which are more common, react with only a limited number of species. P. 315 note

The ideal precipitating reagent 1. Readily filtered and washed free of contaminants 2. Of sufficiently low solubility so that no significant loss of the solid occurs during filtration and washing 3. Unreactive with constituents of the atmosphere 4. Of known composition after it is dried or, if necessary, ignited

12 A-2 Particle size and Filterability of Precipitates • Precipitates consisting of large particles because these particles are easy to filter and wash free of impurities 1. Factors that determine the particle size of precipitates (1) Colloidal suspensions (10 -7 to 10 -4 cm in diameter) show no tendency to settle from solution and are not easily filtered (2) Crystalline suspension Tend to settle spontaneously and are easily filtered. • Particle size of a precipitate is influenced by Precipitate solubility, temp. , reactants conc. , the rate at which the reactants are mixed

Q is the concentration of the solute at any instant and S is its equilibrium solubility p. 316 note: supersaturated (Q-S)/S large: colloid; (Q-S)/S small: crystalline

2. Mechanism of Precipitates formation (1) Nucleation is a process in which a minimum number of atoms, ions, or molecules join together to produce a stable solid. (2) Particle growth • If nucleation predominates, a precipitate containing a large number of small particles results;if growth predominates, a smaller number of large particles is produced. 3. Controlling particle size (1)Elevated temperatures to increase the solubility of the precipitate(2) dilute solution (3) slow addition of the precipitating agent with good stirring.

12 A-3 Colloidal Precipitates 1. Coagulation of colloids: by heating, stirring, adding electrolyte (1) Why colloidal suspensions are stable and do not coagulate spontaneously. (a) Charge (cations or anions that are bound to the surface of the particles. Check) (b) Adsorption is a process in which a substance (gas, liquid, or solid) is held on the surface of a solid. . (c) Absorption involves retention of a substance within the pores of a solid. .

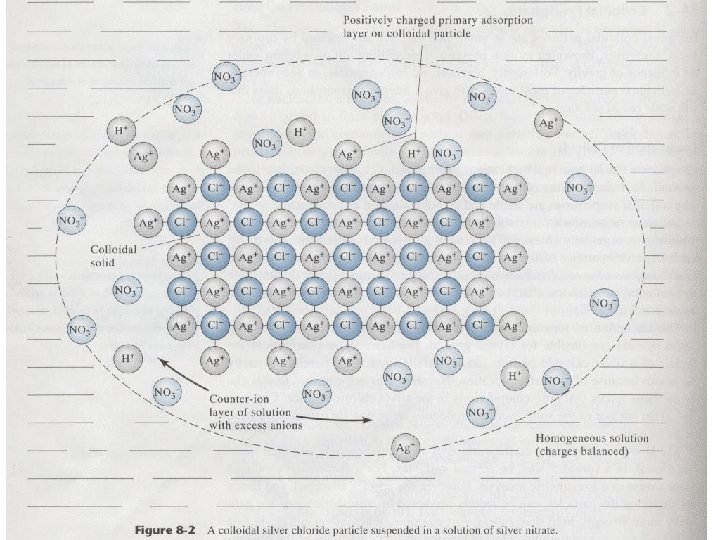
Fig 12 -1 Colloidal silver chloride particle in a solution that contains an excess of silver nitrate. The primary adsorption layer attach directly to the solid surface Ex:consists mainly of adsorbed silver ions. The counter–ion layer surrounding the charged particle is a layer of solution. Ex:which contains sufficient excess of negative ion (principally nitrate)to balance the charge on the surface of the particle. Electrical double layer The primarily adsorbed and the counter-ion layer constitute. This double later exerts an electrostatic repulsive force that prevents particles from colliding and adhering.


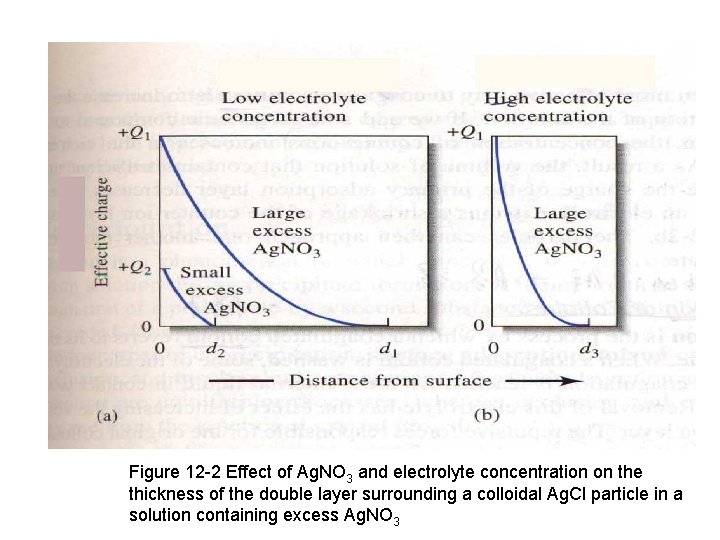
Figure 12 -2 Effect of Ag. NO 3 and electrolyte concentration on the thickness of the double layer surrounding a colloidal Ag. Cl particle in a solution containing excess Ag. NO 3

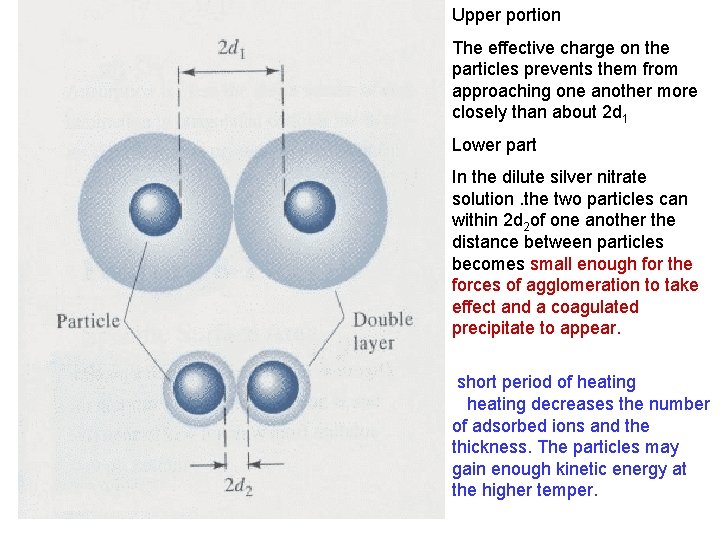
Upper portion The effective charge on the particles prevents them from approaching one another more closely than about 2 d 1 Lower part In the dilute silver nitrate solution. the two particles can within 2 d 2 of one another the distance between particles becomes small enough for the forces of agglomeration to take effect and a coagulated precipitate to appear. short period of heating decreases the number of adsorbed ions and the thickness. The particles may gain enough kinetic energy at the higher temper.

Coagulation of a colloidal suspension can often be brought 1. by short period of heating decreases the number of adsorbed ions and the thickness. The particles may gain enough kinetic energy at the higher temper. 2. Increase the electrolyte concentration of the solution. If we add ionic compound to a colloidal suspension. The concentration of counter – ions increases in the vicinity of each to balance the charge of the primary adsorption layer decreases. The net effect of adding an electrolyte is thus a shrinkage of the counter-ions layer

2. Peptization of colloid Peptization: A coagulated colloid returns to its dispersed state (1) When a coagulated colloid is washed some of the electrolyte responsible for its coagulation is leached from the internal liquid in contact with the solid particles. (2)Washing is needed to minimize contamination on the other, there is a risk of losses resulting from peptization if pure water is used. (3)Solved by washing the precipitate with a solution containing an electrolyte that volatilizes when the precipitate is dried or ignited. commonly 3. Practical treatment of Colloidal precipitates Digestion:It is a process in which a precipitate is heated for an hour or more in the solution from which it was formed(the mother liquor)

12 A-4 Crystalline Precipitates 1、Easily filtered and purified than are coagulated colloids. 2、Method of improving particle size and Filterability (1) minimization of Q (using dilute solution and adding the precipitating reagent slowly and with good mixing. ) (2) maximizing S (precipitating from hot solution or adjusting the p. H of the precipitation medium. )

12 A-5 Coprecipitation It is a process in which normally soluble compounds are carried out of solution by precipitate. There are four types of coprecipitation: 1. surface adsorption, 2. mixed-crystal formation, 3. occlusion, 4. mechanical entrapment.

1. surface adsorption, 1. Adsorption is often the major source of contamination in coagulated colloids but is of no significance in crystalline precipitates. The net effect of surface adsorption is therefore the carrying down of an otherwise soluble compound as a surface contaminant. (1)Minimizing Adsorbed Impurities on Colloids (A) coagulated colloids is improved by digestion (B) Washing a coagulated colloid containing a volatile electrollyte (2)Reprecipitation:A drastic but effective way to minimize the effects of adsorption is Preprecipitation.

2. Mixed-crystal formation (1)Mixed-crystal formation , one of the in the crystal lattice of a solid is replaced by an ion of another element. It is necessary that the two ions have the same charge and that their sizes differ by no more than about 5%. The same crtstal class. Ex (Pb ion replace some of the barium ion) (2)The interfering ion may have to be separated before the final precipitation step. Precipitation reagent.

3. Occlusion is a type of coprecipitation in which a compound is trapped within a pocket formed during rapid crystal growth. 4. Mechanical entrapment occurs when crystals lie close together during growth. Here, several crystals grow together and in so doing trap a portion of the solution in a tiny pocket. Coprecipitation Errors It impurities may cause either negative or positive errors in an analysis.

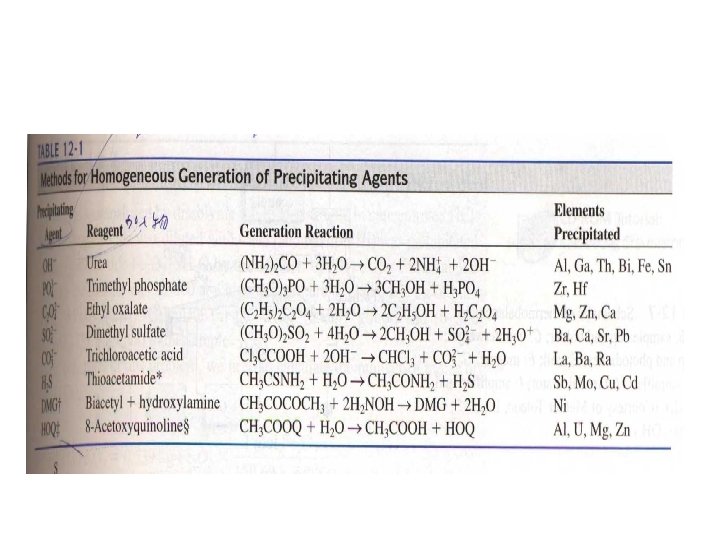
12 A-6 Precipitation from Homogeneous Solution • Homogeneous precipitation is a process in which a precipitate is formed by slow generation of a precipitating reagent homogeneously throughout a solution. • (H 2 N)2 CO+3 H 2 O→CO 2+2 NH 4++2 OH-

Fig 12 -5 shows hydrous oxide precipitates of aluminum formed by direct addition of base and by homogeneous precipitates with urea.


12 A-7 Drying and lgnition of Precipitates Weighing form Some precipitates are also ignited to decompose the solid and form a compound of Known composition Fig 12 -6 Effect of temperature of precipitate mass.

12 BCalculation of results from gravimetric data Ex 12 -1:The calcium in a 200. 0 ML sample of a natural water was determined by precipitating the cation as Ca. C 2 O 4. The precipitate was filtered, washed, and ignited in a crucible with an empty mass of 26. 6002 g. the mass of the crucible plus Cao(56. 077 g/mol)was 26. 7134 g. Calcualte the concentration of Ca(40. 078 g/mol)in water in units of grams per 100 m. L of the water. Ans: The mass of Ca. O is 26. 7134 -26. 6002= 0. 1132 g The number of moles Ca in the sample is equal to the number of moles Ca. O = 0. 1132 g. Ca. O × × = 2. 0186× 10 -3 Conc. Ca= = 0. 04045 g/100 ML amount of Ca

Example 12 -2:Anirom ore was analyzed by dissolving a 1. 1324 g sample in concentrated HCL. The resulting solution was diluted with water, and the iron( Ⅲ)was precipitated as the hydrous oxide Fe 2 O 3. x. H 2 O by the addition of NH 3. After filtration and washing, the residue was ignited at a high temperature to give 0. 5394 g of pure Fe 2 O 3 (159. 69 g/mol. ). Calculate (a) a the %Fe (55. 847 g/mol ) and (b) The % Fe 3 O 4(231. 54 g/mol) in the sample . Ans: amount Fe 2 O 3= 0. 5394 g Fe 2 O 3× (a)The number of moles of Fe is twice the number of moles of Fe 2 O 3, and Mass Fe = 3. 3778× 10 -3× = 0. 37728 Fe %Fe = %= 33. 32% (b) mass. Fe 3 O 4= 3. 3778× 10 -3 %Fe 3 O 4= %= 46. 04%

Ex 12 -3 A 0. 2356 g sample containing only Nacl and Ba. Cl 2 Yielded 0. 4637 g of dried Ag Cl. Calculate the percent of each halogen compound in the sample. Sol:X+y = 0. 2356 g sample ;X= Na. Cl y= Ba. Cl 2 Amount Ag. Cl from Na. Cl =xg. Na. Cl× = 0. 017111 xmol Agcl Mass Ag. Cl from Na. Cl= 0. 017111 xmol Ag. Cl× 143. 32 g Ag. Cl/mol. Ag. Cl = 2. 4524 g Proceeding in the same way, wi can write that the number of moles of Ag. Cl from the Ba. Cl 2 is given by Amount Ag. Cl from Ba. Cl 2 =yg. Ba. Cl 2× Amount Ag. Cl from Ba. Cl 2 = 9. 605× 10 -3× 143. 32= 1. 3766 yg Ag. Cl 2. 4524 x+1. 3766 y= 0. 4637 Y= 0. 2356 -x; 2. 4524 x+1. 3766(0. 2356 -x) = 0. 4637 1. 0758 x= 0. 13942;x=mass Na. Cl= 0. 12960 g. Na. Cl %Na. Cl= %Ba. Cl 2= 100. 00%-55. 01%= 44. 99%

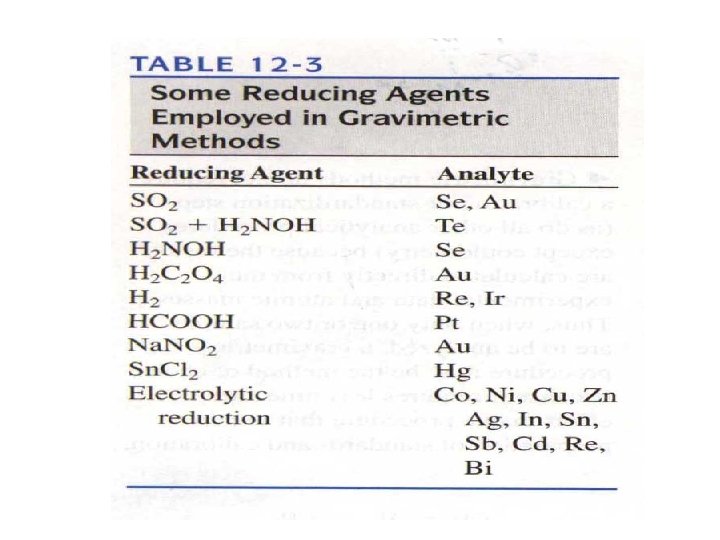
12 C Applications of gravimetric methods 12 C-1 Inorganic Precipitating Agents 12 C-2 Reducing Agents

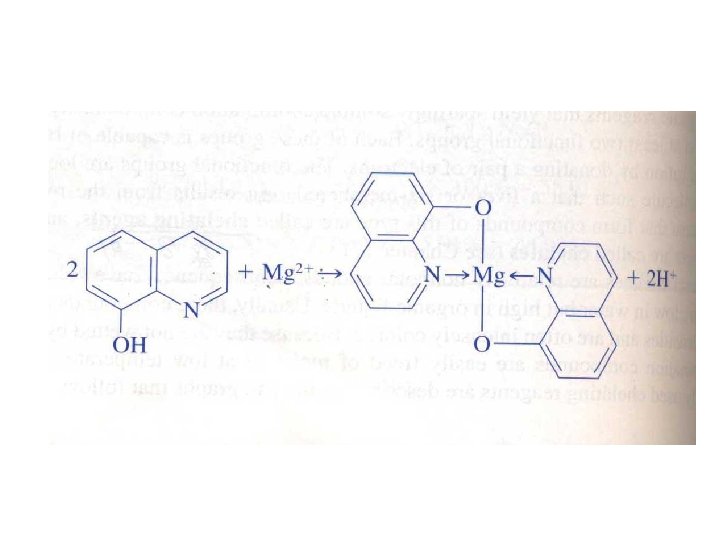
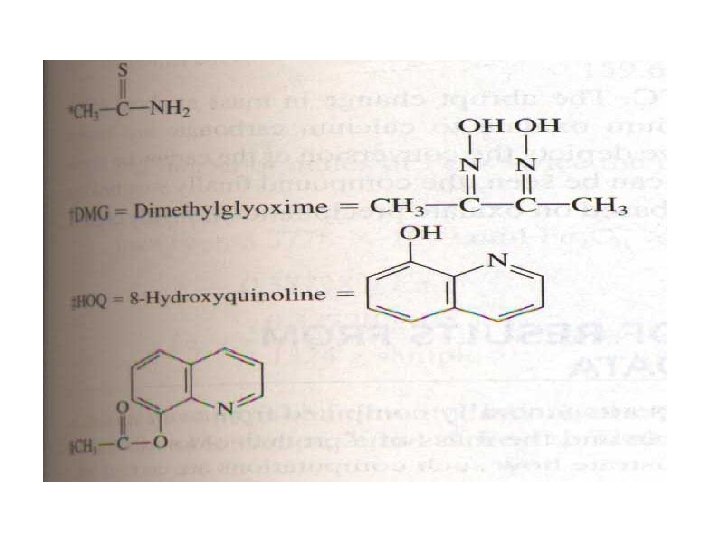
12 C-3 Organic precipitating Agents




12 C-5 Voltilization Gravimetry The two most common gravimetric methods based on volatilization are those for determining water and carbon dioxide (1)water A. Direct determination B. Indirect method (2)CO 2 (It is the determination of the sodium hydrogen carbonate content of antacid tablets) 2 Na. OH+CO 2 → Na 2 CO 3+H 2 O