Air Pollution Inorganic gaseous pollutants Page 127 By

Air Pollution: Inorganic gaseous pollutants Page: 127

By human activities 4 major air pollutants: CO, SO 2, NO 2, & O 3 Other 2 criteria air pollutants - Volatile organic compounds (VOCs) and lead (Pb)
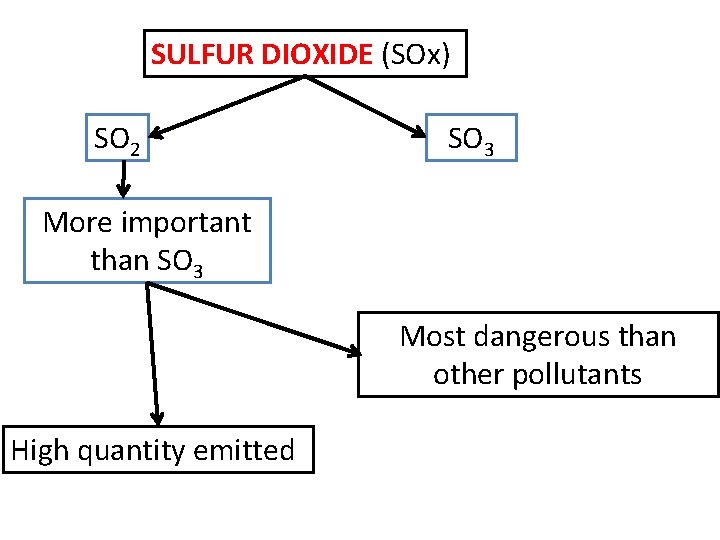
SULFUR DIOXIDE (SOx) SO 2 SO 3 More important than SO 3 Most dangerous than other pollutants High quantity emitted
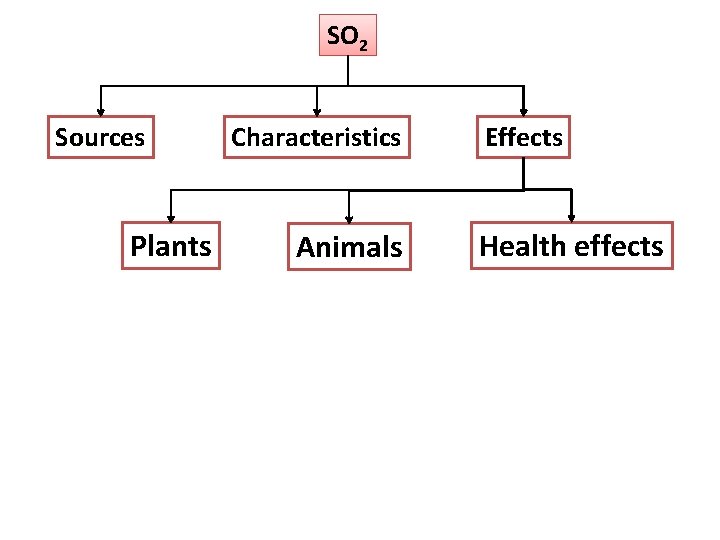
SO 2 Sources Plants Characteristics Animals Effects Health effects
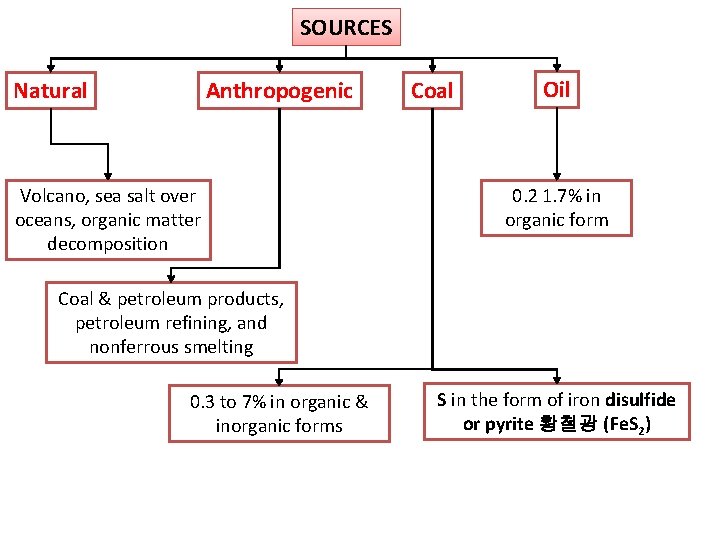
SOURCES Anthropogenic Natural Volcano, sea salt over oceans, organic matter decomposition Coal Oil 0. 2 1. 7% in organic form Coal & petroleum products, petroleum refining, and nonferrous smelting 0. 3 to 7% in organic & inorganic forms S in the form of iron disulfide or pyrite 황철광 (Fe. S 2)
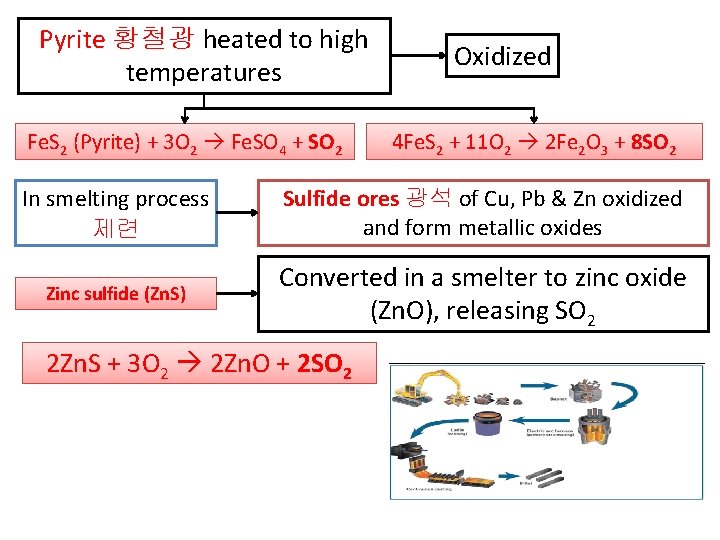
Pyrite 황철광 heated to high temperatures Fe. S 2 (Pyrite) + 3 O 2 Fe. SO 4 + SO 2 Oxidized 4 Fe. S 2 + 11 O 2 2 Fe 2 O 3 + 8 SO 2 In smelting process 제련 Sulfide ores 광석 of Cu, Pb & Zn oxidized and form metallic oxides Zinc sulfide (Zn. S) Converted in a smelter to zinc oxide (Zn. O), releasing SO 2 2 Zn. S + 3 O 2 2 Zn. O + 2 SO 2
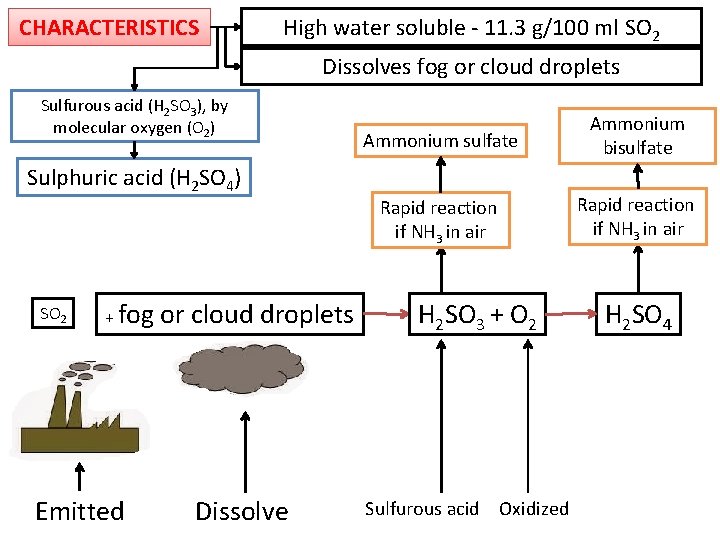
CHARACTERISTICS High water soluble - 11. 3 g/100 ml SO 2 Dissolves fog or cloud droplets Sulfurous acid (H 2 SO 3), by molecular oxygen (O 2) Ammonium sulfate Ammonium bisulfate Rapid reaction if NH 3 in air Sulphuric acid (H 2 SO 4) SO 2 + fog Emitted or cloud droplets Dissolve H 2 SO 3 + O 2 Sulfurous acid Oxidized H 2 SO 4
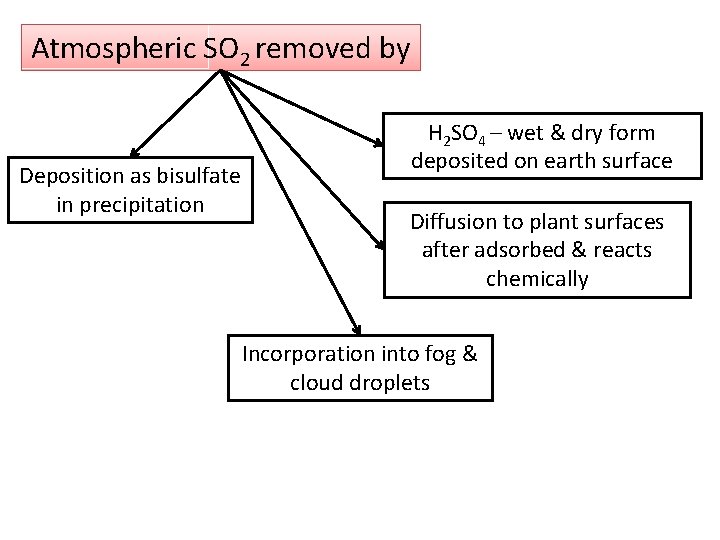
Atmospheric SO 2 removed by Deposition as bisulfate in precipitation H 2 SO 4 – wet & dry form deposited on earth surface Diffusion to plant surfaces after adsorbed & reacts chemically Incorporation into fog & cloud droplets
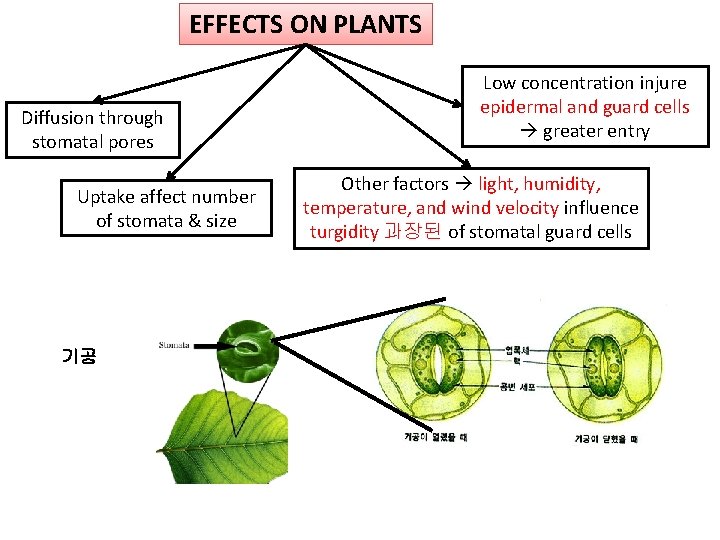
EFFECTS ON PLANTS Diffusion through stomatal pores Uptake affect number of stomata & size 기공 Low concentration injure epidermal and guard cells greater entry Other factors light, humidity, temperature, and wind velocity influence turgidity 과장된 of stomatal guard cells
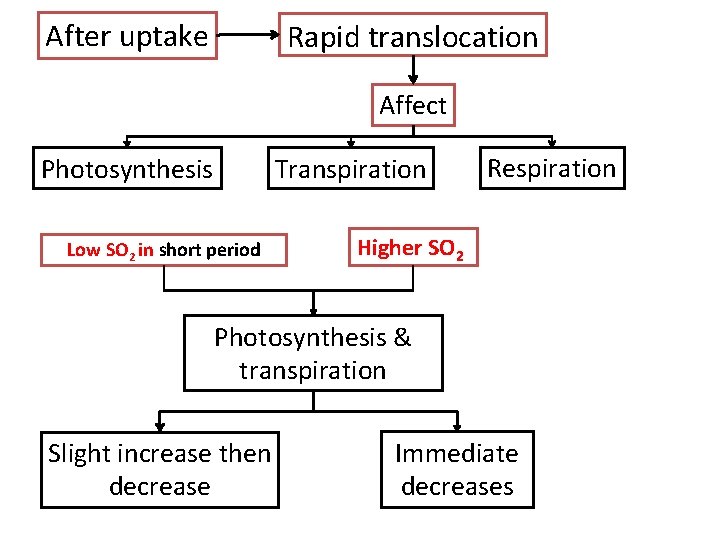
After uptake Rapid translocation Affect Photosynthesis Transpiration Low SO 2 in short period Respiration Higher SO 2 Photosynthesis & transpiration Slight increase then decrease Immediate decreases
Plant injuries Leaf chlorosis & spotty necrotic lesions Synergistic effect on leaf damage SO 2 + O 3 Damage mesophyll cells
Leaf absorbed SO 2 Dissolves intercellular water Form bisulfite (HSO 3), sulfite (SO 32 -) & ionic species
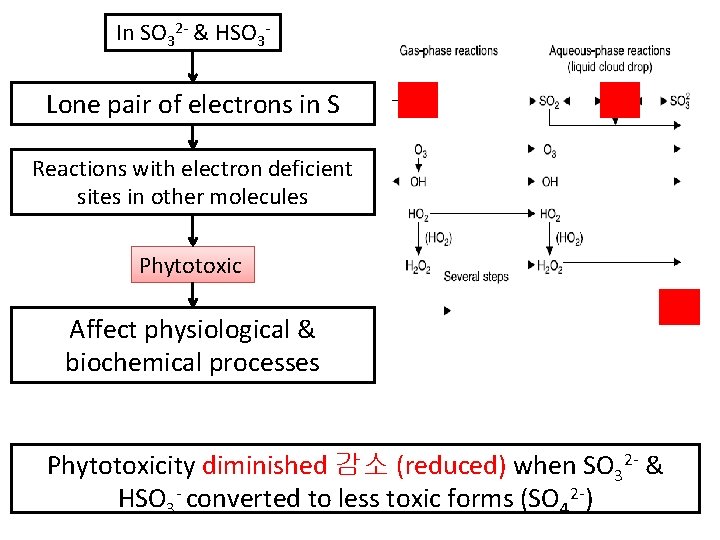
In SO 32 - & HSO 3 - Lone pair of electrons in S Reactions with electron deficient sites in other molecules Phytotoxic Affect physiological & biochemical processes Phytotoxicity diminished 감소 (reduced) when SO 32 - & HSO 3 - converted to less toxic forms (SO 42 -)
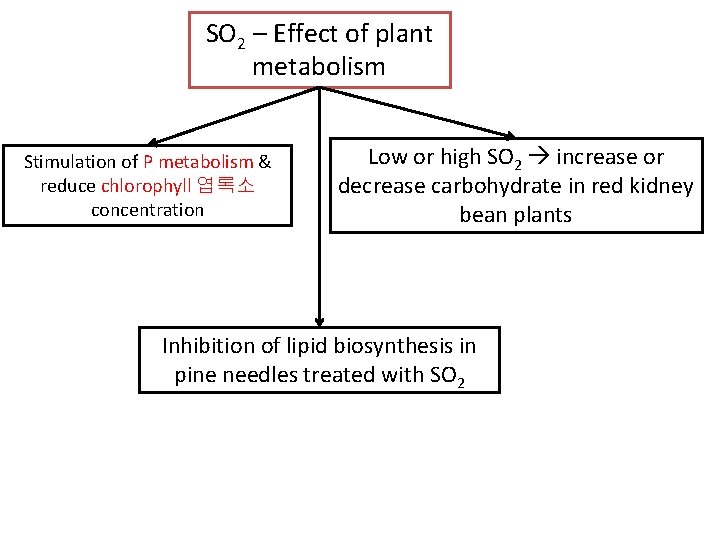
SO 2 – Effect of plant metabolism Stimulation of P metabolism & reduce chlorophyll 엽록소 concentration Low or high SO 2 increase or decrease carbohydrate in red kidney bean plants Inhibition of lipid biosynthesis in pine needles treated with SO 2
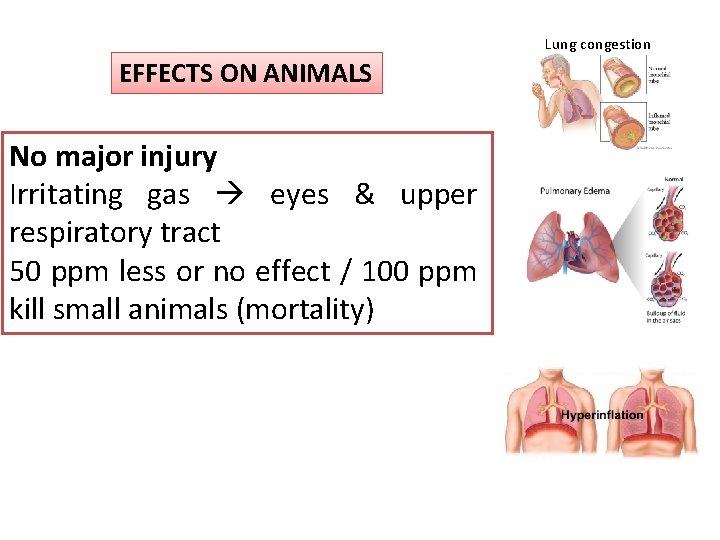
Lung congestion EFFECTS ON ANIMALS No major injury Irritating gas eyes & upper respiratory tract 50 ppm less or no effect / 100 ppm kill small animals (mortality)
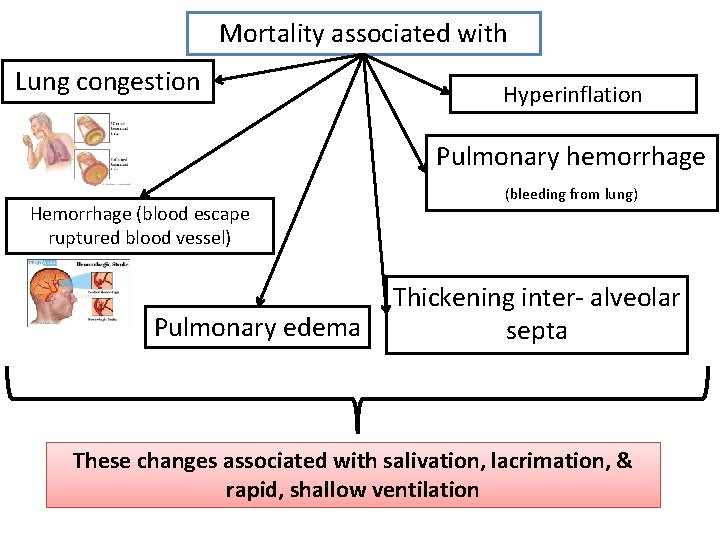
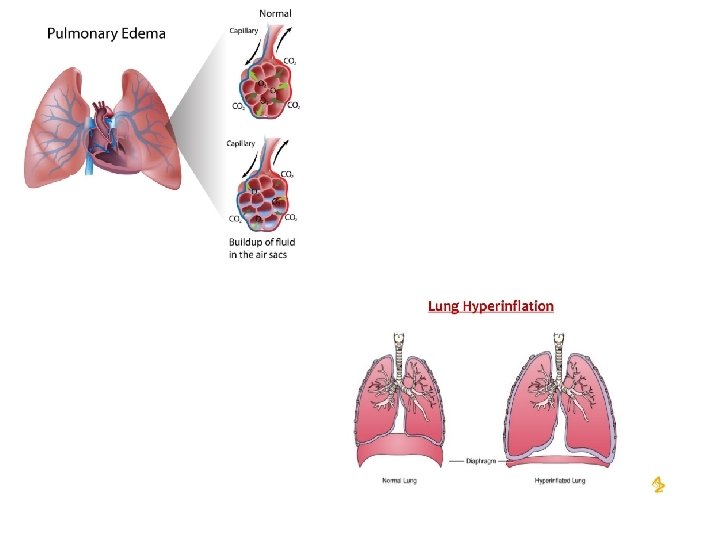
Mortality associated with Lung congestion Hyperinflation Pulmonary hemorrhage Hemorrhage (blood escape ruptured blood vessel) Pulmonary edema (bleeding from lung) Thickening inter- alveolar septa These changes associated with salivation, lacrimation, & rapid, shallow ventilation
NITROGEN DIOXIDE Formation Reactive species in troposphere Effects on plants Biological effects Health effects

FORMS & FORMATION NOx Nitrous oxide (N 2 O) Nitric oxide (NO) Nitrogen dioxide (NO 2) Nitrogen pentoxide (N 2 O 5) Nitrogen tetroxide (N 2 O 4) Nitrogen trioxide (N 2 O 3) NO 2 - most important air pollutant – high toxicity
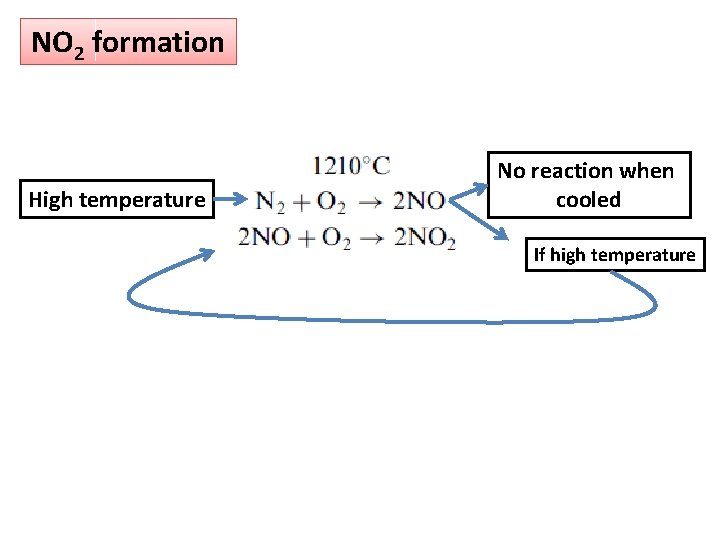
NO 2 formation High temperature No reaction when cooled If high temperature
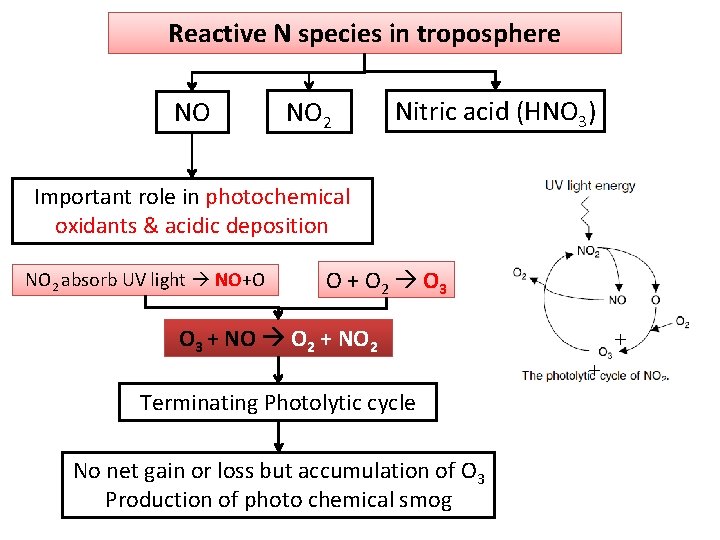
Reactive N species in troposphere NO NO 2 Nitric acid (HNO 3) Important role in photochemical oxidants & acidic deposition NO 2 absorb UV light NO+O O + O 2 O 3 + NO O 2 + NO 2 + + Terminating Photolytic cycle No net gain or loss but accumulation of O 3 Production of photo chemical smog
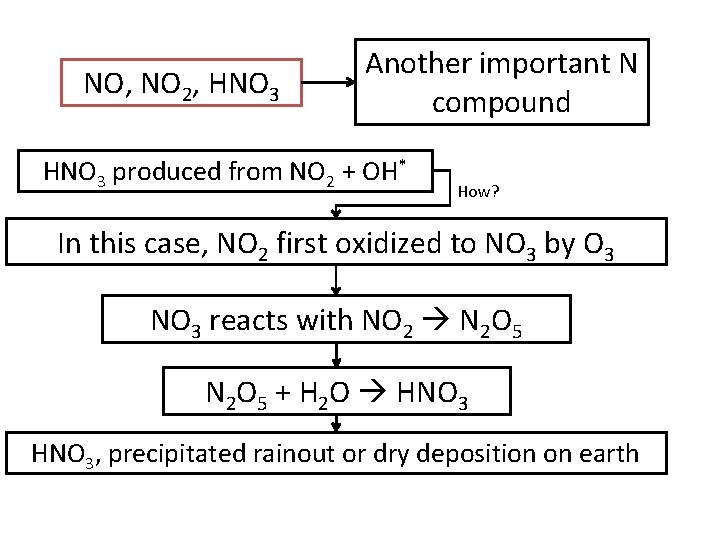
NO, NO 2, HNO 3 Another important N compound HNO 3 produced from NO 2 + OH* How? In this case, NO 2 first oxidized to NO 3 by O 3 NO 3 reacts with NO 2 N 2 O 5 + H 2 O HNO 3, precipitated rainout or dry deposition on earth
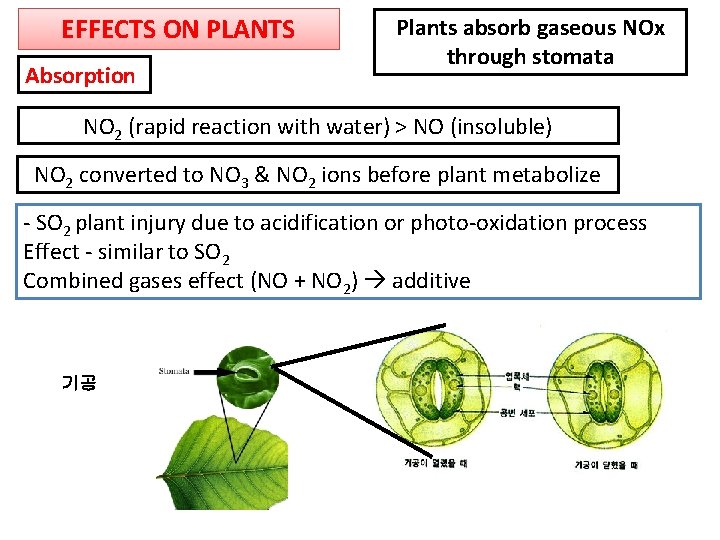
EFFECTS ON PLANTS Absorption Plants absorb gaseous NOx through stomata NO 2 (rapid reaction with water) > NO (insoluble) NO 2 converted to NO 3 & NO 2 ions before plant metabolize - SO 2 plant injury due to acidification or photo-oxidation process Effect - similar to SO 2 Combined gases effect (NO + NO 2) additive 기공
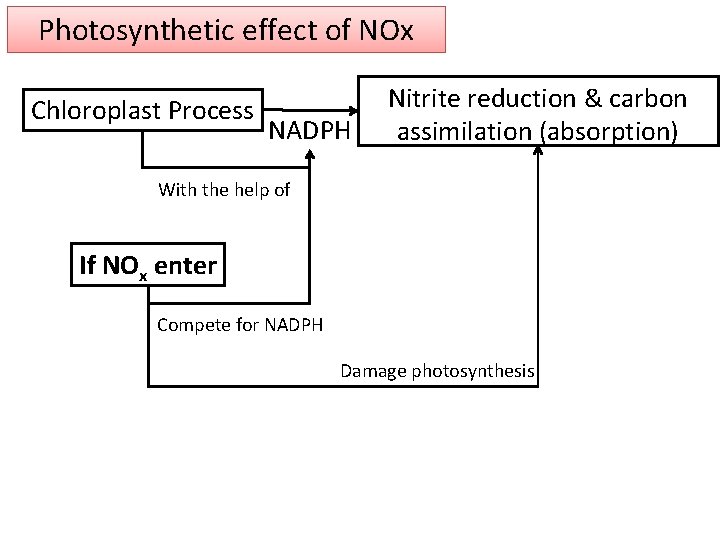
Photosynthetic effect of NOx Chloroplast Process NADPH Nitrite reduction & carbon assimilation (absorption) With the help of If NOx enter Compete for NADPH Damage photosynthesis
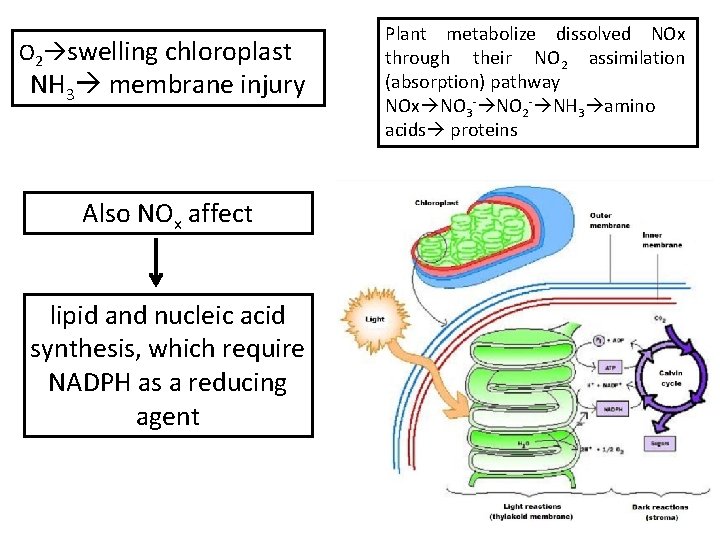
O 2 swelling chloroplast NH 3 membrane injury Also NOx affect lipid and nucleic acid synthesis, which require NADPH as a reducing agent Plant metabolize dissolved NOx through their NO 2 assimilation (absorption) pathway NOx NO 3 - NO 2 - NH 3 amino acids proteins

Health effects Toxic action NO 2 deep lung & peripheral airway NO 2 at 10 to 25 ppm for 24 h induce fibrin (protein) production Pulmonary alveoli Disturbed cilia Pneumonia Respiratory illness in children

BIOLOGICAL EFFECTS Inhaled NO 2 converted to NO 2 & NO 3 ions in lungs Found in the blood and urine after exposure 24 ppm of NO 2 Increased respiration, slow weight gain, rats decreased swimming ability, lowered hemoglobin content & methemoglobin formation occurred only at high concentrations.

OZONE - SOURCES O 3 contributing atmospheric pollution Photochemical Smog 광화학 스모그 O 3 removed from atmosphere by back reaction Back reaction faster than the forward reaction O 3 removal faster than O 3 formation
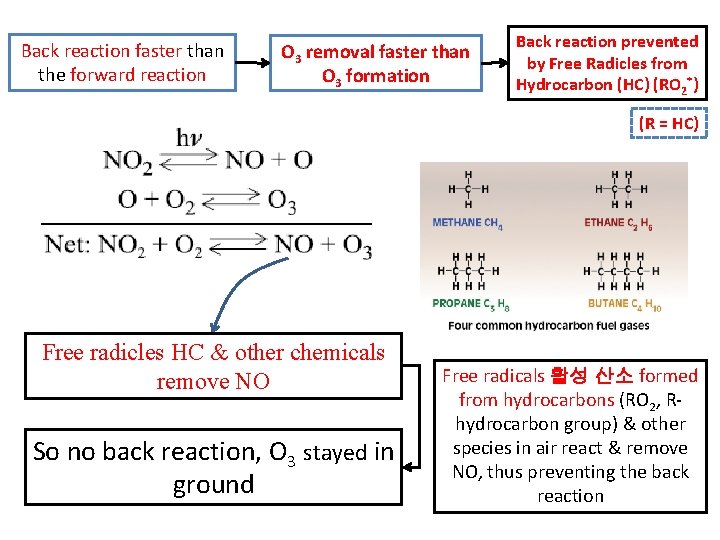
Back reaction faster than the forward reaction O 3 removal faster than O 3 formation Back reaction prevented by Free Radicles from Hydrocarbon (HC) (RO 2*) (R = HC) Free radicles HC & other chemicals remove NO So no back reaction, O 3 stayed in ground Free radicals 활성 산소 formed from hydrocarbons (RO 2, Rhydrocarbon group) & other species in air react & remove NO, thus preventing the back reaction
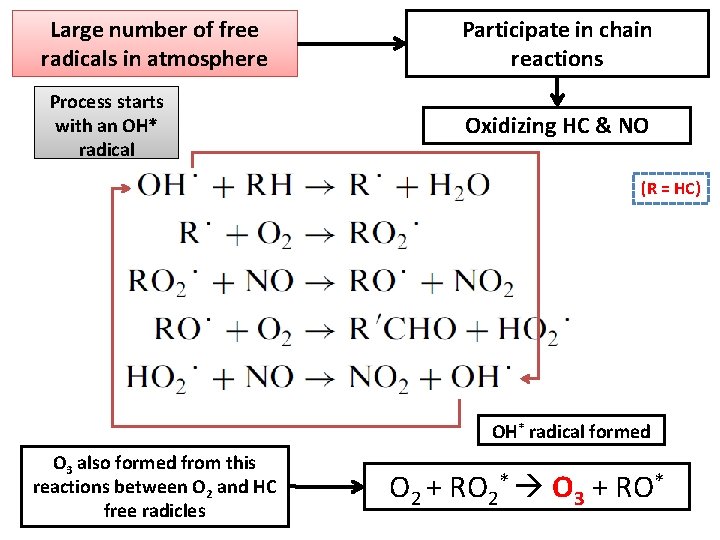
Large number of free radicals in atmosphere Process starts with an OH* radical Participate in chain reactions Oxidizing HC & NO (R = HC) OH* radical formed O 3 also formed from this reactions between O 2 and HC free radicles O 2 + RO 2* O 3 + RO*
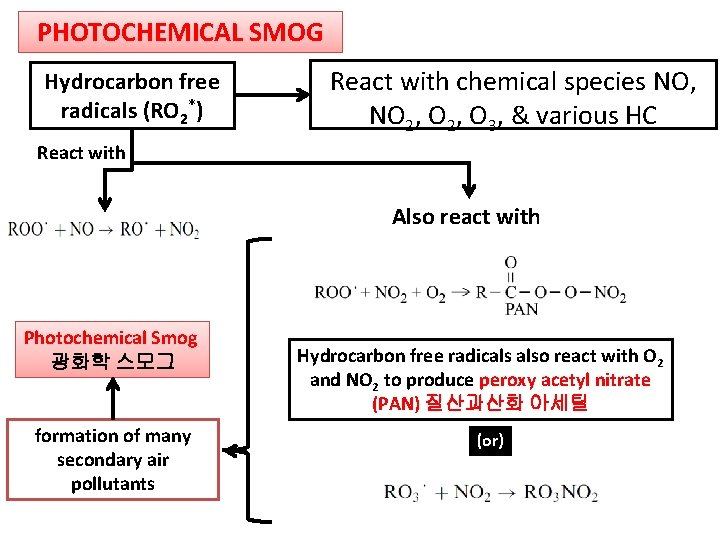
PHOTOCHEMICAL SMOG Hydrocarbon free radicals (RO 2*) React with chemical species NO, NO 2, O 3, & various HC React with Also react with Photochemical Smog 광화학 스모그 formation of many secondary air pollutants Hydrocarbon free radicals also react with O 2 and NO 2 to produce peroxy acetyl nitrate (PAN) 질산과산화 아세틸 (or)
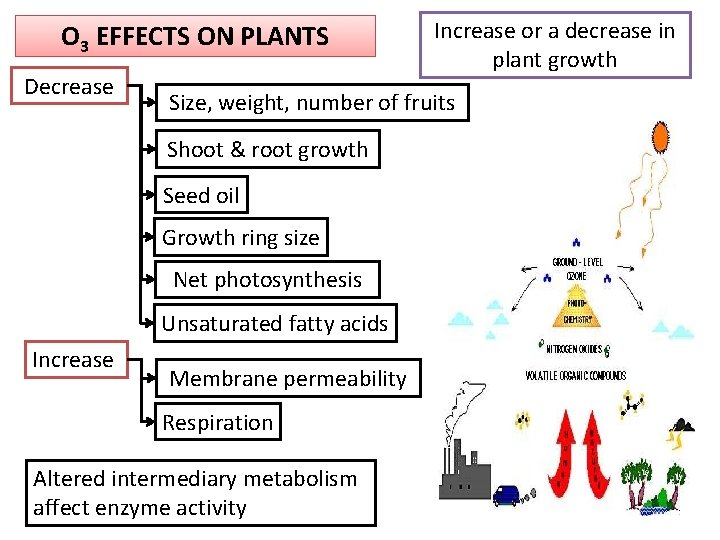
O 3 EFFECTS ON PLANTS Decrease Size, weight, number of fruits Shoot & root growth Seed oil Growth ring size Net photosynthesis Unsaturated fatty acids Increase or a decrease in plant growth Membrane permeability Respiration Altered intermediary metabolism affect enzyme activity

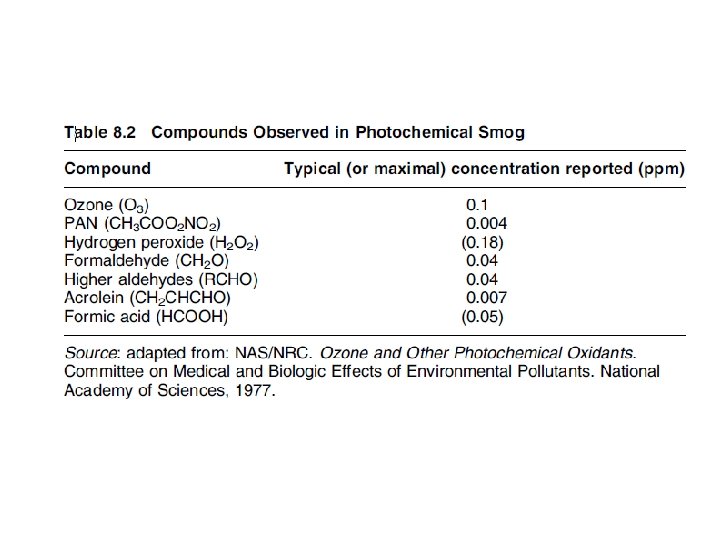
EFFECTS ON ANIMALS AND HUMANS Irritating respiratory tract & eye Threshold limit value (TLV) for O 3 in industry is 0. 1 ppm 0. 6 to 0. 8 ppm O 3 for 60 minutes headache, nausea, anorexia, coughing, chest pain, shortness of breath Experimental evidences in book

BIOLOGICAL EFFECTS Reactions with proteins 단백질 and amino acids 아 미노산 Reactions with lipids 지질 Formation of free radicals Experimental evidences in book

CARBON MONOXIDE Can’t see it Atmospheric concentration - 0. 1 ppm Can’t smell it 500 million metric tons – residence - 36 to 110 days. Can’t feel it Deadly gas kills over 5000 From CH 4 – Oxidation process – CO peoples each year Degradation of chlorophyll - 20% of the total annual release Comes from appliances fuel-burning
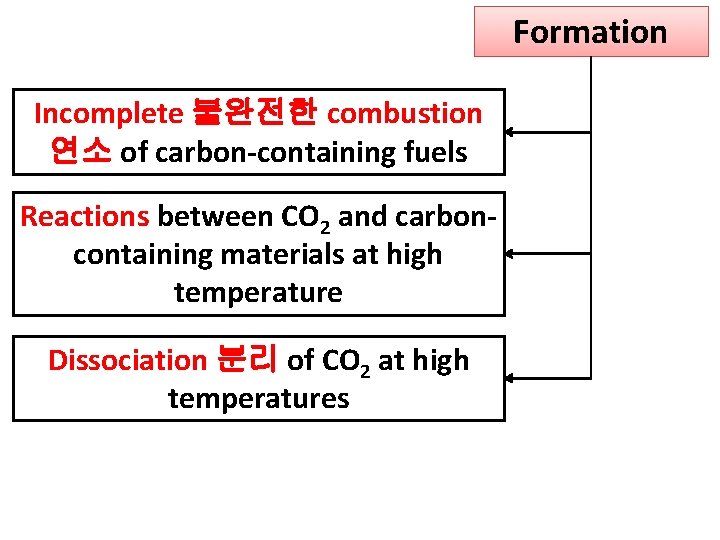
Formation Incomplete 불완전한 combustion 연소 of carbon-containing fuels Reactions between CO 2 and carboncontaining materials at high temperature Dissociation 분리 of CO 2 at high temperatures

Incomplete combustion Available oxygen is less than amount required for complete combustion It will also occur when there is poor mixing of fuel & air.
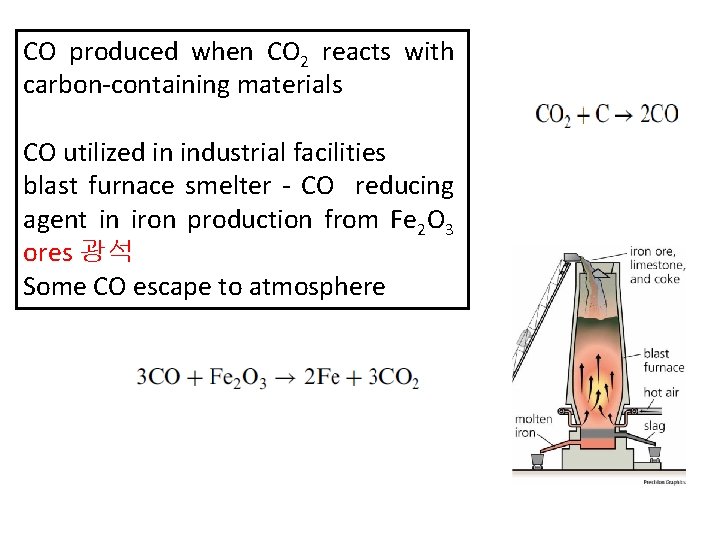
CO produced when CO 2 reacts with carbon-containing materials CO utilized in industrial facilities blast furnace smelter - CO reducing agent in iron production from Fe 2 O 3 ores 광석 Some CO escape to atmosphere

CO also be produced by dissociation of carbon dioxide into CO and O at high temperatures
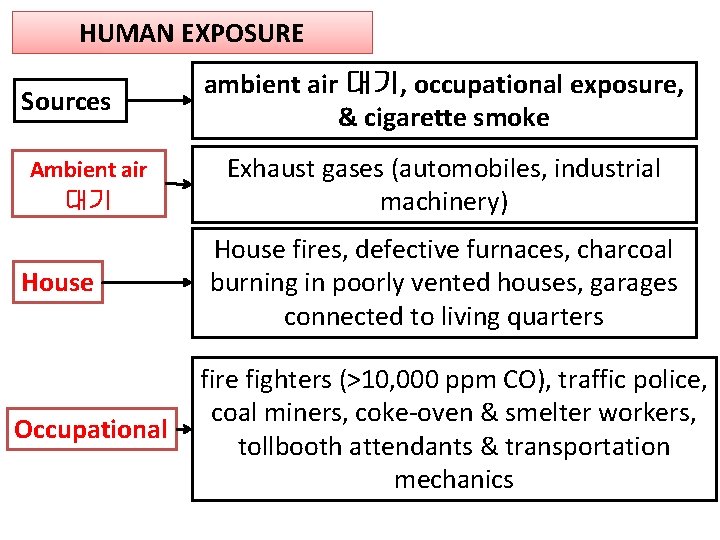
HUMAN EXPOSURE Sources Ambient air 대기 House Occupational ambient air 대기, occupational exposure, & cigarette smoke Exhaust gases (automobiles, industrial machinery) House fires, defective furnaces, charcoal burning in poorly vented houses, garages connected to living quarters fire fighters (>10, 000 ppm CO), traffic police, coal miners, coke-oven & smelter workers, tollbooth attendants & transportation mechanics
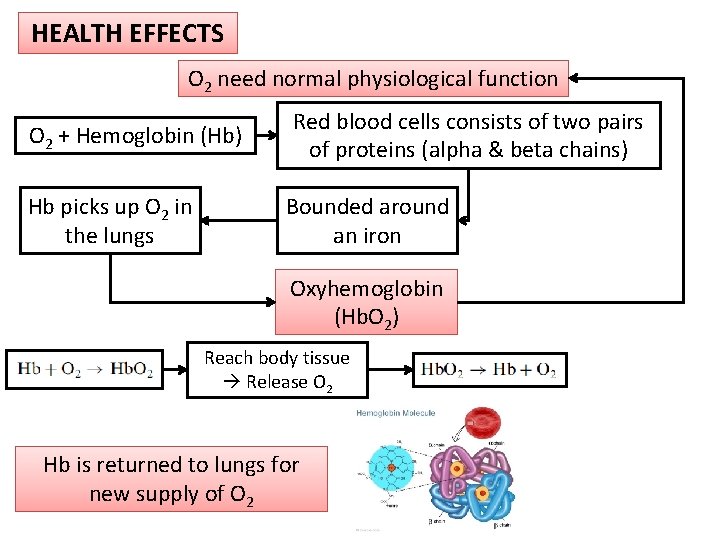
HEALTH EFFECTS O 2 need normal physiological function O 2 + Hemoglobin (Hb) Hb picks up O 2 in the lungs Red blood cells consists of two pairs of proteins (alpha & beta chains) Bounded around an iron Oxyhemoglobin (Hb. O 2) Reach body tissue Release O 2 Hb is returned to lungs for new supply of O 2

CO Toxicity CO enters bloodstream & reduces red blood cells ability to deliver oxygen to body’s organs & tissues The toxic action of CO involves the formation of carboxyhemoglobin (COHb or Hb. CO): Affinity of CO for Hb is more than 200 times greater than O 2 Presence of CO, Hb. O 2 readily releases the bound O 2 and picks up CO to form Hb. CO
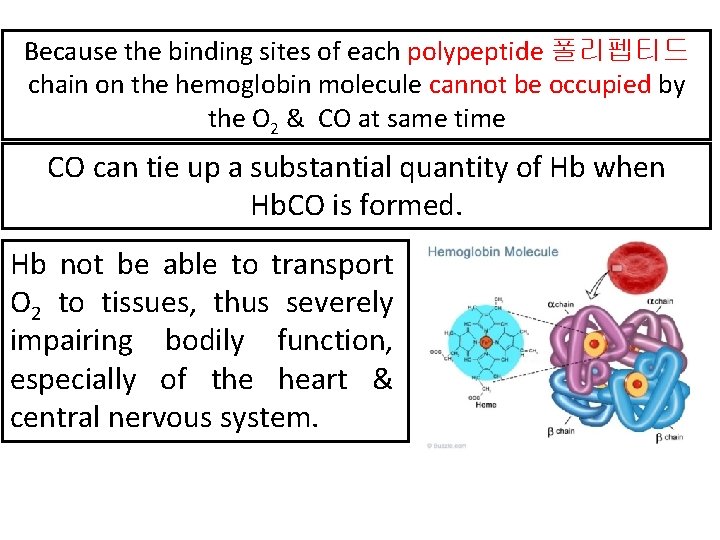
Because the binding sites of each polypeptide 폴리펩티드 chain on the hemoglobin molecule cannot be occupied by the O 2 & CO at same time CO can tie up a substantial quantity of Hb when Hb. CO is formed. Hb not be able to transport O 2 to tissues, thus severely impairing bodily function, especially of the heart & central nervous system.
- Slides: 44