Cofactors Cofactors Cofactors are organic or inorganic molecules













- Slides: 35

Cofactors

Cofactors • Cofactors are organic or inorganic molecules that are required for the activity of a certain conjugated enzymes • Apoenzyme = enzyme (-) cofactor • Holoenzyme = enzyme (+) cofactor • Inorganic cofactors – essential ions • Organic cofactors – coenzymes

Essential Ion Cofactors • Activator ions – bind reversibly to enzyme and often participate in substrate binding. • Metal ions of metalloenzymes – cations that are tightly bound to enzyme and participate directly in catalysis (Fe, Zn, Cu, Co). • Metal activated enzymes – require or are stimulated by addition of metal ions (i. e. Mg 2+, is required by many ATP requiring enzymes)

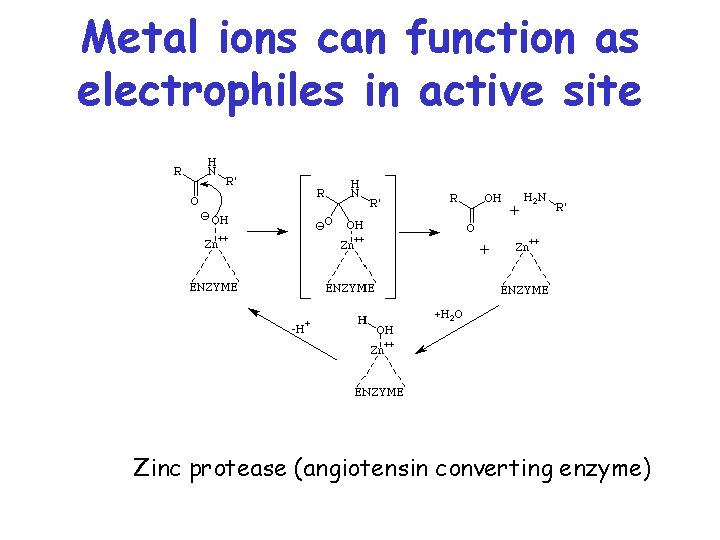
Metal ions can function as electrophiles in active site Zinc protease (angiotensin converting enzyme)

Coenzymes Cosubstrates- altered in rxn and regenerated to original structure in subsequent rxn - disassociated from active site - shuttle chemical groups among different enzyme rxns. Prosthetic groups- remains bound to enzyme - must return to original form Both cosubstrates and prosthetic groups supply reactive groups not present on amino acid side chains

Coenzymes • Metabolite coenzymes – synthesized from common metabolites • Nucleoside triphosphates – (ATP) can donate phosphates, pyrophosphates, adenosyl grroups • S-adenosylmethionine (SAM) – donates methyl groups • Nucleotide sugars (uridine diphosphate glucose = UDP-glucose) - transfer sugars in carbohydrate metabolism

Vitamin derived coenzymes • Must be obtained from diet • Synthesized by microorganisms and plants • Vitamin deficiencies lead to disease state • Most vitamins must be enzymatically transformed to function as a coenzyme

Vitamin Ascorbic acid (C) Niacin Riboflavin (B 2) Thiamin (B 1) Pyridoxal (B 6) Biotin Folate Cobalamin (B 12) Vitamin A Vitamin K Pantothenate (B 3) Vitamins Coenzyme not a coenzyme NAD(P)+/NAD(P)H FMN & FAD Thiamin-pyrophosphate Pyridoxal phosphate Biotin Tetrahydrafolate adenosyl-and methylcobalamin Retinal Vitamin K Coenzyme A

Niacin (nicotinic acid) • Deficiencies lead to pellagra (dermatitis, diarrhea, dementia) • Required in relatively high amounts compared to other vitamins • Not true enzyme because can be synthesized from tryptophan in the liver

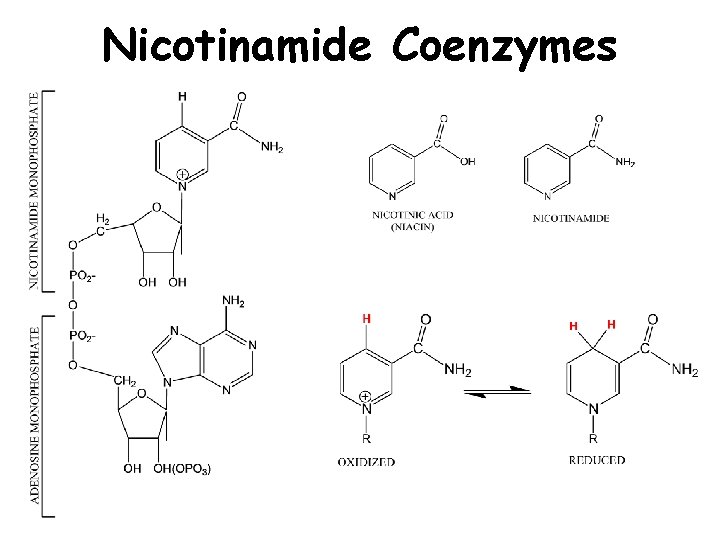
Nicotinamide Coenzymes

+ NAD / + NADP • Serve as cofactors in oxidation/reduction reactions • Act as co-substrates for dehydrogenases • Reduction of NAD+/NADP+ and oxidation of NADH/NADPH occurs 2 e- at a time. • Function in hydride ion transfer • Rxns forming NADH/NADPH are catabolic • NADH is coupled with ATP production in mitochondria • NADPH is an impt reducing agent in biosynthetic reactions • Reduced forms (NADH/NADPH) absorb light at 340 nm, oxidized forms (NAD+/NADP+) do not

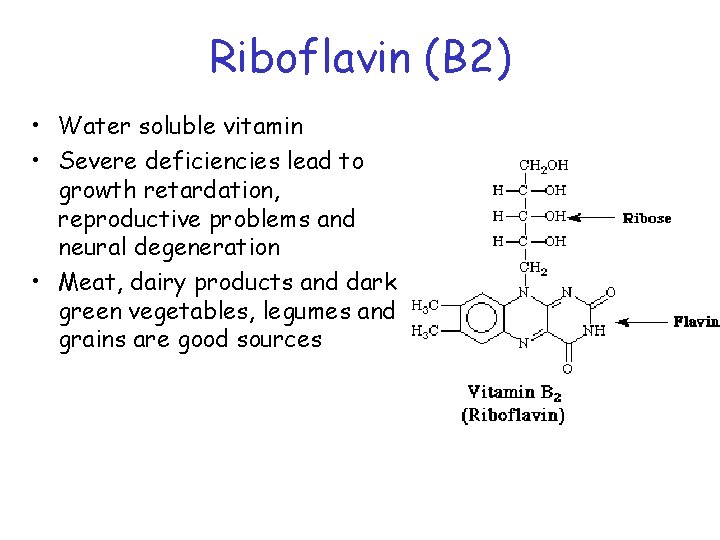
Riboflavin (B 2) • Water soluble vitamin • Severe deficiencies lead to growth retardation, reproductive problems and neural degeneration • Meat, dairy products and dark green vegetables, legumes and grains are good sources

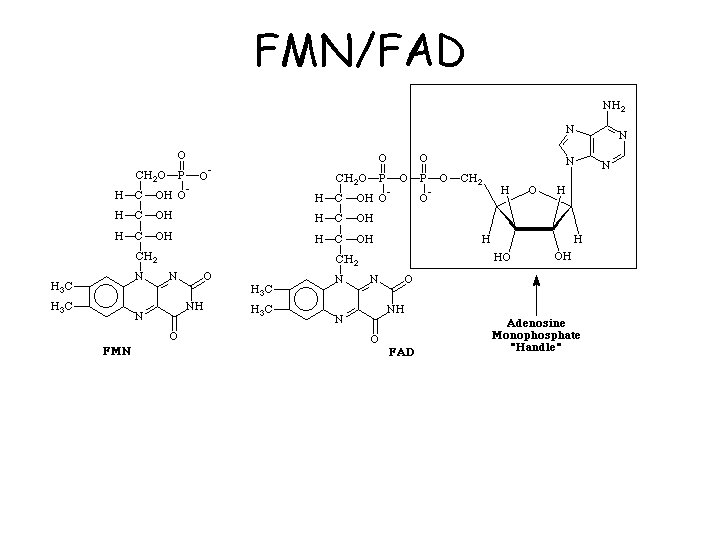
FMN/FAD

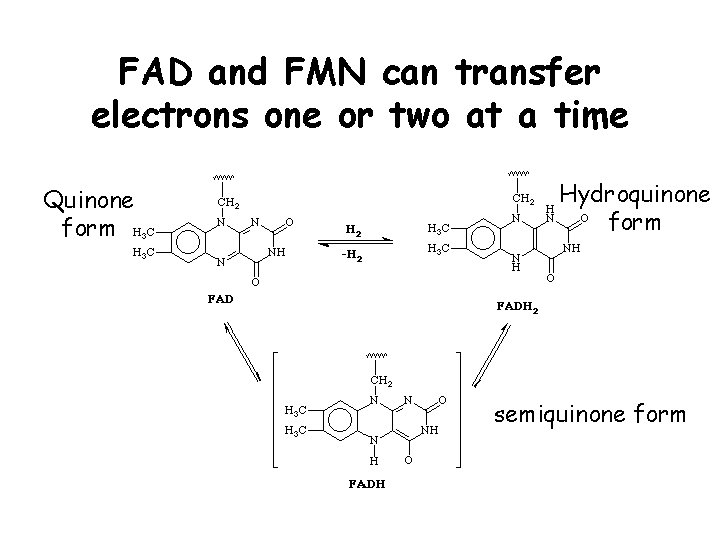
FAD and FMN can transfer electrons one or two at a time Quinone form Hydroquinone form semiquinone form

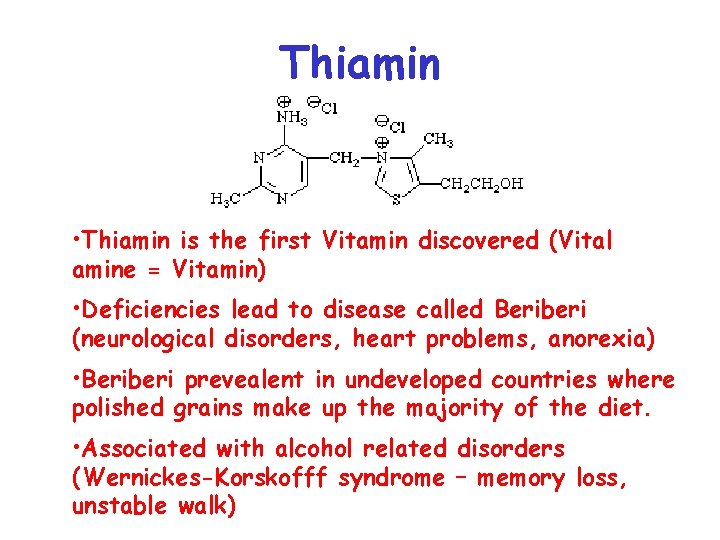
Thiamin • Thiamin is the first Vitamin discovered (Vital amine = Vitamin) • Deficiencies lead to disease called Beriberi (neurological disorders, heart problems, anorexia) • Beriberi prevealent in undeveloped countries where polished grains make up the majority of the diet. • Associated with alcohol related disorders (Wernickes-Korskofff syndrome – memory loss, unstable walk)

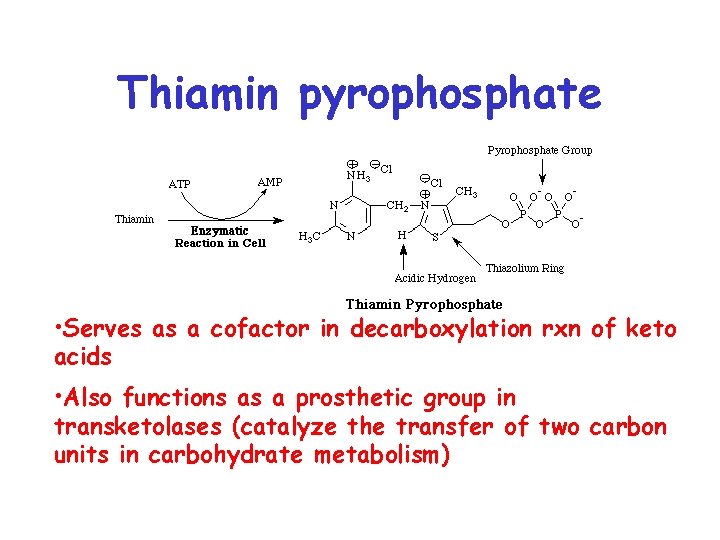
Thiamin pyrophosphate • Serves as a cofactor in decarboxylation rxn of keto acids • Also functions as a prosthetic group in transketolases (catalyze the transfer of two carbon units in carbohydrate metabolism)

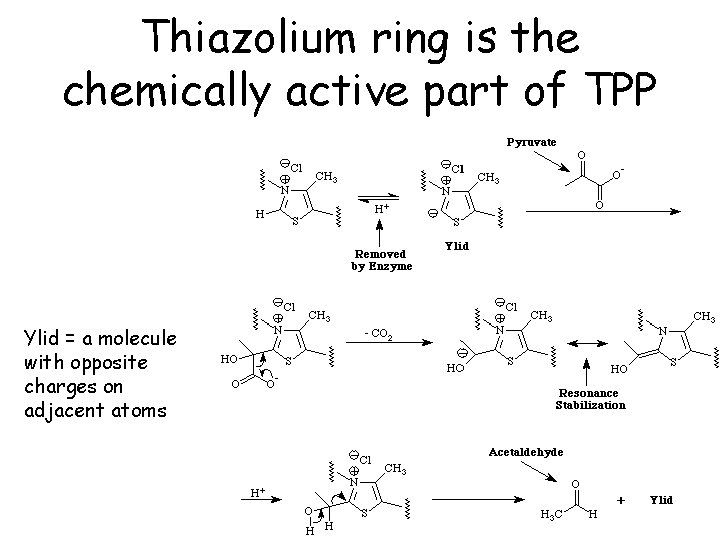
Thiazolium ring is the chemically active part of TPP Ylid = a molecule with opposite charges on adjacent atoms

Pyridoxal

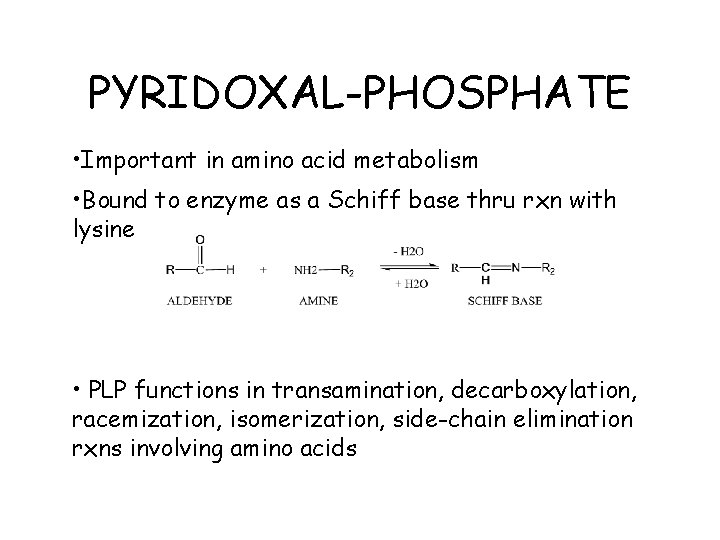
PYRIDOXAL-PHOSPHATE • Important in amino acid metabolism • Bound to enzyme as a Schiff base thru rxn with lysine • PLP functions in transamination, decarboxylation, racemization, isomerization, side-chain elimination rxns involving amino acids

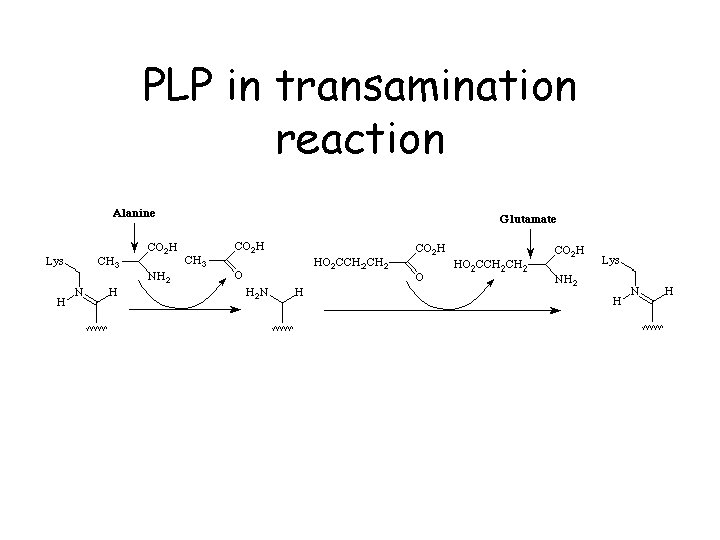
PLP in transamination reaction

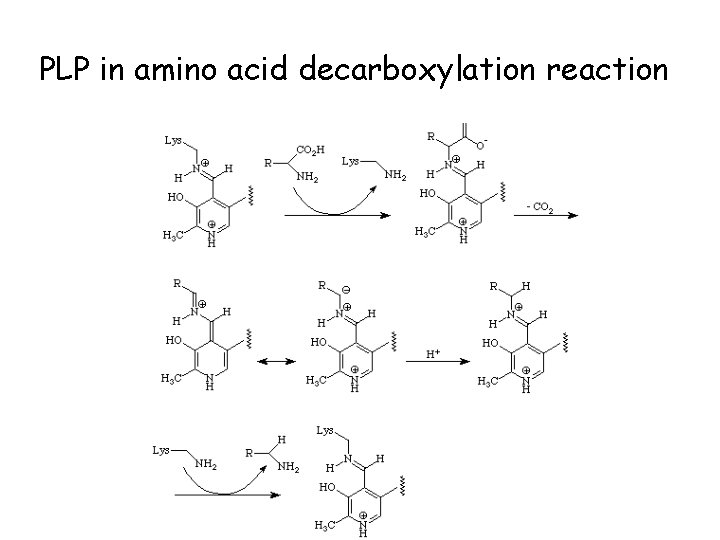
PLP in amino acid decarboxylation reaction

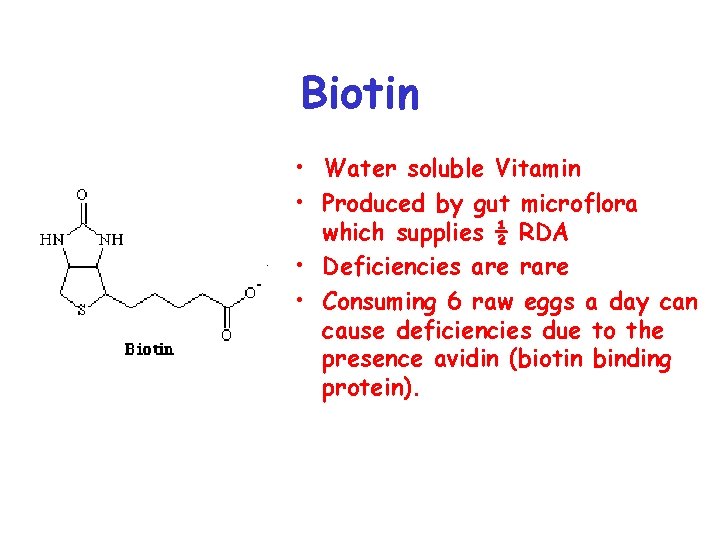
Biotin • Water soluble Vitamin • Produced by gut microflora which supplies ½ RDA • Deficiencies are rare • Consuming 6 raw eggs a day can cause deficiencies due to the presence avidin (biotin binding protein).

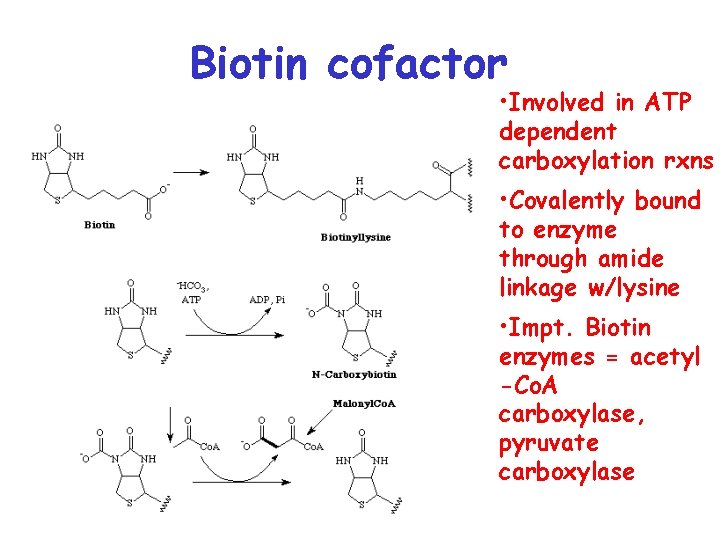
Biotin cofactor • Involved in ATP dependent carboxylation rxns • Covalently bound to enzyme through amide linkage w/lysine • Impt. Biotin enzymes = acetyl -Co. A carboxylase, pyruvate carboxylase

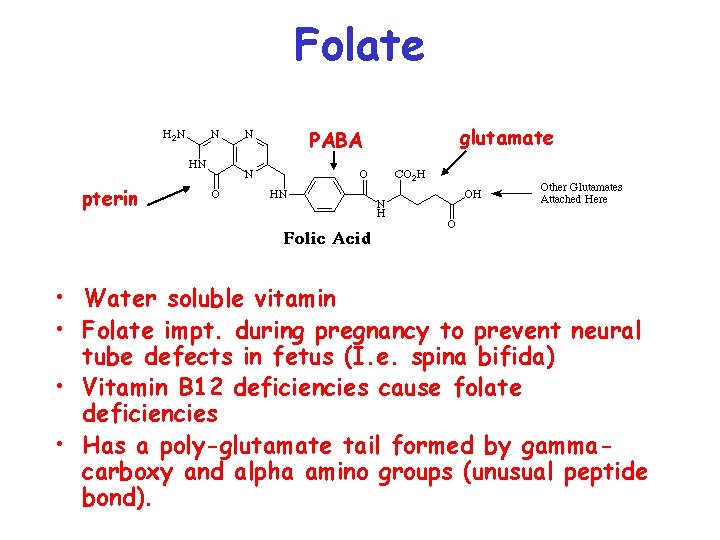
Folate PABA glutamate pterin • Water soluble vitamin • Folate impt. during pregnancy to prevent neural tube defects in fetus (I. e. spina bifida) • Vitamin B 12 deficiencies cause folate deficiencies • Has a poly-glutamate tail formed by gammacarboxy and alpha amino groups (unusual peptide bond).

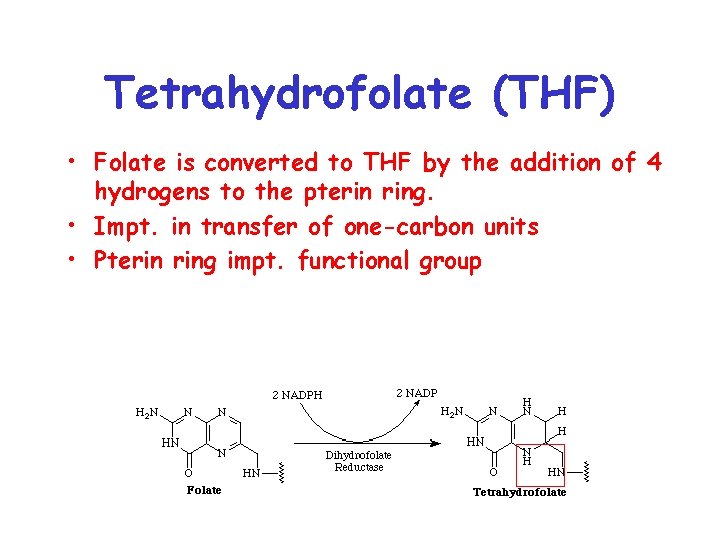
Tetrahydrofolate (THF) • Folate is converted to THF by the addition of 4 hydrogens to the pterin ring. • Impt. in transfer of one-carbon units • Pterin ring impt. functional group

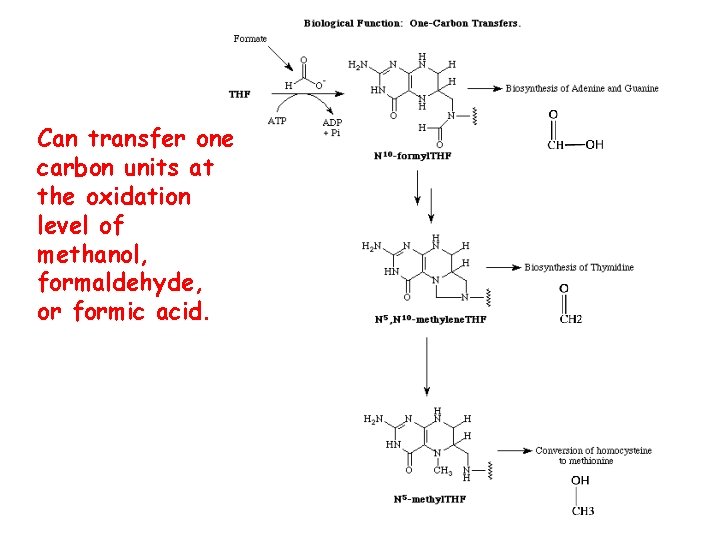
Can transfer one carbon units at the oxidation level of methanol, formaldehyde, or formic acid.

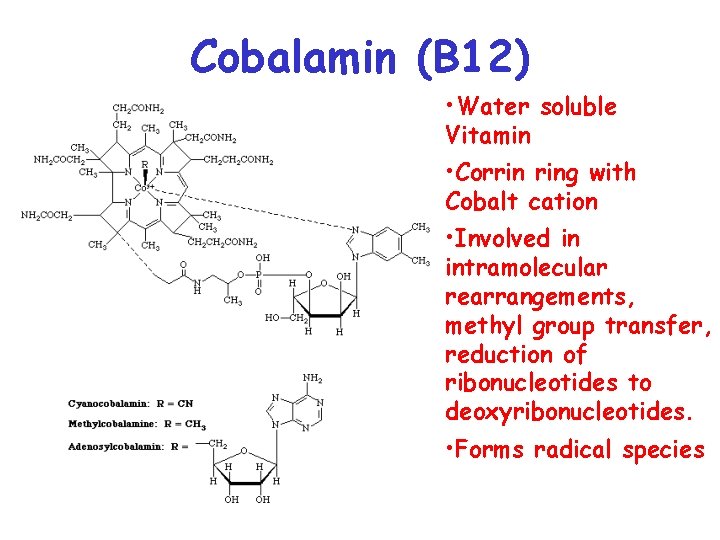
Cobalamin (B 12) • Water soluble Vitamin • Corrin ring with Cobalt cation • Involved in intramolecular rearrangements, methyl group transfer, reduction of ribonucleotides to deoxyribonucleotides. • Forms radical species

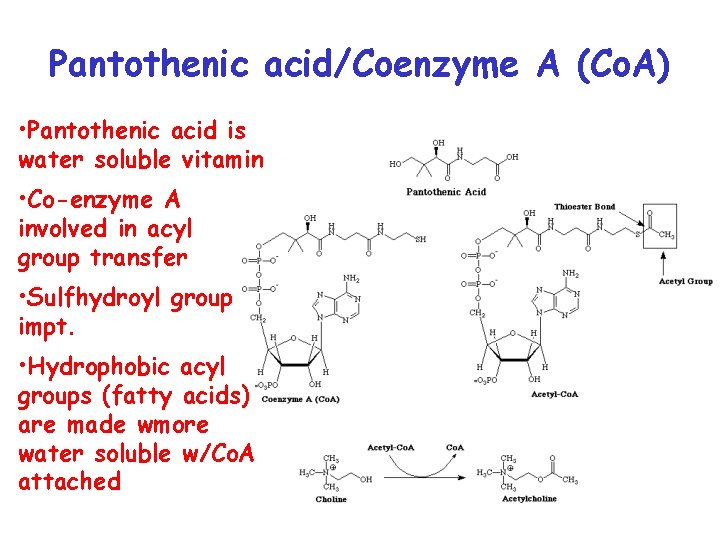
Pantothenic acid/Coenzyme A (Co. A) • Pantothenic acid is water soluble vitamin • Co-enzyme A involved in acyl group transfer • Sulfhydroyl group impt. • Hydrophobic acyl groups (fatty acids) are made wmore water soluble w/Co. A attached

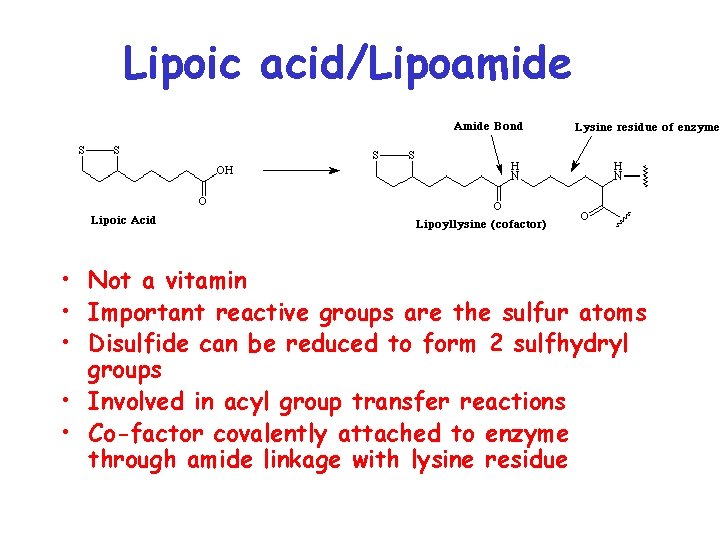
Lipoic acid/Lipoamide • Not a vitamin • Important reactive groups are the sulfur atoms • Disulfide can be reduced to form 2 sulfhydryl groups • Involved in acyl group transfer reactions • Co-factor covalently attached to enzyme through amide linkage with lysine residue

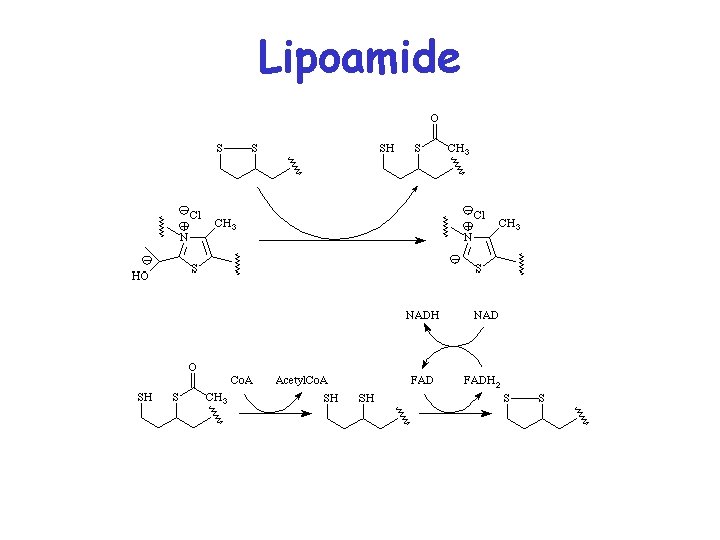
Lipoamide

Fat soluble Vitamins • Vitamin A (retinol) derived from bcarotene impt for vision, regulation of gene expression during cell differentiation, teratogenic • Vitamin D – impt in Ca absorption, regulates intestinal absorption and deposition in bones • Vitamin E – antioxidant

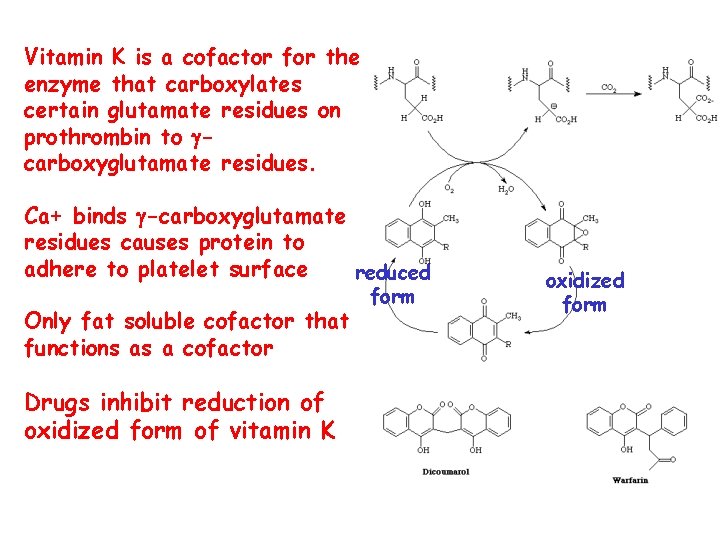
Vitamin K is a cofactor for the enzyme that carboxylates certain glutamate residues on prothrombin to gcarboxyglutamate residues. Ca+ binds g-carboxyglutamate residues causes protein to adhere to platelet surface reduced Only fat soluble cofactor that functions as a cofactor Drugs inhibit reduction of oxidized form of vitamin K form oxidized form

Ubiquinone/Plastoquinone • Lipid soluble electron carriers. • Impt in electron transport chains • Can accept or donate electrons one or two at a time (See page 223 of text)

Protein coenzymes • Usually small proteins • Active groups are either prosthetic groups or part of protein backbone • Participate in group transfer and oxidation/reduction rxns • acyl carrier protein • biotin carboxyl carrier protein

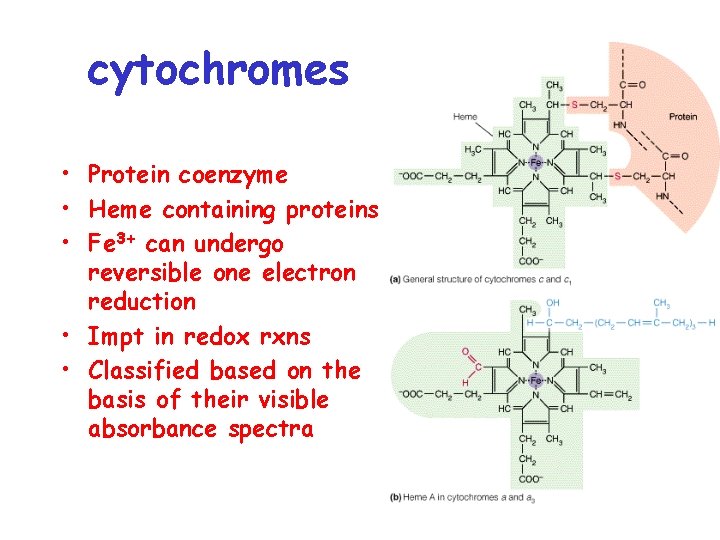
cytochromes • Protein coenzyme • Heme containing proteins • Fe 3+ can undergo reversible one electron reduction • Impt in redox rxns • Classified based on the basis of their visible absorbance spectra