Rocks and Minerals Minerals by def naturally occurring

Rocks and Minerals ÜMinerals (by def) - naturally occurring - inorganic - solid - definite chemical composition - a crystal structure

Rocks and Minerals 1. Naturally Occurring: not manufactured mineral: quartz pyrite Not mineral: cement, steel

Rocks and Minerals 2. Inorganic: - minerals not made by living things (organic) Ex: coal, pearls Amber (not minerals)

Rocks and Minerals 3. Chemical Composition- may be a single element or combination

MINERALS NATIVE ELEMENTS Gold (Au) Silver (Ag) Platinum (Pt) Diamond (C) Graphite (C) Sulfur (S) Copper (Cu) Gold Copper Silver
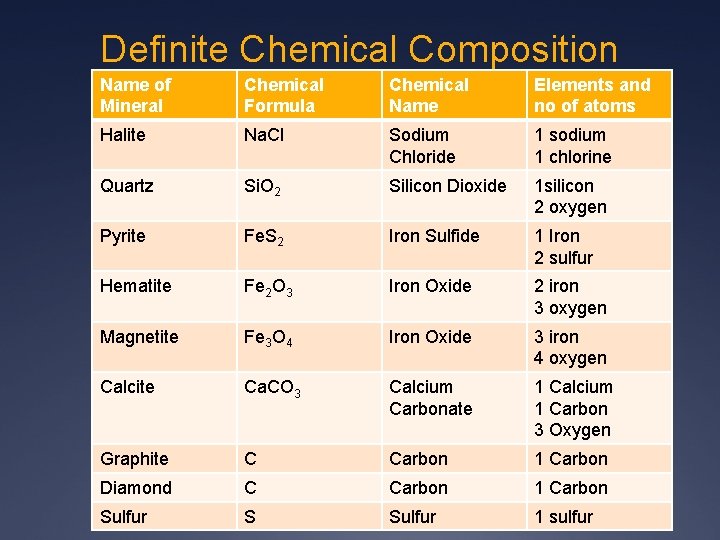
Definite Chemical Composition Name of Mineral Chemical Formula Chemical Name Elements and no of atoms Halite Na. Cl Sodium Chloride 1 sodium 1 chlorine Quartz Si. O 2 Silicon Dioxide 1 silicon 2 oxygen Pyrite Fe. S 2 Iron Sulfide 1 Iron 2 sulfur Hematite Fe 2 O 3 Iron Oxide 2 iron 3 oxygen Magnetite Fe 3 O 4 Iron Oxide 3 iron 4 oxygen Calcite Ca. CO 3 Calcium Carbonate 1 Calcium 1 Carbon 3 Oxygen Graphite C Carbon 1 Carbon Diamond C Carbon 1 Carbon Sulfur S Sulfur 1 sulfur

Rocks and Minerals 4. Solids: Have definite size/volume and a definite shape Ex: oil, lava, magma

Rocks and Minerals 5 Crystal Shape: Atoms arranged in repeating patterns. Atoms have specific arrangement or crystal structure

General Facts About Minerals 2, 000 + minerals have been identified Ü A few are “native elements” -- made of only one element, such as sulfur, gold. copper, and graphite (carbon) Ü Most are compounds, especially the silicate group (Si, O). (largest group) Ü Other important groups are oxides, carbonates, and sulfides.

MINERAL FORMATION Ü CRYSTALLIZE FROM MAGMA or LAVA solidification of liquid magma/lava to solid with a definite internal arrangement of atoms into a regular repeating pattern Ü PRECIPITATE FROM SOLUTION dissolved minerals come out of solution (water) to form solids

Identifying Minerals Identified by Physical and Chemical properties Physical: Color, Streak, Luster, Hardness, Density Chemical: reactivity, special properties

Minerals are identified by their Physical Properties Ü Crystal Form – determines physical properties Ü Color Ü Streak Ü Luster – metallic, non-metallic Ü Hardness – Mohs Hardness Scale (1 -10) Ü Cleavage/Fracture Ü Acid Test for carbonate minerals

Physical Properties: COLOR Ü Color is not usually a definitive property of a mineral. Ü Some minerals have characteristics colors Ü Others vary due to chemical differences or impurities (atoms mixed inside the main elements) Ü However most minerals have a variety of colors.

Physical Properties a. Some minerals have only one color - olivine: green - sulfur: yellow b. Others have many colors - quartz: clear, pink, purple, white, smoky - hematite: black, grey red-brown, dark red

Some Colors of Quartz

Physical Properties 2. Streak: color of powder when mineral is rubbed on a streak plate - Streak never changes Ex: Hematite: color varies Dark red, reddish brown, grey Streak: always reddish brown

STREAK Ü For opaque minerals, if you rub the sample across a streak plate, it will leave a colored powder. This streak is distinctive for minerals and is used to identify minerals.

Varieties of Hematite – all same color streak

LUSTER – Does this look like it could be made from a hard metal? Ü Describes how light reflects off the surface Ü Categories: Metallic or Non-metallic Metallic – luster of metal – shines like a hard metal (light does not penetrate) Many non-metallic minerals are SHINY because they are transparent or semi-transparent Ü Non-metallic: vitreous or glassy; silky; pearly; greasy; waxy, dull; earthy

Examples of metallic luster

Non-metallic and metallic luster – earthy hematite – metallic hematite

Example of non-metallic luster Ü Vitreous--quartz

Example of non-metallic luster Ü Silky--example plagioclase feldspar

More Examples of Metallic Luster Pyrite (Fe. S 2) Galena (Pb. S) PYRITE GALENA

4. HARDNESS – Mineral’s resistance to scratching or abrasion. Minerals with higher numbers will scratch minerals below
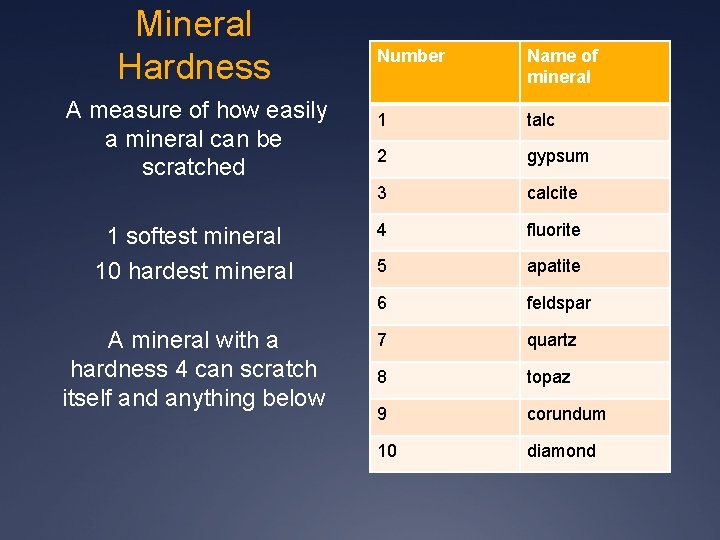
Mineral Hardness A measure of how easily a mineral can be scratched 1 softest mineral 10 hardest mineral A mineral with a hardness 4 can scratch itself and anything below Number Name of mineral 1 talc 2 gypsum 3 calcite 4 fluorite 5 apatite 6 feldspar 7 quartz 8 topaz 9 corundum 10 diamond

Mineral Hardness/Common Objects 2. 5 Fingernail 3. 5 Copper Penny 4. 5 Iron Nail 5. 5 Glass Plate 6. 5 Steel File 7 Streak Plate

5. Cleavage and Fracture Ü Some minerals split along flat surfaces (called cleavage planes) when struck hard--this is called mineral cleavage Ü Other minerals break unevenly along rough or curved surfaces-this is called fracture

a. Cleavage: when a mineral splits along smooth flat surfaces. - Ex: Mica: one direction

Ex: Galena: three directions Determined by internal structure of mineral bonds between the atoms

What Determines Cleavage – weak bonds in the crystal structure Halite (Na. Cl) Fluorite (Ca. F 2) HALITE FLUORITE

Cleavage does not = Crystal Shape Crystal way mineral grows Cleavage way mineral breaks


Cleavage BIOTITE MUSCOVITE

Fracture: mineral breaks unevenly curved or irregular pieces rough/jagged surfaces
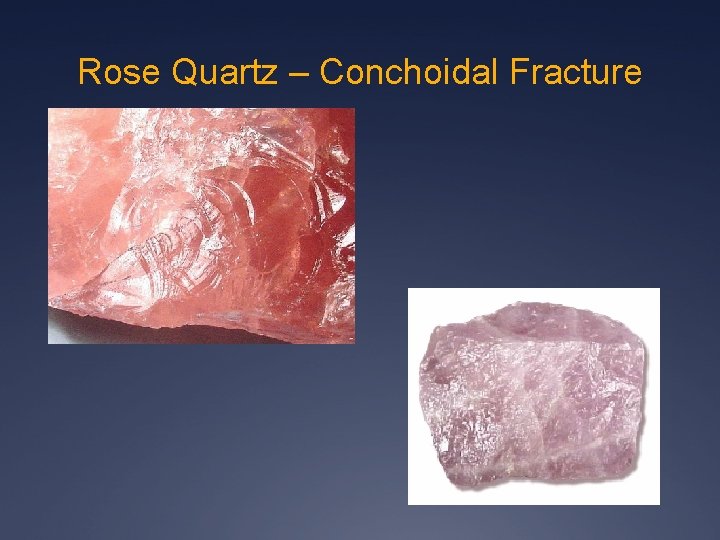
Rose Quartz – Conchoidal Fracture

Conchoidal Fracture - Quartz Obsidian
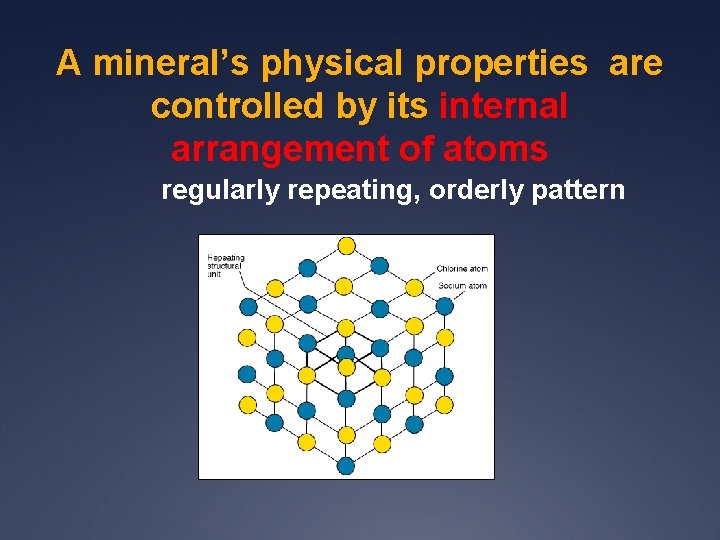
A mineral’s physical properties are controlled by its internal arrangement of atoms regularly repeating, orderly pattern

Minerals can have the same chemical composition (Carbon) but different physical properties because of their crystal structure Diamond Graphite

Chemical Properties Calcite: reacts with HCL. It forms bubbles of carbon dioxide gas Ca. CO 3 + 2 HCl -> Ca. Cl 2 + H 2 O + CO 2

Special Properties Lodestone, a form of the mineral magnetite is naturally magnetic Calcite, shows double refraction Pitchblende: is radioactive

Uses of Minerals A. Ore: A mineral that contains Metals and nonmetals that can be removed in usable amounts for a profit. 1. Metals: elements shiny surfaces conduct heat and electricity

Metals 1. Iron: hematite: Fe 2 O 3 Magnetite: Fe 3 O 4 2. Aluminum: bauxite: Al(OH)3 3. Copper: Chalcopyrite: Cu. Fe. S 2 malachite: Cu 2 CO 3(OH)2

Nonmetals Elements dull surfaces, poor conductors of heat, electricity 1. Halite: Salt: Na. CL 2. Gypsum: drywall 3. Sulfur: matches 4. Talc: talcum powder 5. Graphite: pencils

Gems Minerals with desirable qualities hardness, color, luster, rare, durable Precious stones: diamonds, rubies sapphires Semiprecious: amethyst, garnet topaz Gems not minerals: pearls

7 Mineral compositions Ü Silicates Ü Carbonates Ü Sulfates Ü Halides Ü Oxides Ü Sulfides Ü Native Elements

Silicates ÜA Silicon atom surrounded by 4 oxygen atoms ÜExamples ÜOlivine ÜQuartz
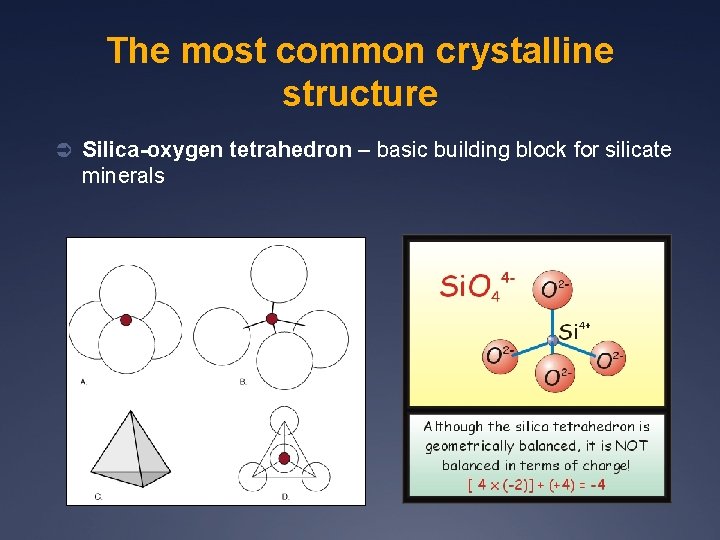
The most common crystalline structure Ü Silica-oxygen tetrahedron – basic building block for silicate minerals

Carbonates ÜContain a Carbon atom with 3 Oxygen atoms ÜExamples ÜDolomite ÜCalcite

NONSILICATE MINERALS CARBONATES Minerals are soft (3 -4). Minerals are soluble in acidic water. Leads to cave development. Calcite (Ca. CO 3) if transparent can Break light into 2 images (double refraction) CALCITE

Acid Test for Carbonates Ü Special Characteristics: Carbonates react with dilute HCl and other acids by fizzing or bubbling (releasing CO 2 gas)

Oxides ÜOxygen and compounds of certain elements ÜExample ÜMagnetite ÜHematite

Sulfides ÜSulfur and compounds of certain elements ÜExample ÜGalena ÜPyrite

Halides ÜA Chlorine, Fluorine, Iodine, or Bromine ion and compounds of certain elements ÜExample ÜHalite ÜFluorite

Sulfates Ü Contain a Sulfur atom with 4 Oxygen atoms Ü Many sulfates form out of precipitation out of water at or near the Earth’s surface Ü Example Ü Gypsum

Less than a dozen are common in most rocks Ü Quartz Pyrite Ü Feldspar (group) Ü Muscovite (silver white mica) Ü Biotite (black mica) Olivine Ü Calcite Amphibole Ü Pyrite Pyroxene Ü Magnetite, limonite, and other iron oixdes
- Slides: 56