GAS LAWS KINETIC MOLECULAR THEORY Particles in an





















- Slides: 26

GAS LAWS

KINETIC MOLECULAR THEORY Particles in an ideal gas… have no volume. have elastic collisions. are in constant, random, straight-line motion. don’t attract or repel each other.

REAL GASES Particles in a REAL gas… have their own volume attract each other Gas behavior is most ideal… at low pressures at high temperatures in nonpolar atoms/molecules

CHARACTERISTICS OF GASES Gases expand to fill any container. random motion, no attraction Gases are fluids (like liquids). They flow. CO 2 demo Cats are Fluids: no attraction Gases have very low densities. They flow and fill the shape of their container

CHARACTERISTICS OF GASES Gases can be compressed. no volume = lots of empty space Gases undergo diffusion random motion

CHARACTERISTICS OF GASES

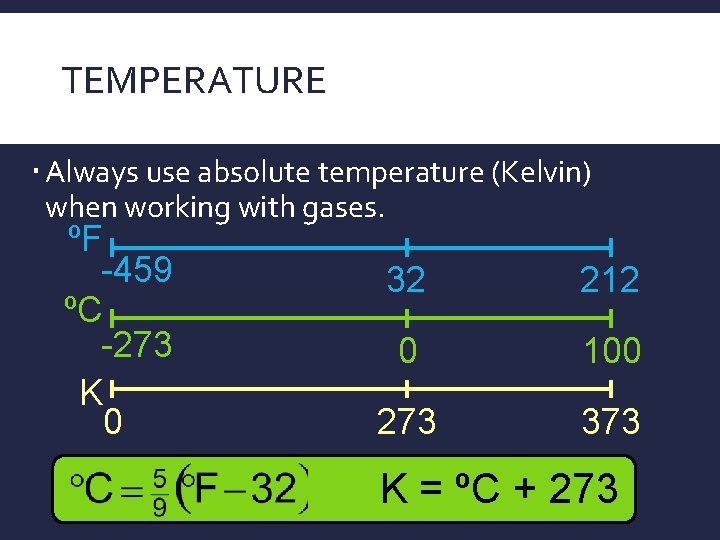
TEMPERATURE Always use absolute temperature (Kelvin) when working with gases. ºF -459 ºC -273 K 0 32 212 0 100 273 373 K = ºC + 273

PRESSURE Which shoes create the most pressure?

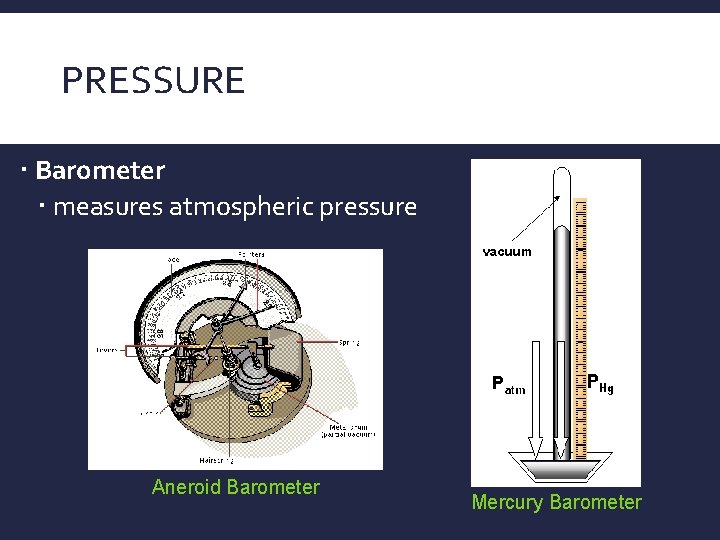
PRESSURE Barometer measures atmospheric pressure Aneroid Barometer Mercury Barometer

PRESSURE KEY UNITS AT SEA LEVEL 101. 325 k. Pa (kilopascal) 1 atm 760 mm Hg 760 torr 14. 7 psi

STP Standard Temperature & Pressure 1 atm 273 K

BOYLE’S LAW

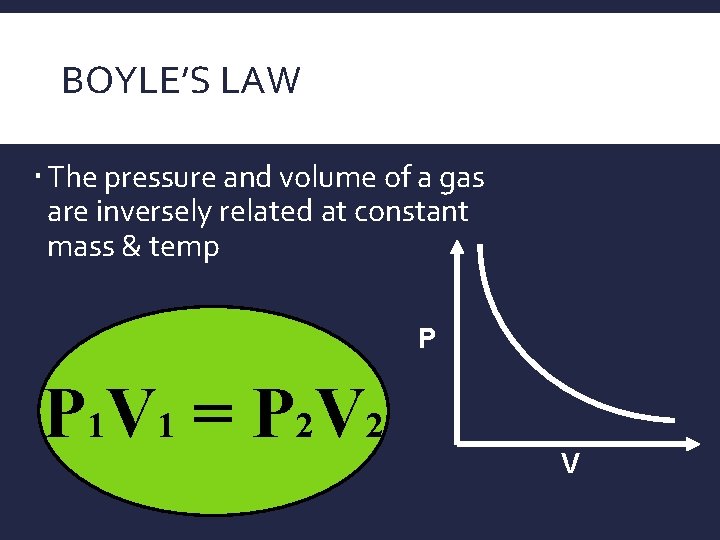
BOYLE’S LAW The pressure and volume of a gas are inversely related at constant mass & temp P P 1 V 1 = P 2 V 2 V

PRACTICE QUESTION If a Helium tank that has an initial volume of 100 m. L and a pressure of 5 atm loses 50 m. L of Helium what is its new pressure? (5)(100) = P 2 (50) 500/50 = 10 P 2 = 10 atm

CHARLES’ LAW

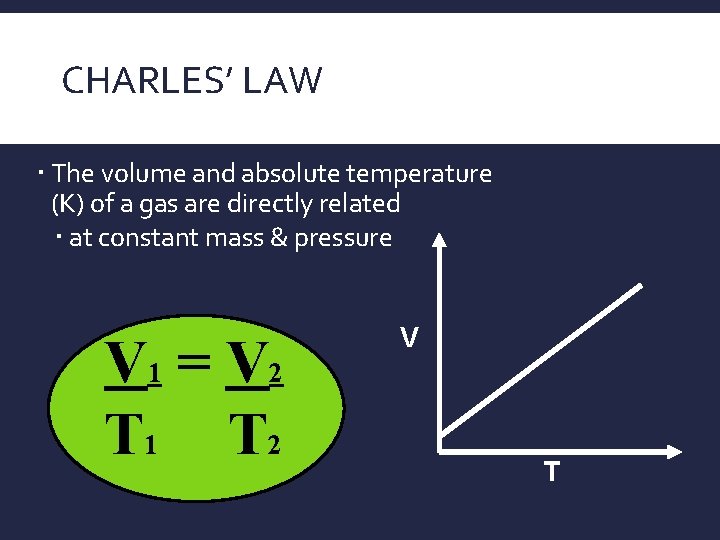
CHARLES’ LAW The volume and absolute temperature (K) of a gas are directly related at constant mass & pressure V 1 = V 2 T 1 T 2 V T

PRACTICE QUESTION If a Helium balloon that has an intial volume of 100 m. L and a temperature of 240 K is placed in the sun where it heats to 300 K what is its new volume? (100) = V 2 (240) (300) 0. 42 = V 2 (300) V 2 = 125 m. L

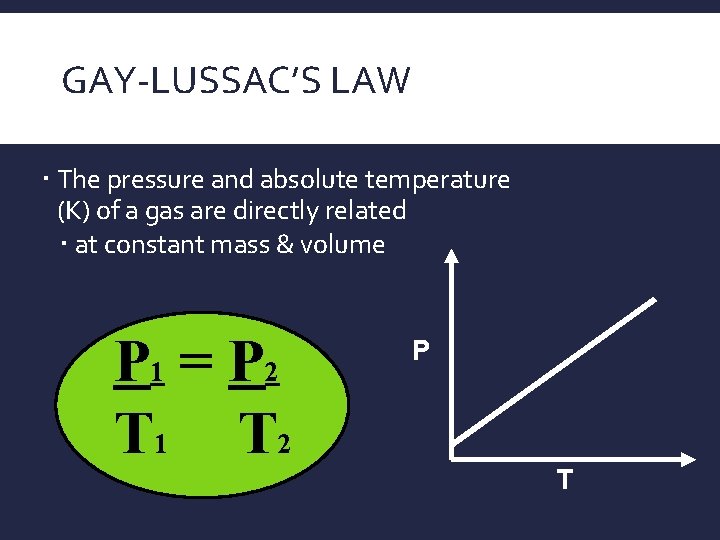
GAY-LUSSAC’S LAW

GAY-LUSSAC’S LAW The pressure and absolute temperature (K) of a gas are directly related at constant mass & volume P 1 = P 2 T 1 T 2 P T

PRACTICE QUESTION If a Helium tank that has an initial pressure of 5 atm and a temperature of 240 K is placed in the sun where it heats to 300 K what is the new pressure? (5) = P 2 (240) (300) 0. 021 = P 2 (300) P 2 = 6. 25 atm

DALTON’S LAW The total pressure of a mixture of gases equals the sum of the partial pressures of the individual gases. Ptotal = P 1 + P 2 +. . .

PRACTICE QUESTION A scuba tank is filled with a mixture of atmospheric gases. Nitrogen exerts a pressure of 70 atm. Oxygen gas exerts a pressure of 22 atm. Argon exerts a pressure of 4 atm. A fourth gas exerts an unknown amount of pressure. We know the total pressure of the tank is 98 atm. What is the partial pressure of the fourth gas? Ptotal = P 1 + P 2 + P 3 + P 4 98 = 70 +22 + 4 + p 4 98 = 96 + P 4 98 -96 = P 4 2 = P 4

IDEAL GAS LAW PV=n. RT UNIVERSAL GAS CONSTANT R=0. 0821 L atm/mol K 3 R=8. 315 dm k. Pa/mol K

PRACTICE QUESTION How many moles of gas does it take to occupy 120 L at a pressure of 2. 3 atm and a temperature of 340 K? PV = n. RT (2. 3)(120) = n (0. 0821)(340) 276 = n(27. 914) 276/(27. 914) = n n = 9. 89 moles

GAS STOICHIOMETRY Moles Liters of a Gas STP - use 22. 4 L/mol Non-STP - use ideal gas law

PRACTICE QUESTION You are given 1 L of carbon monoxide at STP, how many moles of carbon monoxide do you have? 1 L x 1 mol = 0. 045 mol carbon monoxide 22. 4 L