ERT 1083 PHYSICAL CHEMISTRY SECOND LAW OF THERMODYNAMICS





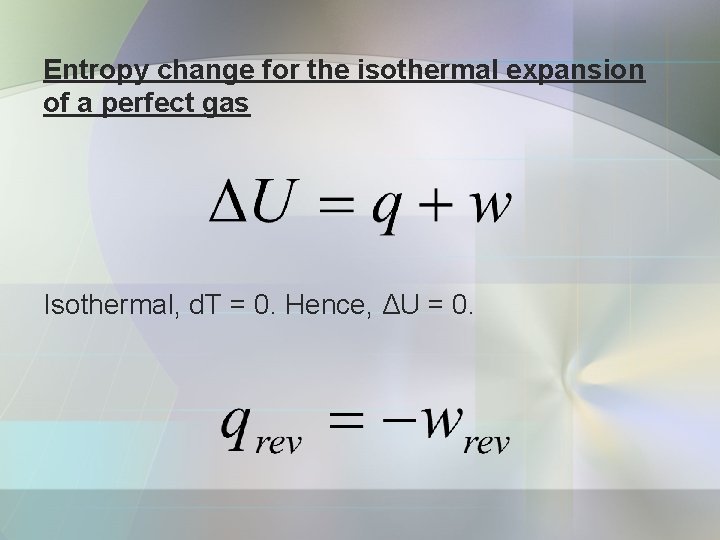



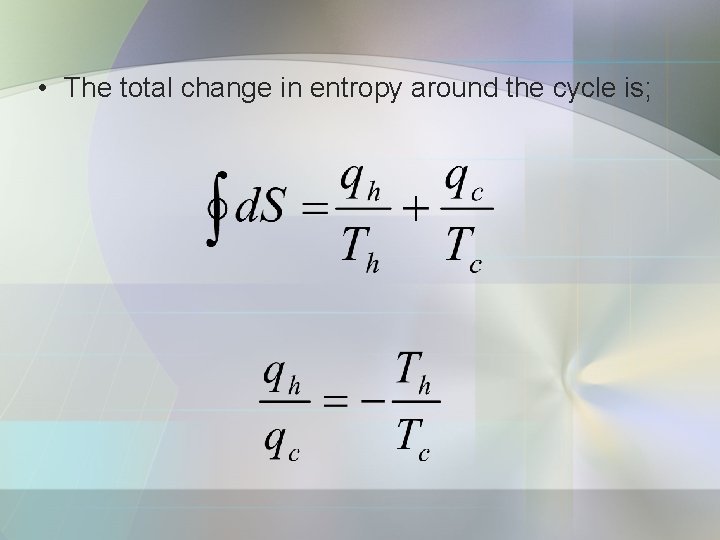





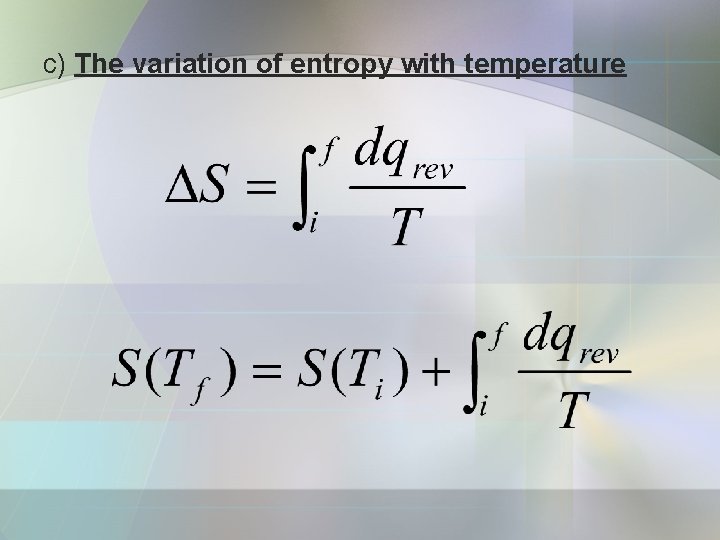







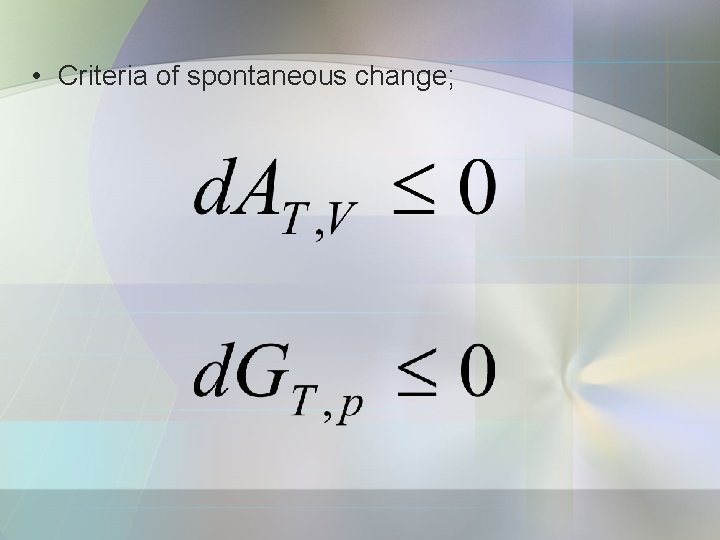



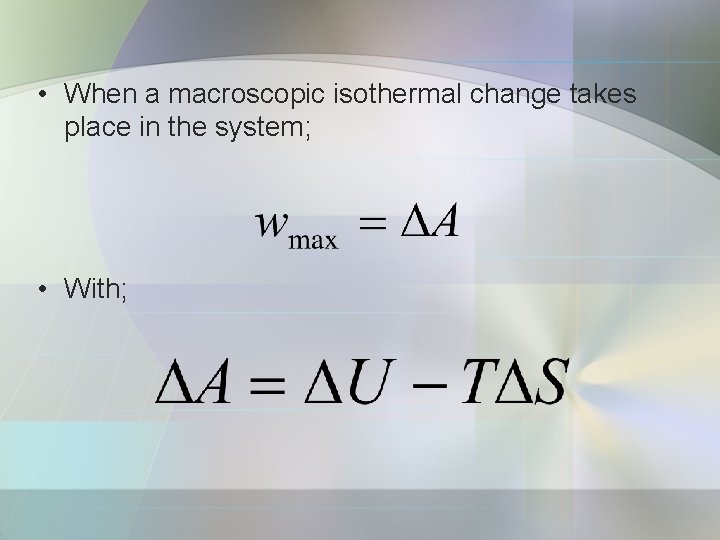



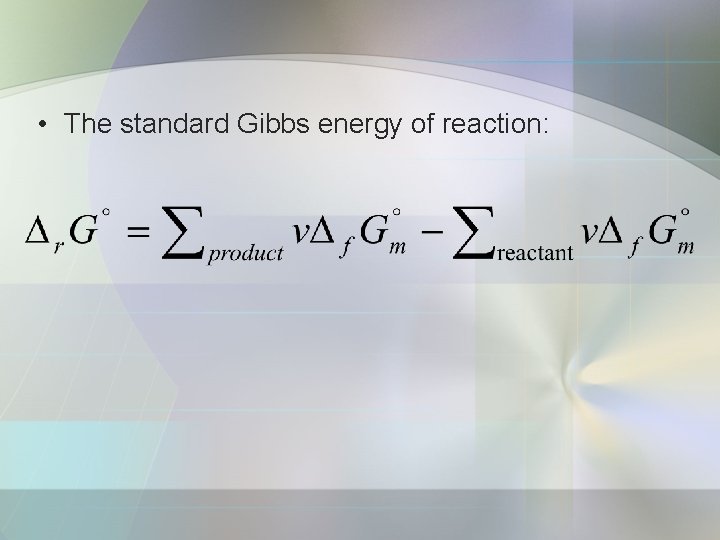
- Slides: 36

ERT 108/3 PHYSICAL CHEMISTRY SECOND LAW OF THERMODYNAMICS Prepared by: Pn. Hairul Nazirah Abdul Halim

About This Chapter • To explain the origin of the spontaneity of physical and chemical change. • • Heat engines Entropy Calculation of entropy changes Helmholtz and Gibbs energies

The direction of spontaneous change • What is the direction of spontaneous change of the following reaction? ½ N 2 + 3/2 H 2 NH 3 • Answer to this question is obtained by calculating the entropy change in the system and the surroundings.

Entropy • Second Law of Thermodynamics use entropy, S to identify the spontaneous changes. • The entropy of an isolated system increases in the course of a spontaneous change: • Where ΔStot = total entropy of the system and its surroundings.

• Thermodynamic definition of entropy Unit of entropy: J K-1 Unit of molar entropy: J K-1 mol-1
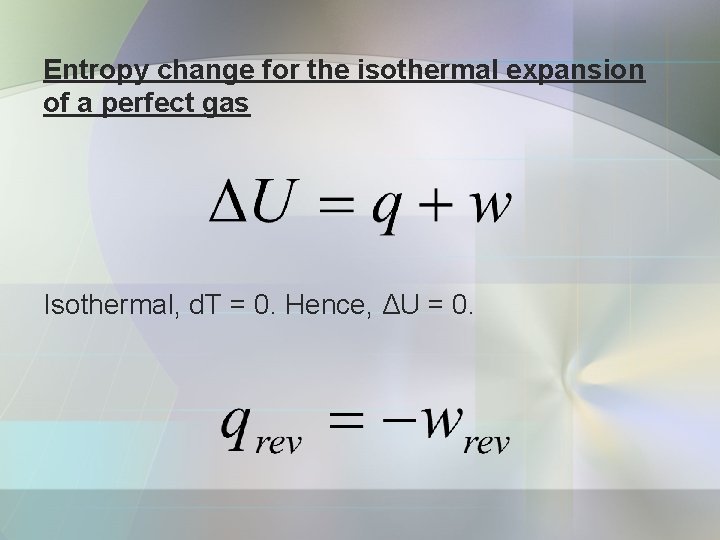
Entropy change for the isothermal expansion of a perfect gas Isothermal, d. T = 0. Hence, ΔU = 0.

Because the temp. is constant;

Heat Engines Automobile engine • Automobile engine operates in a cyclic process of fuel intake, compression, ignition and expansion, and exhaust. • Occurs several thousand times per minutes. • Is used to perform work on the surroundings. • The system consists of piston & cylinder assembly with diathermal walls. • The expansion and contraction of the gas caused by changes in its temperature drives the piston in or out of cylinder.

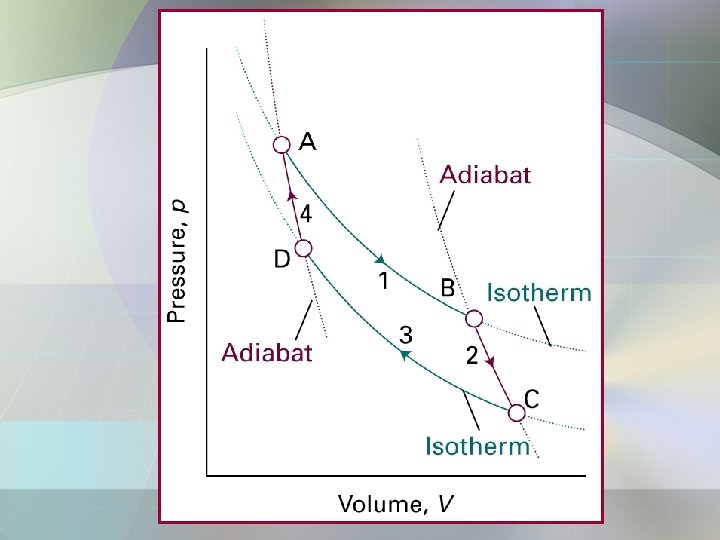
A reversible Carnot Cycle consist of 4 reversible stages: 1. Reversible isothermal expansion from A to B. d. S = qh/Th. 2. Reversible adiabatic expansion from B to C. d. S = 0. 3. Reversible isothermal compression from C to D. d. S = qc/Tc 4. Reversible adiabatic compression from D to A. d. S = 0.

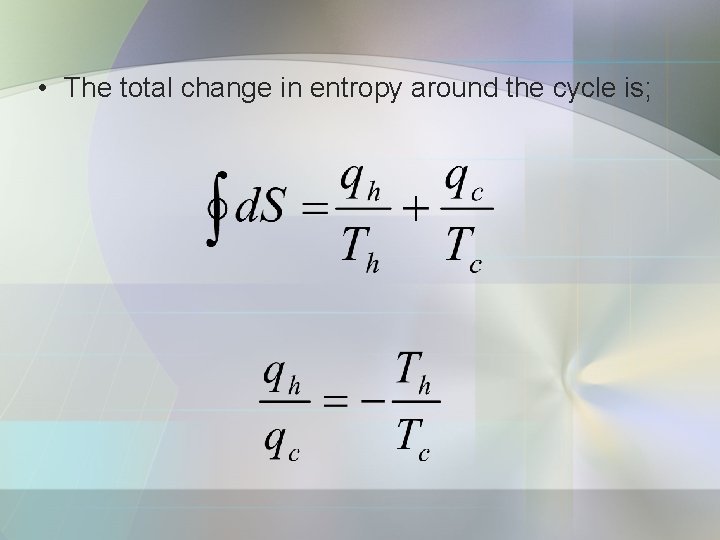
• The total change in entropy around the cycle is;

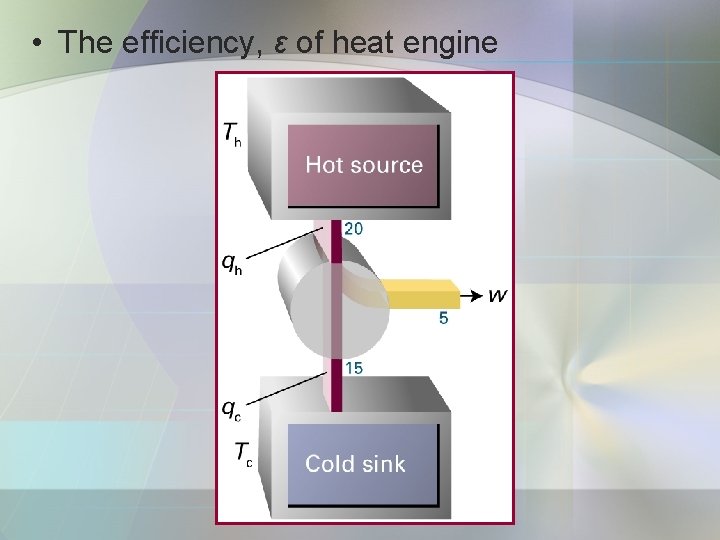
• The efficiency, ε of heat engine

• The efficiency, ε of heat engine:

c) Thermodynamic Temperature Thermodynamic temperature scale is defined as;

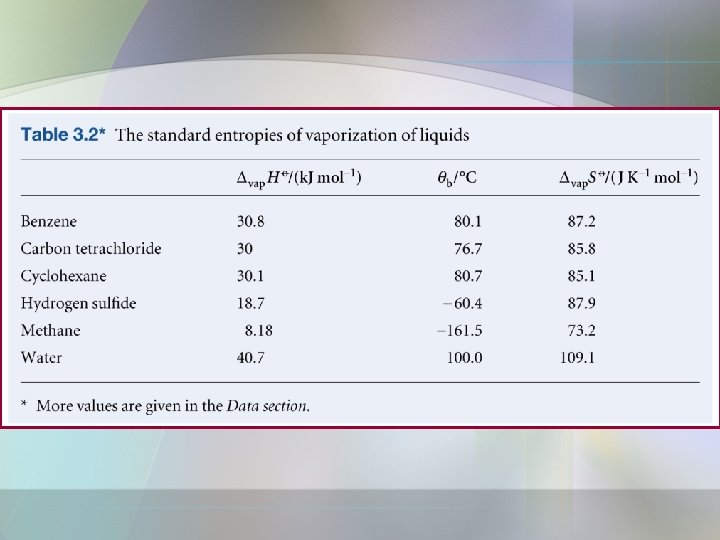
Entropy Changes a) Entropy change of phase transition at the transition temp. • • Phase transition such as solid melts to liquid, liquid phase turns into a gas. At constant pressure; • The change in molar entropy of the system;



b) The expansion of a perfect gas The change in entropy of a perfect gas that expands isothermally from Vi to Vf is;
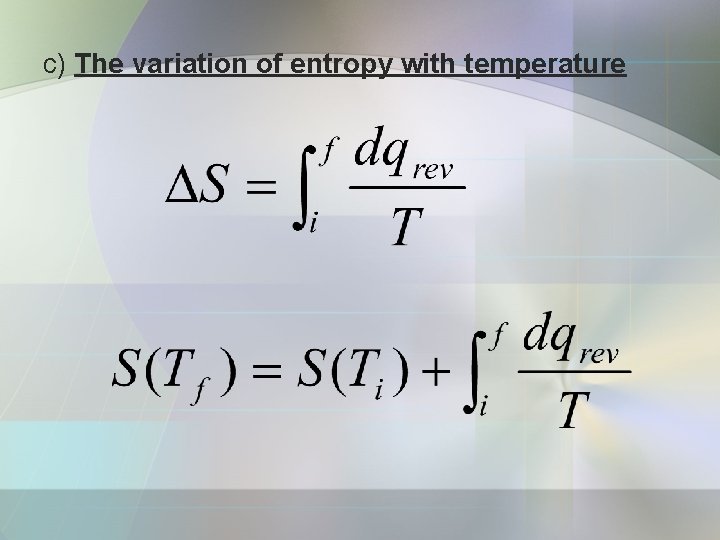
c) The variation of entropy with temperature

Constant pressure heat capacity; Hence, at constant pressure;

d) The measurement of entropy If a substance melts at Tf and boils at Tb, then its entropy above its boiling temperature is;

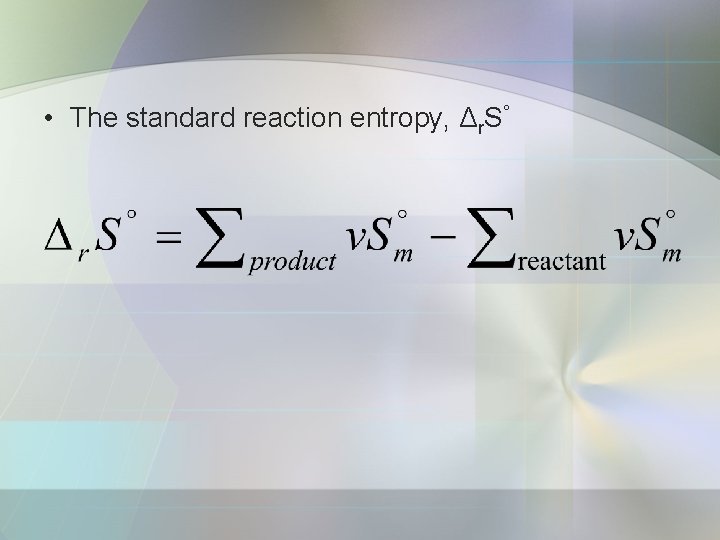
• The standard reaction entropy, Δr. S°

The Helmholtz and Gibss Energies • Consider a system in thermal equilibrium with its surroundings at a temperature T. • When a change in the system occurs and there is a transfer of energy as heat between system and its surrounding;

• Criteria for spontaneity Consider heat transfer at constant volume, dqv = d. U; At constant V and no additional work;

• When heat is transferred at constant pressure, dqp = d. H;

• Helmholtz energy, A is defined as; • When the states of the system changes at constant temp. ;

• Gibbs energy, G is defined as; • When the states of the system changes at constant temp. ;
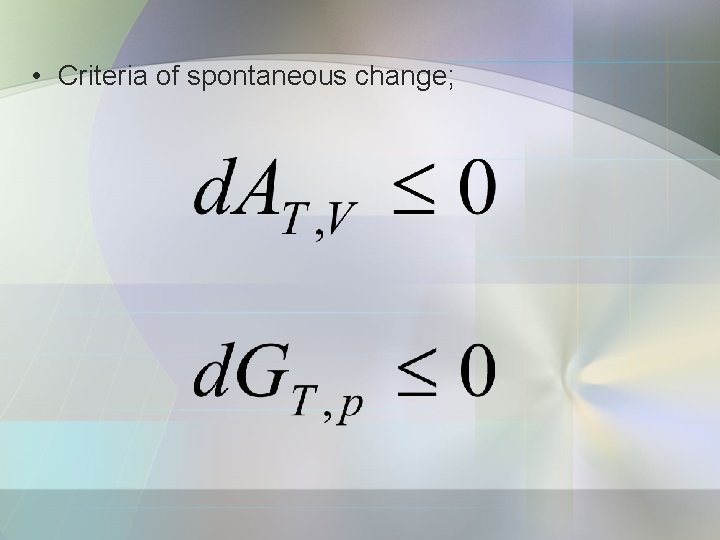
• Criteria of spontaneous change;

b) Some remarks on the Helmholz energy • A change in system at constant temp. and volume is spontaneous if. • The criterion of equilibrium,

& Both of the above equations are interpreted as follows: • –ve value of d. A is favoured by –ve value of d. U and +ve value of Td. S.

c) Maximum work The change in the Helmholtz function is equal to the maximum work accompanying a process; A sometimes called the ‘maximum work function’ or ‘work function’.
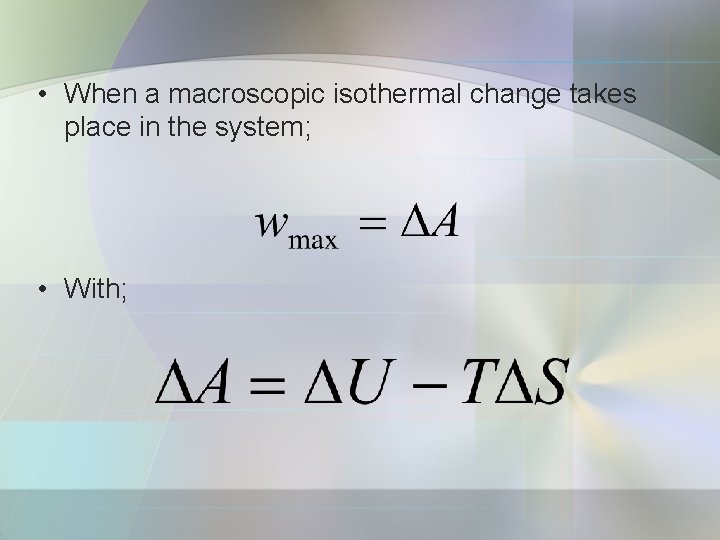
• When a macroscopic isothermal change takes place in the system; • With;

c) Some remarks on the Gibbs energy • • Gibbs energy = free energy At constant pressure and temperature, chemical reactions are spontaneous in the direction of decreasing Gibbs energy. If G decreases as the reaction proceeds: spontaneous tendency to convert reactants to products. If G increases, the reverse reaction is spontaneous.

e) Maximum additional (non-expansion) work Wad, max = Maximum additional (non-expansion) work, is given by the change in Gibbs energy;

Standard Molar Gibbs Energies • The standard entropies and enthalpies of reaction can be combined to obtain the standard Gibbs energy of reaction, Δr. G°;
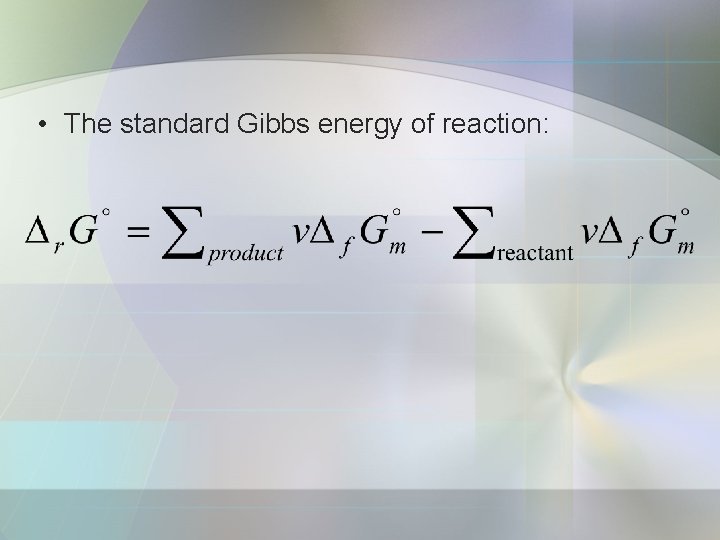
• The standard Gibbs energy of reaction: