ERT 1083 PHYSICAL CHEMISTRY Chemical Kinetics By Mrs

ERT 108/3 : PHYSICAL CHEMISTRY Chemical Kinetics By; Mrs Hafiza Binti Shukor ERT 108/3 PHYSICAL CHEMISTRY SEM 2 (2010/2011)

TOPIC COVERED…; Experimental Chemical and Kinetics Reactions First Order Reactions Second Order Reactions Reaction Rates and Reaction Mechanisms Light Spectroscopy and Adsorption Chemistry (Experimental methods for fast reactions). ERT 108/3 PHYSICAL CHEMISTRY SEM 2 (2010/2011)

CHEMICAL KINETICS? ? Also called reaction kinetics Study of the rates & mechanisms of chemical reactions 2 types of reaction; a)homogeneous – reaction occurs in 1 phase (gas @liquid phase) b)heterogeneous – reaction occurs ERT 108/3 PHYSICAL CHEMISTRY SEM 2 (2010/2011)

Experimental Chemical and Kinetics Reactions Rates of chemical Reactions: Ø the rate of speed with which a reactant disappears or a product appears. Ø the rate at which the concentration of one of the reactants decreases or of one of the products increases with time. Ø mol L-1 s-1. ERT 108/3 PHYSICAL CHEMISTRY 4 SEM 2 (2010/2011)

Example 1 • The decomposition of dinitrogen pentoxide (N 2 O 5) in an inert solvent (carbon tetrachloride) at 450 C: • The data of the formation of O 2(g) and the disappearance of N 2 O 5 is shown in Table 1. • The initial concentration [N 2 O 5] = 1. 40 M. What is the concentration, [N 2 O 5] at time, t=423 s? ERT 108/3 PHYSICAL CHEMISTRY 5 SEM 2 (2010/2011)
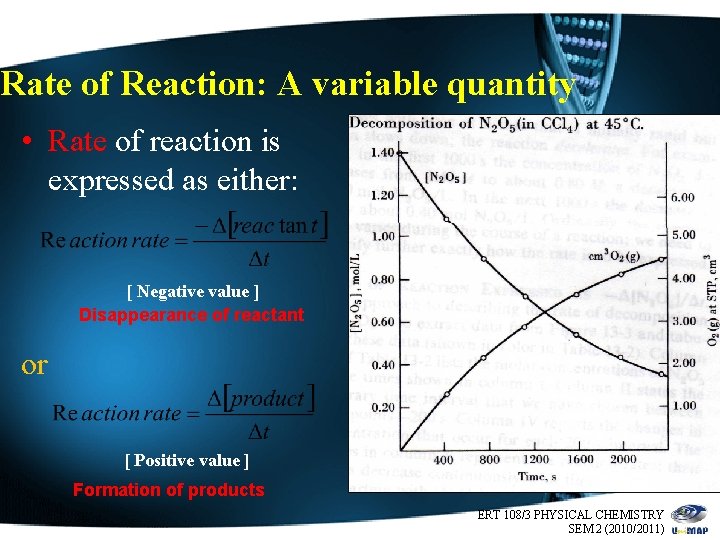
Rate of Reaction: A variable quantity • Rate of reaction is expressed as either: [ Negative value ] Disappearance of reactant or [ Positive value ] Formation of products ERT 108/3 PHYSICAL CHEMISTRY 6 SEM 2 (2010/2011)
![Answer (Example 1) • At t=0, Initial [N 2 O 5] = 1. 40 Answer (Example 1) • At t=0, Initial [N 2 O 5] = 1. 40](http://slidetodoc.com/presentation_image_h2/703897c1a8f4e78c92ffb5a03b52022d/image-7.jpg)
Answer (Example 1) • At t=0, Initial [N 2 O 5] = 1. 40 M • At t = ∞, Final [N 2 O 5] = 0 M [decomposes completely] • 5. 93 cm 3 O 2(g) is obtained at STP. • After 423 s, the volume of O 2 (g) collected is 1. 32 cm 3 of a possible 5. 93 cm 3. • The fraction of the N 2 O 5 decomposed is 1. 32/5. 93. • The decrease in concentration of N 2 O 5 at this point = (1. 32/5. 93) x 1. 40 M = 0. 312 M. • After 423 s, [N 2 O 5] remaining undecomposed = 1. 40 -0. 31 = 1. 09 M. ERT 108/3 PHYSICAL CHEMISTRY 7 SEM 2 (2010/2011)
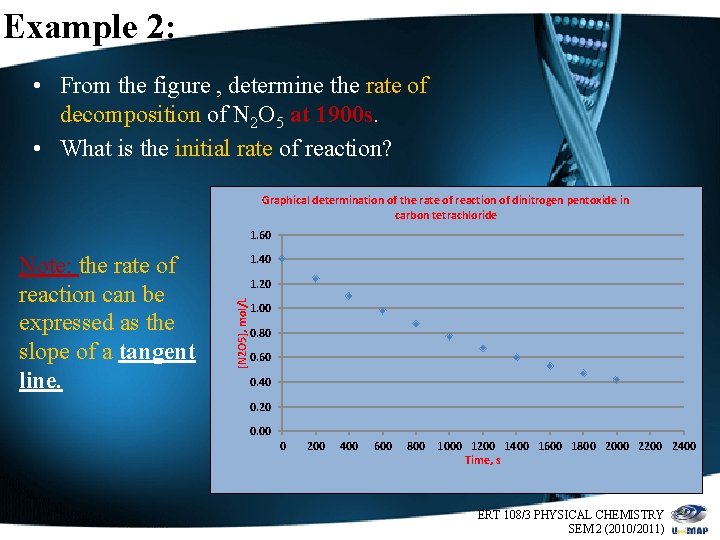
Example 2: • From the figure , determine the rate of decomposition of N 2 O 5 at 1900 s. • What is the initial rate of reaction? Graphical determination of the rate of reaction of dinitrogen pentoxide in carbon tetrachloride 1. 60 1. 40 1. 20 [N 2 O 5], mol/L Note: the rate of reaction can be expressed as the slope of a tangent line. 1. 00 0. 80 0. 60 0. 40 0. 20 0. 00 0 200 400 600 800 1000 1200 1400 1600 1800 2000 2200 2400 Time, s ERT 108/3 PHYSICAL CHEMISTRY 8 SEM 2 (2010/2011)
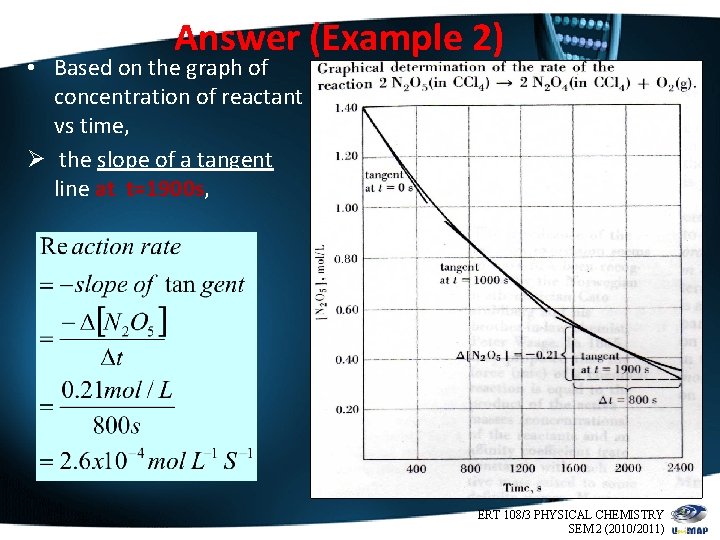
Answer (Example 2) • Based on the graph of concentration of reactant vs time, Ø the slope of a tangent line at t=1900 s, ERT 108/3 PHYSICAL CHEMISTRY 9 SEM 2 (2010/2011)
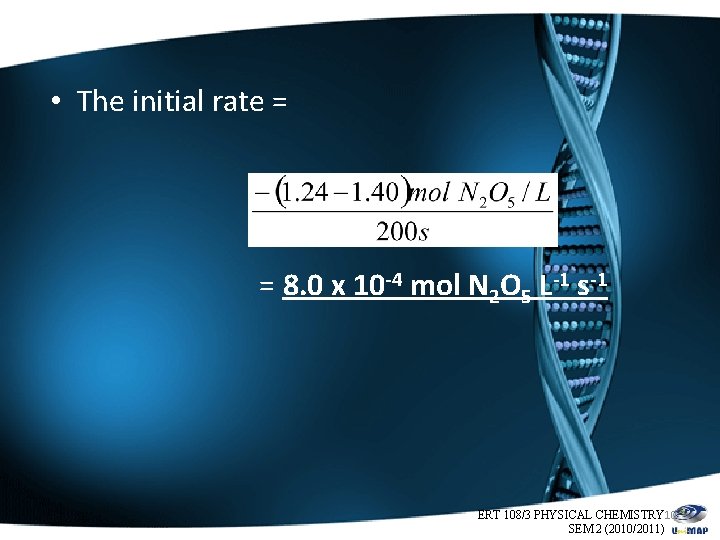
• The initial rate = = 8. 0 x 10 -4 mol N 2 O 5 L-1 s-1 ERT 108/3 PHYSICAL CHEMISTRY 10 SEM 2 (2010/2011)

The Rate Law for Chemical Reactions • The rate law or rate equation – mathematical equation. Reaction rate, r = k[A]m[B]n …. . Ø The rate, r at time t is experimentally found to be related to the concentrations of species present at that time, t. Ø The exponents in the rate reaction are called the order of the reaction. Ø The term k in the equation is called the rate constant. Ø it is a proportionality constant that is characteristic of the particular reaction & is significantly dependent only on temperature. ERT 108/3 PHYSICAL CHEMISTRY 11 SEM 2 (2010/2011)

Method of Initial Rates • This simple method of establishing the exponents in a rate equation involves measuring the initial rate of reaction for different sets of initial concentration. ERT 108/3 PHYSICAL CHEMISTRY 12 SEM 2 (2010/2011)

Example 3 • The data of three reactions involving S 2 O 82 and I- were given in the below table. (i) Use the data to establish the order of reaction with respect to S 2 O 82 -, the order with respect to I- & the overall order. ERT 108/3 PHYSICAL CHEMISTRY 13 SEM 2 (2010/2011)

Cont…Example 3 (ii) Determine the value of k for the above reaction. (iii) What is the initial rate of disappearance of S 2 O 82 - reaction in which the initial concentrations are [S 2 O 82 - ] =0. 050 M & [I-]=0. 025 M? (iv) What is the rate of formation of SO 42 - in Experiment 1? ERT 108/3 PHYSICAL CHEMISTRY 14 SEM 2 (2010/2011)
![Answer (Example 3) (i) In the experiments 1 & 2, [I-] is held constant Answer (Example 3) (i) In the experiments 1 & 2, [I-] is held constant](http://slidetodoc.com/presentation_image_h2/703897c1a8f4e78c92ffb5a03b52022d/image-15.jpg)
Answer (Example 3) (i) In the experiments 1 & 2, [I-] is held constant & [S 2 O 82 -] is increased by a factor of 2, from 0. 038 to 0. 076 M. Ø The reaction rate, R increased by a factor of 2 also. R 2 = k (0. 076)m(0. 060)n = k (2 x 0. 038)m(0. 060)n = k (2)m (0. 038)m (0. 060)n = 2. 8 x 10 -5 mol L-1 s-1 R 1 = k (0. 038)m(0. 060)n =1. 4 x 10 -5 mol L-1 s-1 If 2 m =2, then m =1. The reaction is first order in S 2 O 82 -. ERT 108/3 PHYSICAL CHEMISTRY 15 SEM 2 (2010/2011)

R 2 = k (0. 076)m(0. 060)n = k (0. 076)m(2 x 0. 030)n = k (0. 076)m (2)n(0. 030)n = 2. 8 x 10 -5 mol L-1 s-1 R 3 = k (0. 076)m(0. 030)n =1. 4 x 10 -5 mol L-1 s-1 If 2 n =2, then n =1. The reaction is first order in I-. The overall order of the reaction is m + n = 1+1 = 2 (second order). ERT 108/3 PHYSICAL CHEMISTRY 16 SEM 2 (2010/2011)

(ii) Use any one of the three experiments: k = 6. 1 x 10 -3 L mol-1 s-1. (iii) Once the k value is determined, the rate law can be used to predict the rate of reaction. Reaction rate, R = 6. 1 x 10 -3 L mol-1 s-1 x 0. 050 mol L-1 x 0. 025 mol L-1 = 7. 6 x 10 -6 mol L-1 s-1. TRT 401 Physical Chemistry BBLee@Uni. MAP ERT 108/3 PHYSICAL CHEMISTRY 17 SEM 2 (2010/2011)

• Based on the stoichiometry, 2 moles of SO 42 - are produced for every mole of S 2 O 82 - consumed. = 2. 8 x 10 -5 mol SO 42 -(L-1 s-1). ERT 108/3 PHYSICAL CHEMISTRY 18 SEM 2 (2010/2011)
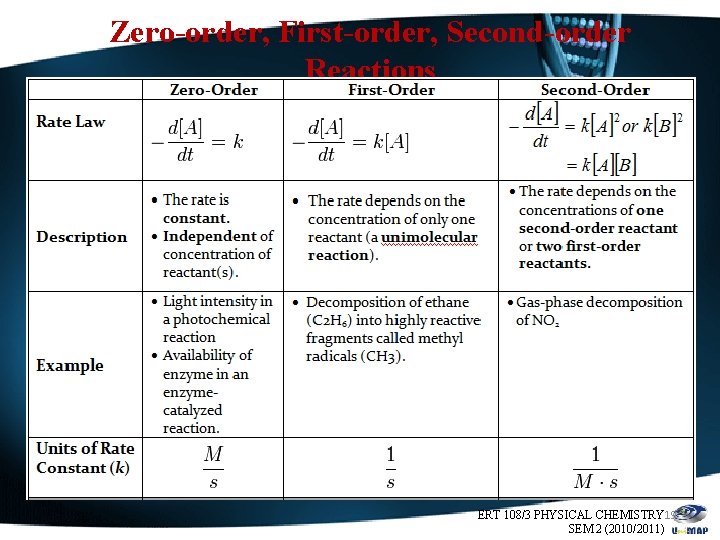
Zero-order, First-order, Second-order Reactions ERT 108/3 PHYSICAL CHEMISTRY 19 SEM 2 (2010/2011)

Cont……… ERT 108/3 PHYSICAL CHEMISTRY 20 SEM 2 (2010/2011)
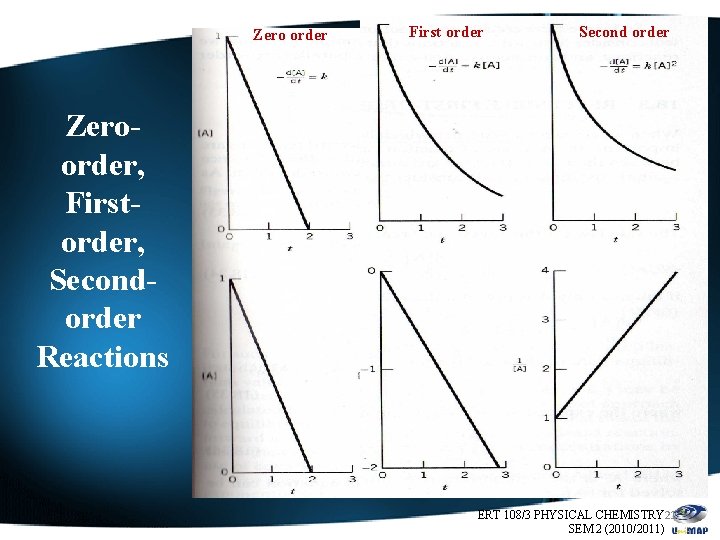
Zero order First order Second order Zeroorder, Firstorder, Secondorder Reactions ERT 108/3 PHYSICAL CHEMISTRY 21 SEM 2 (2010/2011)
![Example 4 (a) When [N 2 O 5] =0. 44 M, the rate of Example 4 (a) When [N 2 O 5] =0. 44 M, the rate of](http://slidetodoc.com/presentation_image_h2/703897c1a8f4e78c92ffb5a03b52022d/image-22.jpg)
Example 4 (a) When [N 2 O 5] =0. 44 M, the rate of decomposition of N 2 O 5 is 2. 6 x 10 -4 mol L-1 s-1. Ø what is the value of k for this first-order reaction? (b) N 2 O 5 initially at a concentration of 1. 0 mol/L in CCl 4, is allowed to decompose at 450 C. At what time will [N 2 O 5] be reduced to 0. 50 M? ERT 108/3 PHYSICAL CHEMISTRY 22 SEM 2 (2010/2011)

Answer (Example 4) (a) Rate of disappearance of N 2 O 5 (R) = k [N 2 O 5] = 5. 9 x 10 -4 s-1 (b) For 1 st order of reaction, to determine t, we can use: log [A]0 = log [N 2 O 5]0 = log 1. 0 = 0. log [A]t = log [N 2 O 5]t = log 0. 50 = -0. 30. use, k = 5. 9 x 10 -4 s-1. ERT 108/3 PHYSICAL CHEMISTRY 23 SEM 2 (2010/2011)
![Example 5 Time, min [A], M log [A] 1/[A] 0 1. 00 0. 00 Example 5 Time, min [A], M log [A] 1/[A] 0 1. 00 0. 00](http://slidetodoc.com/presentation_image_h2/703897c1a8f4e78c92ffb5a03b52022d/image-24.jpg)
Example 5 Time, min [A], M log [A] 1/[A] 0 1. 00 0. 00 1. 00 5 0. 63 -0. 20 1. 59 10 0. 46 -0. 34 2. 17 15 0. 36 -0. 44 2. 78 25 0. 25 -0. 60 4. 00 • The data of the above table were obtained for the decomposition reaction: A → 2 B + C. (a) Establish the order of the reaction. (b) What is the rate constant, k? ERT 108/3 PHYSICAL CHEMISTRY 24 SEM 2 (2010/2011)
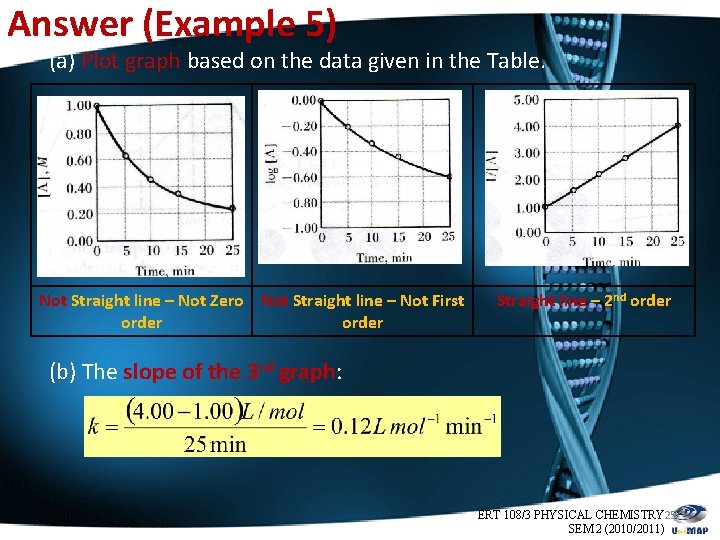
Answer (Example 5) (a) Plot graph based on the data given in the Table. Not Straight line – Not Zero Not Straight line – Not First order Straight line – 2 nd order (b) The slope of the 3 rd graph: ERT 108/3 PHYSICAL CHEMISTRY 25 SEM 2 (2010/2011)

Reaction rates: Effect of temperature • Chemical reactions tend to go faster at higher temperature. Ø slow down some reactions by lowering the temperature. • Increasing the temperature increases the fraction of the molecules that have energies in excess of the activation energy. Ø this factor is so important that for many chemical reactions it can lead to a doubling or tripling of the reaction rate for a ERT 108/3 PHYSICAL CHEMISTRY 26 0 SEM 2 (2010/2011) temperature increase of only 10 C.

Cont…. • In 1889, Arrhenius noted that the k data for many reactions fit the equation: where, A & Ea are constants characteristics of the reaction R = the gas constant. Ea = the Arrhenius activation energy (k. J/mol or kcal/mol) A = the pre-exponential factor (Arrhenius factor). ( the unit of A is the same as those of k. ) • Taking log of the above equation: ERT 108/3 PHYSICAL CHEMISTRY 27 SEM 2 (2010/2011)

Cont…. • If the Arrhenius equation is obeyed: Ø a plot of log 10 k versus 1/T is a straight line with slope: -Ea/2. 303 R and intercept log 10 A. Ø This enables Ea and A to be found. • Another useful equation: Ø Ø (eliminate the constant A). T 2 and T 1 - two kelvin temperatures. k 2 and k 1 - the rate constants at these temperatures. Ea – the activation energy (J/mol) R – the gas constant (8. 314 Jmol-1 K-1). ERT 108/3 PHYSICAL CHEMISTRY 28 SEM 2 (2010/2011)
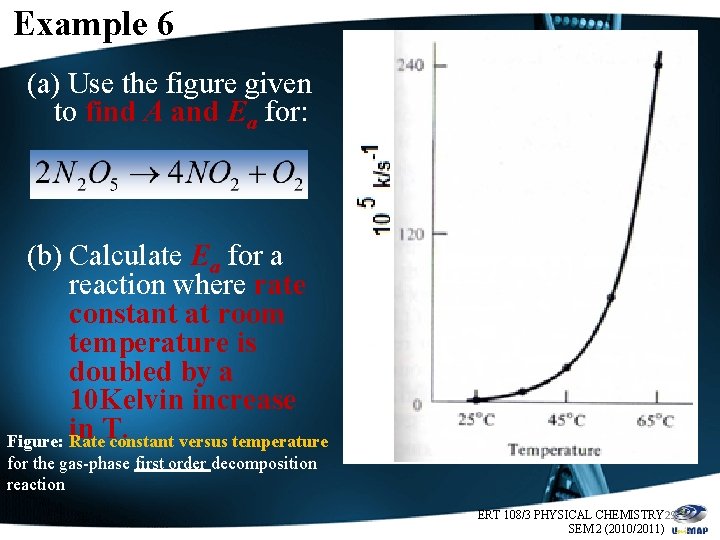
Example 6 (a) Use the figure given to find A and Ea for: (b) Calculate Ea for a reaction where rate constant at room temperature is doubled by a 10 Kelvin increase in T. Figure: Rate constant versus temperature for the gas-phase first order decomposition reaction ERT 108/3 PHYSICAL CHEMISTRY 29 SEM 2 (2010/2011)

Answer (Example 6 a) • Tabulate the data as follows. Temp, 0 C Temp, K 1/Temp, 1/K k, s-1 log 10 k 25 298 0. 0034 0. 001 -3 • Construct the Arrhenius plot of log 10 k versus 1/T for the reaction. Ø Intercept (log 10 A)=13. 5 A = 3 x 1013 s-1 Ø Slope=-5500 K, Ea=25 kcal/mol =105 k. J/mol Figure: Arrhenius plot of log 10 k versus 1/T for this reaction. Note: the long extrapolation needed to find A. ERT 108/3 PHYSICAL CHEMISTRY 30 SEM 2 (2010/2011)

Answer (Example 6 b) • Based on the given info: Ø k 2 = 2 k 1 , ØT 1 = room temperature (298 K), ØT 2=298+10 = 308 K, • The Arrhenius equation: • Substitute: Ea = 53 k. J/mol ERT 108/3 PHYSICAL CHEMISTRY 31 SEM 2 (2010/2011)

Reaction Mechanisms • Each molecular event that significantly alters a molecule’s energy or geometry is called an elementary process (reaction). • The mechanism of a reaction: Ø the sequence of elementary reactions that add up to give the overall reaction. Ø A mechanism is a hypothesis about the elementary steps through which chemical change occurs. ERT 108/3 PHYSICAL CHEMISTRY 32 SEM 2 (2010/2011)

Reaction Mechanisms • Elementary processes in which a single molecule dissociates (unimolecular) or two molecules collide (bimolecular) much more probable than a process requiring the simultaneous collision of three bodies (termolecular). • All elementary processes are reversible and may reach a steady-state condition. In the steady state the rates of the forward & reverse processes become equal. • One elementary process may occur much more slower than all the others. In this case, it determines the rate at which the overall reaction proceeds & is called the rate-determining/ limiting step. ERT 108/3 PHYSICAL CHEMISTRY 33 SEM 2 (2010/2011)

The Hydrogen-Iodine Reaction H 2 (g) + I 2 (g) → 2 HI (g) • Rate of formation of HI = k [H 2][I 2] • The hydrogen-iodine reaction is proposed to be a two-step mechanism [Sullivan J. (1967). J. Chem. Phys. 46: 73]. Ø 1 st step: iodine molecules are believed to dissociate into iodine atoms. Ø 2 nd step: simultaneous collision of two iodine atoms and a hydrogen molecule. (this termolecular step is expected to occur much more slowly – the rate-determining step). ERT 108/3 PHYSICAL CHEMISTRY 34 SEM 2 (2010/2011)
![The Hydrogen-Iodine Reaction 1 st step: [Fast] 2 nd step: [Slow] Net: • If The Hydrogen-Iodine Reaction 1 st step: [Fast] 2 nd step: [Slow] Net: • If](http://slidetodoc.com/presentation_image_h2/703897c1a8f4e78c92ffb5a03b52022d/image-35.jpg)
The Hydrogen-Iodine Reaction 1 st step: [Fast] 2 nd step: [Slow] Net: • If the reversible step reaches a steady state condition: Ø rate of disappearance of I 2 = rate of formation of I 2 ERT 108/3 PHYSICAL CHEMISTRY 35 SEM 2 (2010/2011)

The Hydrogen-Iodine Reaction • For the rate-determining step: Rate of formation of HI = k 3 [I]2[H 2] = K[H 2][I 2] where (K=k 1 k 3/k 2) ERT 108/3 PHYSICAL CHEMISTRY 36 SEM 2 (2010/2011)

Example 7 • The thermal decomposition of ozone to oxygen: 2 O 3 (g) → 3 O 2 (g) • The observed rate law: Rate of disappearance of O 3 = • Show that the following mechanism is consistent with this experiment rate law. 1 st: 2 nd: ERT 108/3 PHYSICAL CHEMISTRY 37 SEM 2 (2010/2011)

Answer (Example 7) • Assume the 1 st step reaches the steady state condition: Rate of formation of O = Rate of disappearance of O k 1 [O 3] = k 2 [O 2] [O] • Assume the 2 nd step is the rate-determining step: Rate of disappearance of O 3 = k 3 [O][O 3] (where k = k 1 k 3/k 2) ERT 108/3 PHYSICAL CHEMISTRY 38 SEM 2 (2010/2011)

Experimental methods for fast reactions • Many reactions are too fast to follow by the classical methods. • Several ways to study fast reactions : 1. Rapid flow methods: (i) Continuous flow (ii) Stopped flow 2. Relaxation methods: (i) Temperature jump (T-jump) method (ii) Pressure jump method (iii) Electric field jump method 3. Flash photolysis 4. Shock tube 5. Nuclear-magnetic-resonance (NMR) spectroscopy ERT 108/3 PHYSICAL CHEMISTRY 39 SEM 2 (2010/2011)

ASSIGNMENT 1 Write a short note for the following fast reaction: a) b) c) d) e) Rapid flow methods Relaxation methods Flash photolysis Shock tube Nuclear-magnetic-resonance (NMR) spectroscopy ERT 108/3 PHYSICAL CHEMISTRY 40 SEM 2 (2010/2011)

The End ERT 108/3 PHYSICAL CHEMISTRY 41 SEM 2 (2010/2011)
- Slides: 41