ERT 1083 PHYSICAL CHEMISTRY FIRST LAW OF THERMODYNAMICS




























- Slides: 33

ERT 108/3 PHYSICAL CHEMISTRY FIRST LAW OF THERMODYNAMICS Prepared by: Pn. Hairul Nazirah Abdul Halim

Thermochemistry • Thermochemistry is the study of heat produced or required by chemical reactions. • If we know the ΔU and ΔH for a reaction, we can predict the heat of reaction. • Enthalpy of a system decreases when heat is releases. ΔH < 0. (Exothermic process). • Enthalpy of a system increases when heat is absorbed. ΔH > 0. (Endothermic process).

Standard Enthalpy Changes • Standard enthalpy change, is the change in enthalpy for a process in which the initial and final substances are in their standard states. • Standard states of a substance at a specified temperature is its pure form at 1 bar. • For example; - the standard state of liquid ethanol at 298 K is pure liquid ethanol at 298 K and 1 bar.

• Example of standard enthalpy change, ; the standard enthalpy of vaporization, is the enthalpy change per mole when a pure liquid at 1 bar vaporizes to a gas at 1 bar.

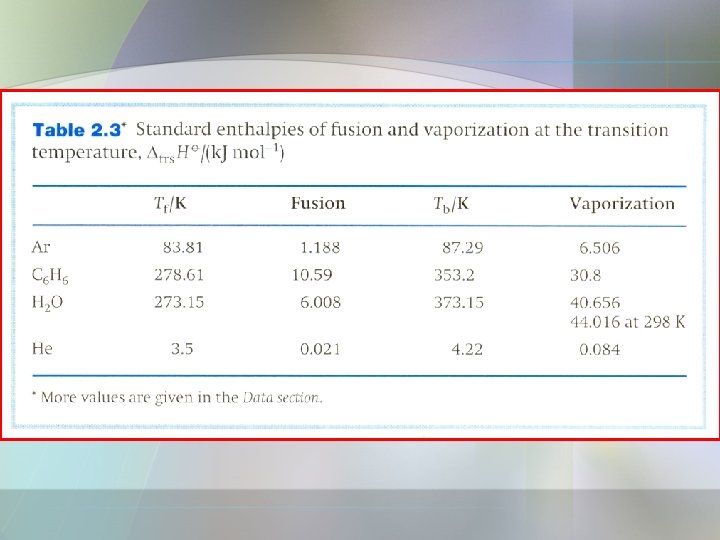
a) Enthalpy of Physical Change • The standard enthalpy transition, is the standard enthalpy change that accompanies a change of physical state. • Example, - standard enthalpy of vaporization, - standard enthalpy of fusion,


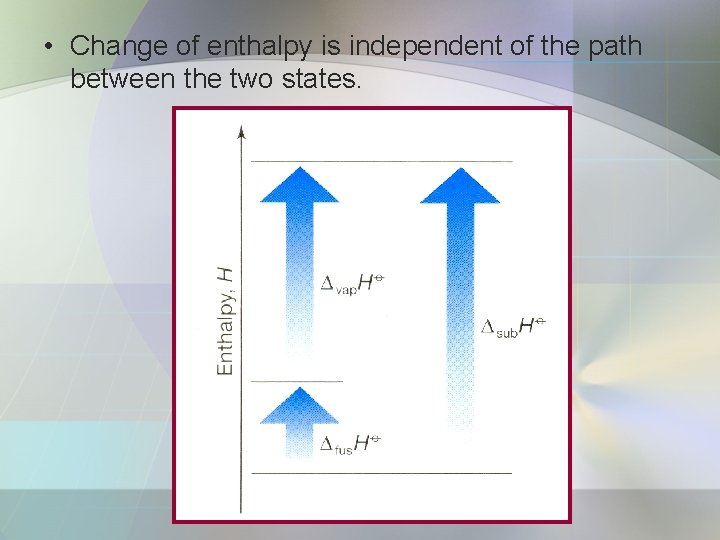
• Change of enthalpy is independent of the path between the two states.

• For example, the direct conversion from solid to vapor; • Or as two steps, first fusion (melting) and then vaporization; • Conclusion;

• The standard enthalpy changes of a forward process and its reverse must differ only in sign. • For example, at 298 K; - enthalpy of vaporization of water is +44 k. J/mol - enthalpy of condensation is -44 k. J/mol


b) Enthalpy of Chemical Change Thermochemical equation is a combination of a chemical equation and the corresponding change in standard enthalpy: = the standard enthalpy change

• Alternatively, we can write the chemical equation with standard reaction enthalpy, • For combustion of methane, we write;

• The standard reaction enthalpy is; • Where, v = stoichiometric coefficients = the standard molar enthalpy of species J at the temp. of interest.

• For the reaction; • The standard reaction enthalpy is; • Where is the standard molar enthalpy of species J at the temp. of interest.

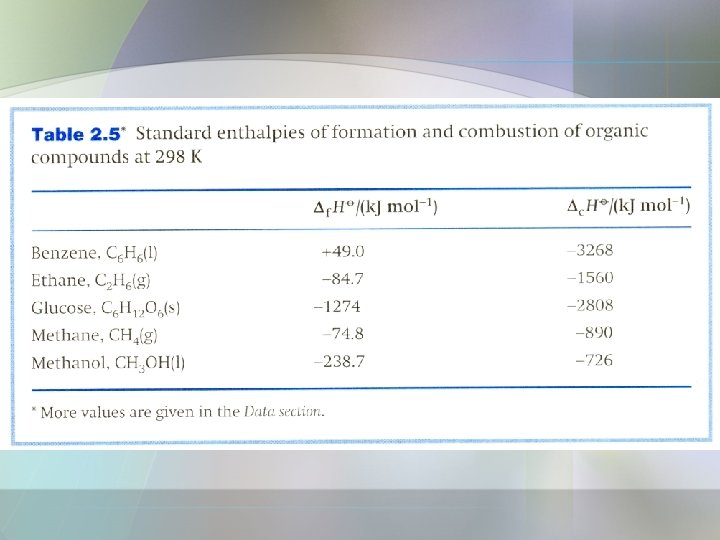
• Standard enthalpy of combustion, is the standard reaction enthalpy for the complete oxidation of an organic compound to CO 2 gas and liquid H 2 O. • Organic compound contains C, H, O and N 2 (if N is present). • Example, the combustion of glucose;


c) Hess’s Law • Standard enthalpies of individual reactions can be combined to obtain the enthalpy of another reaction. • This application of the First Law is called Hess’s Law: The standard enthalpy of an overall reaction is the sum of the standard enthalpies of the individual reactions into which a reaction may be divided.

Example Using Hess’s Law The standard reaction enthalpy for the hydrogenation of propene is -124 k. J mol-1, The standard reaction enthalpy for the combustion of Propane is -2220 k. J mol-1; Calculate the standard enthalpy of combustion of propene.

Solution • Add or subtract the reactions given, together with any others needed, so as to reproduce the reaction required. • Then add or subtract the reaction enthalpies in the same way. Additional data are in Table 2. 5. • The combustion reaction we require is;

• This reaction can be recreated from the following sum:

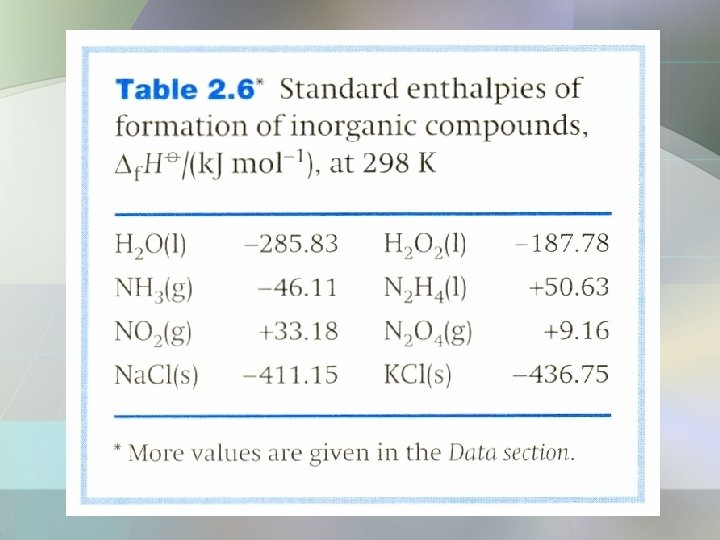
Standard Enthalpy of Formation • The standard enthalpy of formation, of a substance is the standard reaction enthalpy for the formation of the compound from its elements in their reference states. • The reference state of an element is its most stable state at the specified temp. and 1 bar. • For example, at 298 K the reference state of nitrogen is a gas of N 2 molecules, mercury is liquid mercury, carbon is graphite.


a) The reaction enthalpy in terms of enthalpies of formation

Example The standard reaction enthalpy of: is calculated as follows: -

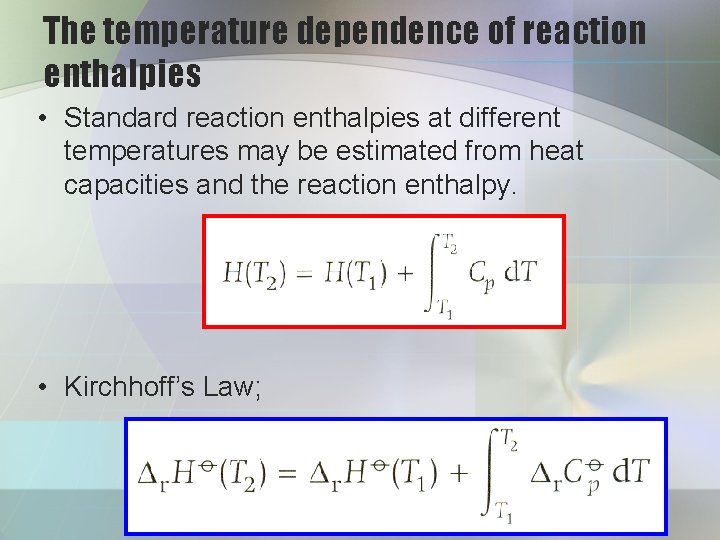
The temperature dependence of reaction enthalpies • Standard reaction enthalpies at different temperatures may be estimated from heat capacities and the reaction enthalpy. • Kirchhoff’s Law;

is the difference of the molar heat capacities of products and reactants under standard conditions weighted by the stoichiometric numbers;

Example Using Kirchhoff’s Law The standard enthalpy of formation of gaseous H 20 at 298 K is -241. 82 k. J mol-1. Estimate its value at 100°C given the following values of the molar heat capacities at constant pressure: H 20(g): 33. 58 J K-1 mol-1 ; H 2(g): 28. 84 J K -1 mol-1; 02 (g): 29. 37 J K-1 mol-1. Assume that the heat capacities are independent of temperature.

Solution • When is independent of temperature in the range T 1 to T 2, . Therefore,

• To proceed, write the chemical equation, identify the stoichiometric coefficients, and calculate from the data. • The reaction is; • So;

Example Calculate for the following reaction at 1450 K and 1 bar; ½ H 2 (g) + ½ Cl 2(g) HCl(g) Given that (HCl, g) = -92. 3 k. J/mol at 298. 15 K and that; over this temperature range.

Solution Use the following equation; T 1 = 298. 15 K T 2 = 1450 K

