Chromatography and Instrumentation in Organic Chemistry 1 Chromatography














- Slides: 28

Chromatography and Instrumentation in Organic Chemistry

1. Chromatography • The word chromatography comes from two Greek words which mean “colour writing” • It was invented by a Russian botanist Mikhail Twsett in 1903 (separating pigments in plants) • Chromatography now widely used to separate substances in a mixture • Also used to measure the amounts of substances in a mixture

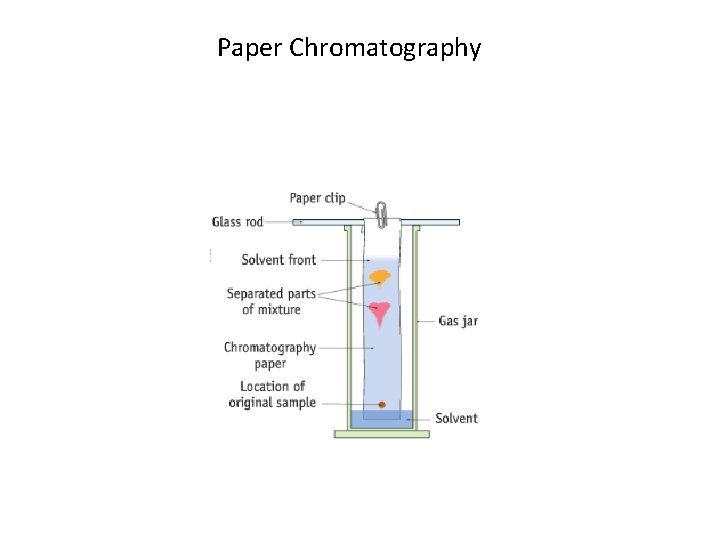
1. Chromatography • In chromatography, a mobile phase carries substances in a mixture at different rates through a stationary phase • Examples of mobile phase: water, ethanol • Examples of stationary phase: paper, aluminium oxide Chromatography = a separation technique in which a mobile phase carrying a mixture moves in contact with a selectively adsorbent stationary phase

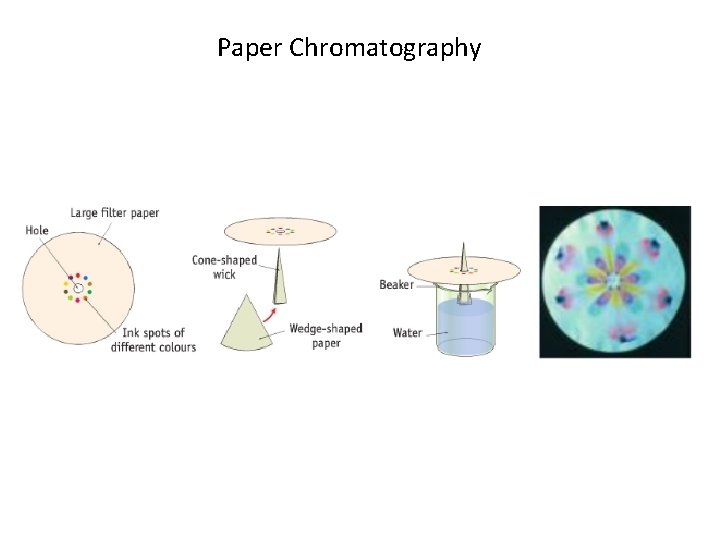
Paper Chromatography

Paper Chromatography

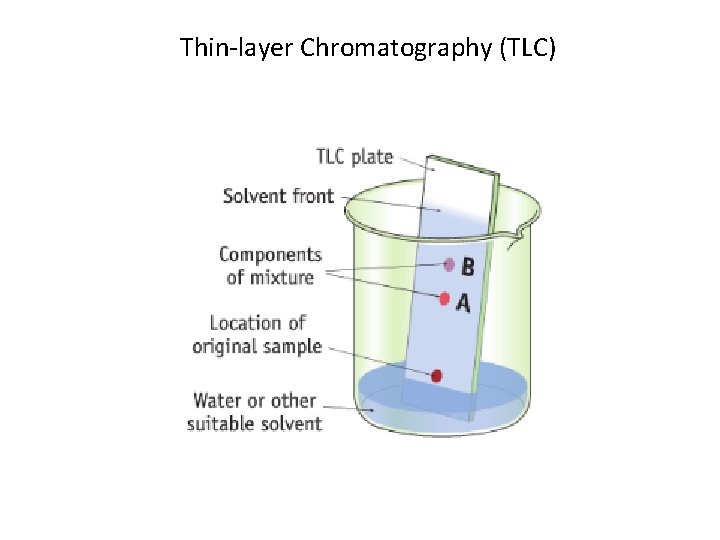
Thin-layer Chromatography (TLC)

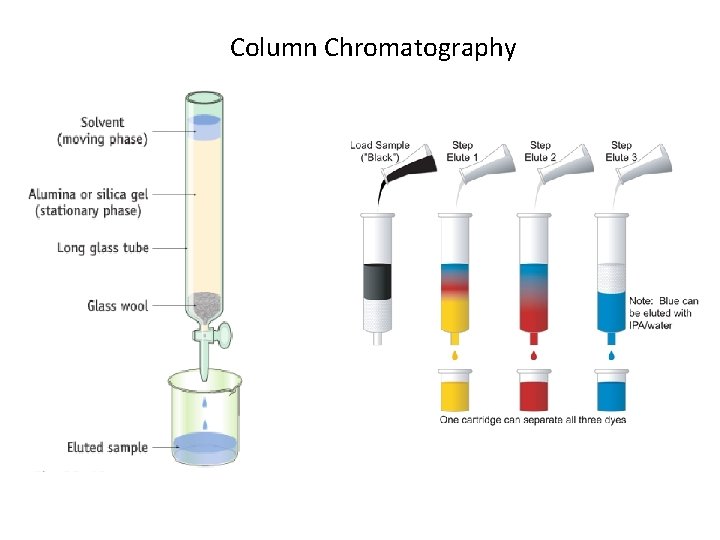
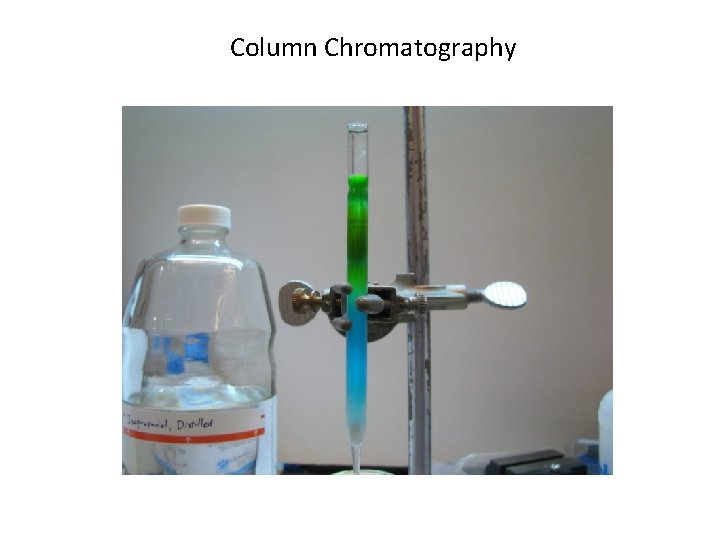
Column Chromatography

Column Chromatography

Column Chromatography

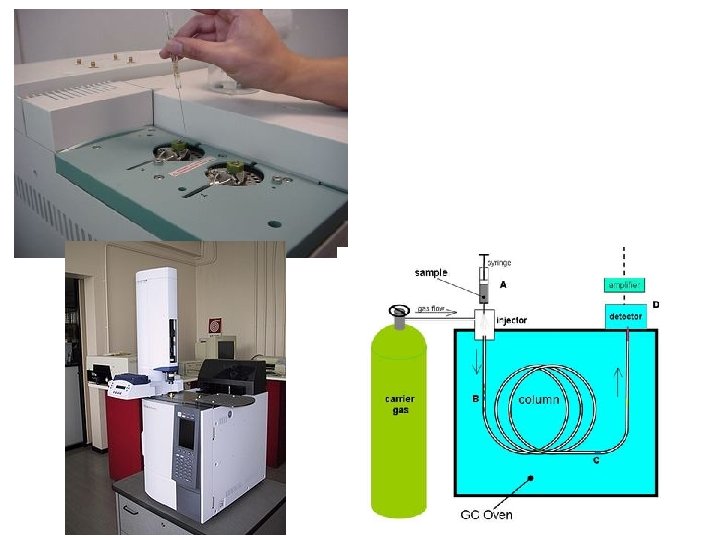
Chromatography in industrial laboratories 1. Gas chromatography (GC) • Stationary phase = high boiling point liquid (eg. Longchain alkane) spread on solid particles (eg. silica gel) • Mobile phase = a gas • Both are packed into a long, coiled tube called a column • The column is several metres in length • Column is inside an oven


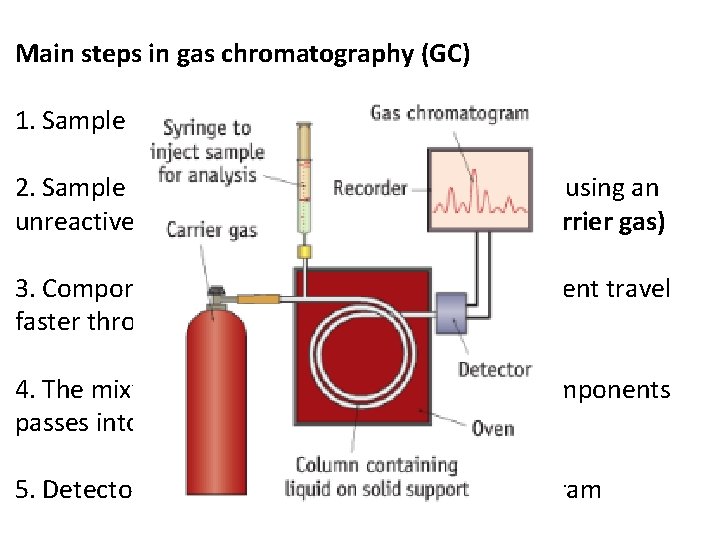
Main steps in gas chromatography (GC) 1. Sample is injected using syringe 2. Sample is vaporised and carried through tube using an unreactive gas eg. Nitrogen, helium or argon (carrier gas) 3. Components that are more soluble in the solvent travel faster through the tube 4. The mixture is separated and the separate components passes into a detector 5. Detector plots a chart called a gas chromatogram

• GC is used to measure alcohol in blood and urine samples • Also used to analyse drug-test samples from athletes

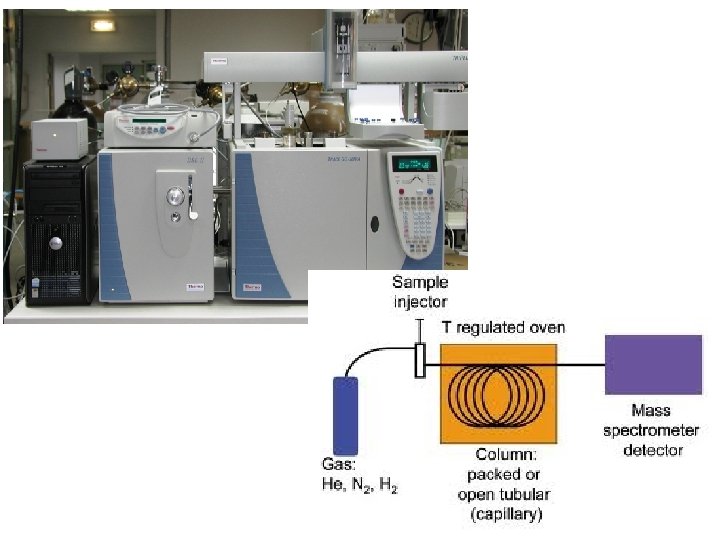
• Gas chromatography is often combined with mass spectrometry for analysis (GC-MS) • Separated components from gas chromatography passed through mass spectrometer • Each compound can be identified separately • GC-MS used in forensic science to identify drugs • GC-MS used in drug research to study metabolism (drug given to patient and blood samples analysed) • GC-MS used to detect gases from waste dumps and pollutants in water

• GC-MS even used on Mars!


Chromatography in industrial laboratories 2. High performance liquid chromatography (HPLC) • Mobile phase = liquid • Stationary phase = small solid particles • Better separation occurs when particles in stationary phase are very small • Pump needed to force liquid through column

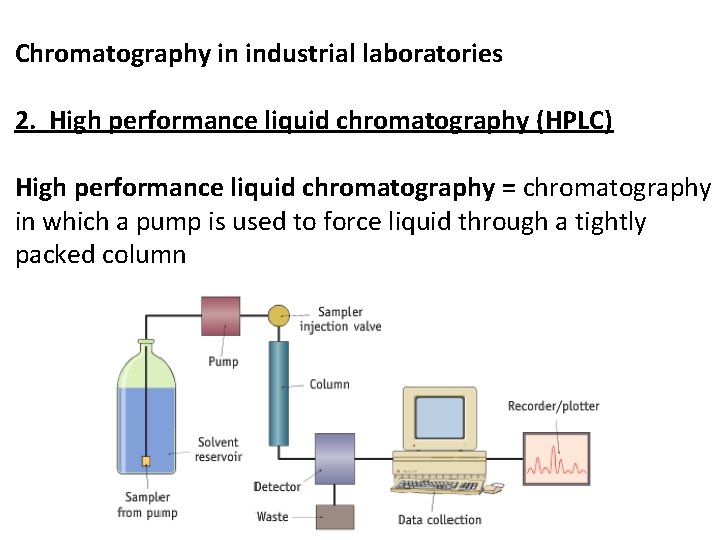
Chromatography in industrial laboratories 2. High performance liquid chromatography (HPLC) High performance liquid chromatography = chromatography in which a pump is used to force liquid through a tightly packed column

Chromatography in industrial laboratories 2. High performance liquid chromatography (HPLC) • HPLC column is shorter than GC column because separation is more efficient • HPLC column only 10 – 30 cm long • HPLC column made of stainless steel to withstand pressure • HPLC uses lower temperature than GC so can analyse substances that would be damaged by higher temperatures • HPLC used to analyse foods

Infra-red Spectrometry (IR) • Infra-red radiation has a longer wavelength than visible light

Infra-red Spectrometry (IR) • Infra-red radiation is invisible but given off by hot objects

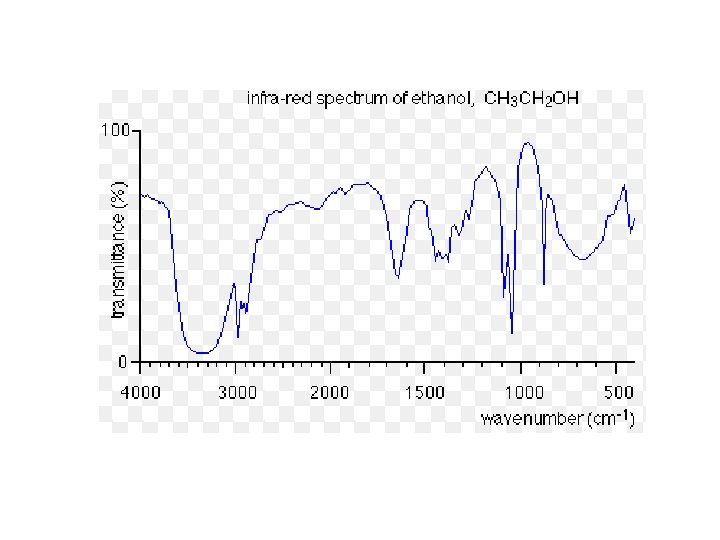
Infra-red Spectrometry (IR) • Organic compounds absorb infra-red radiation • Infra-red radiation absorbed by the vibrations of the bonds of molecules Eg. A C=O bond will absorb a different frequency of infra-red radiation to an O-H bond • Every organic compound has its own IR spectrum



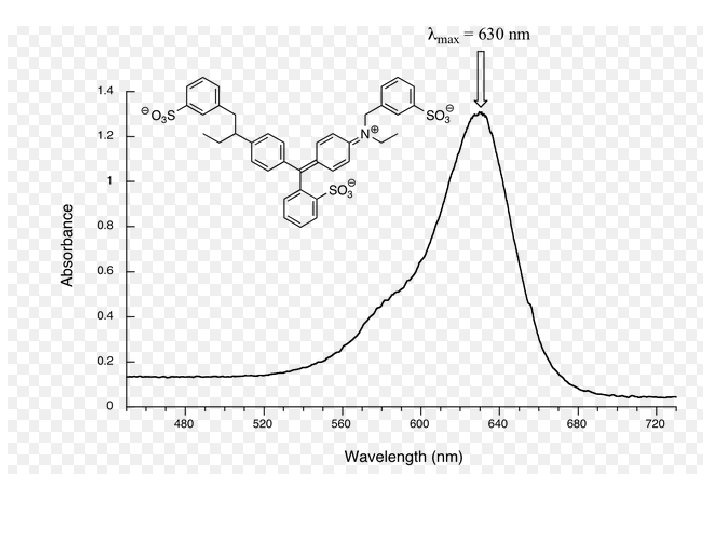
Ultraviolet Spectrometry (UV) • Ultraviolet radiation has a wavelength shorter than visible light

Ultraviolet Spectrometry (UV) • UV spectrometry analyses how different compounds absorb UV light • UV spectrometry used to detect the presence of particular functional groups in molecules • Also used to measure the concentration of substances in solution eg. Drugs

