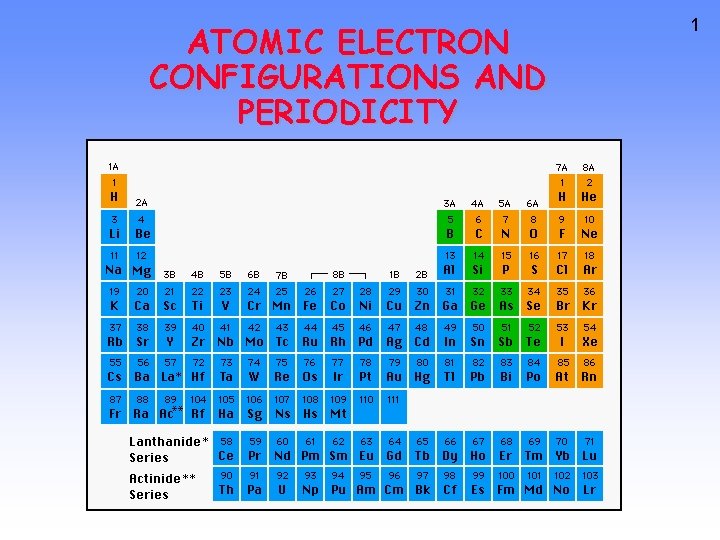
ATOMIC ELECTRON CONFIGURATIONS AND PERIODICITY 1 Arrangement of










![Transition Metals Table 8. 4 All 4 th period elements have the configuration [argon] Transition Metals Table 8. 4 All 4 th period elements have the configuration [argon]](https://slidetodoc.com/presentation_image_h2/4deada5f3d3515d269ad47a1e29bcbec/image-18.jpg)
![Lanthanides and Actinides 20 All these elements have the configuration [core] nsx (n - Lanthanides and Actinides 20 All these elements have the configuration [core] nsx (n -](https://slidetodoc.com/presentation_image_h2/4deada5f3d3515d269ad47a1e29bcbec/image-20.jpg)




- Slides: 29

ATOMIC ELECTRON CONFIGURATIONS AND PERIODICITY 1

Arrangement of Electrons in Atoms Each orbital can be assigned no more than 2 electrons! This is tied to the existence of a 4 th quantum number, the electron spin quantum number, ms. 2

3 Electron Spin Quantum Number, ms Can be proved experimentally that electron has a spin. Two spin directions are given by ms where ms = +1/2 and -1/2.

Electron Spin Quantum Number Diamagnetic: NOT attracted to a magnetic field Paramagnetic: substance is attracted to a magnetic field. Substance has unpaired electrons. 4

5 Pauli Exclusion Principle No two electrons in the same atom can have the same set of 4 quantum numbers. That is, each electron has a unique address.

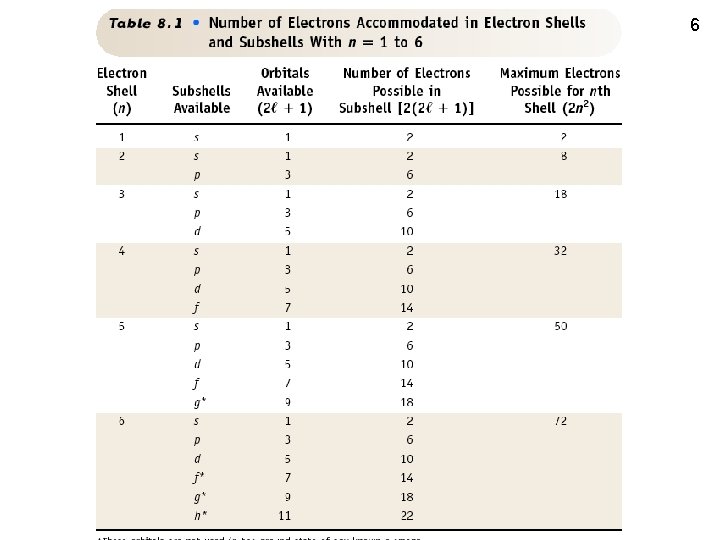
6

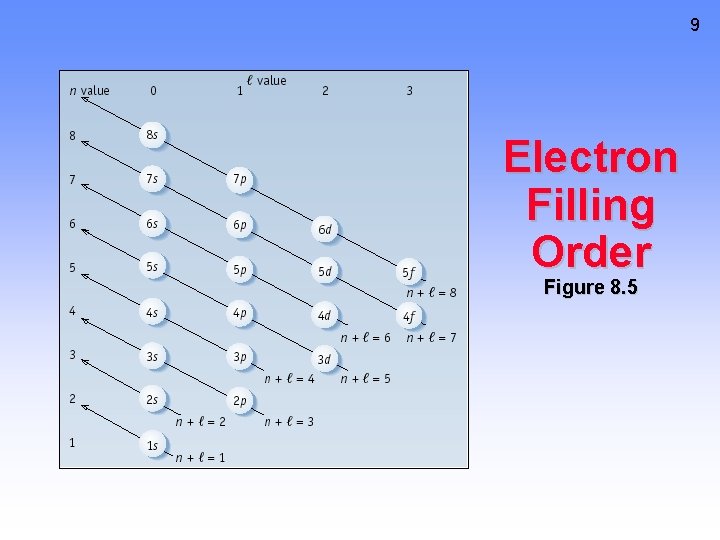
7 Assigning Electrons to Atoms • Electrons generally assigned to orbitals of successively higher energy. • For H atoms, E = - C(1/n 2). E depends only on n. • For many-electron atoms, energy depends on both n and l. • See Figure 8. 5, page 295 and Screen 8. 7.

Assigning Electrons to Subshells • In H atom all subshells of same n have same energy. • In many-electron atom: a) subshells increase in energy as value of n + l increases. b) for subshells of same n + l, subshell with lower n is lower in energy. 8

9 Electron Filling Order Figure 8. 5

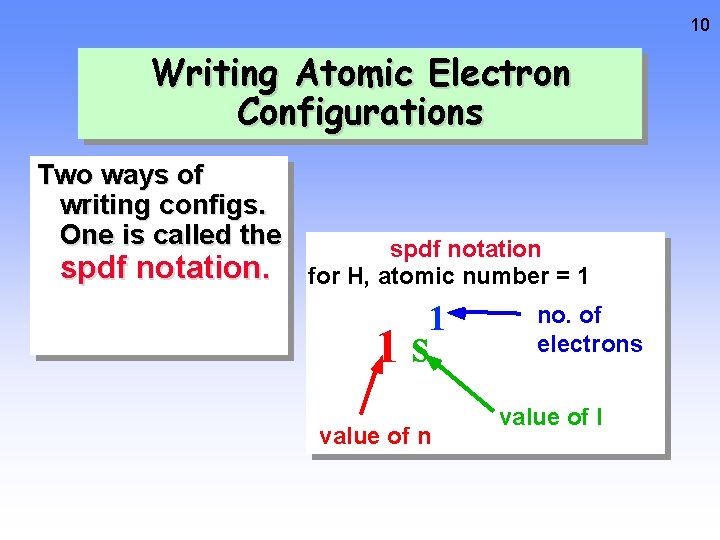
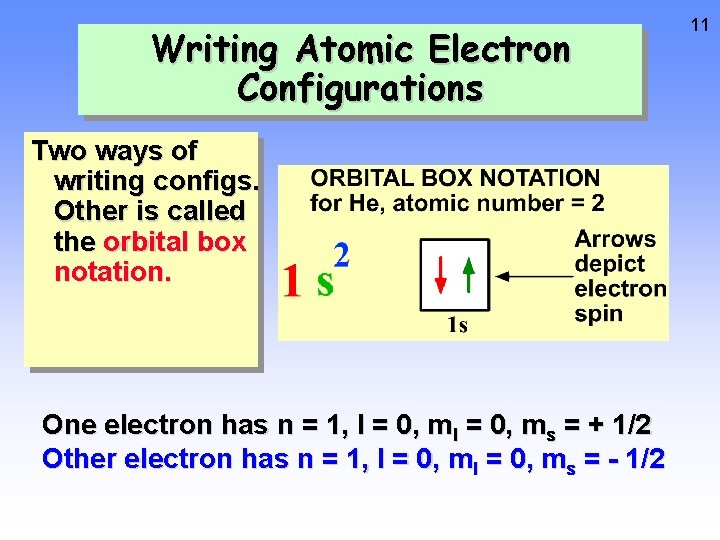
10 Writing Atomic Electron Configurations Two ways of writing configs. One is called the spdf notation for H, atomic number = 1 1 1 s value of n no. of electrons value of l

Writing Atomic Electron Configurations Two ways of writing configs. Other is called the orbital box notation. One electron has n = 1, l = 0, ms = + 1/2 Other electron has n = 1, l = 0, ms = - 1/2 11

12 See “Toolbox” for Electron Configuration tool.

13 Electron Configurations and the Periodic Table Figure 8. 7

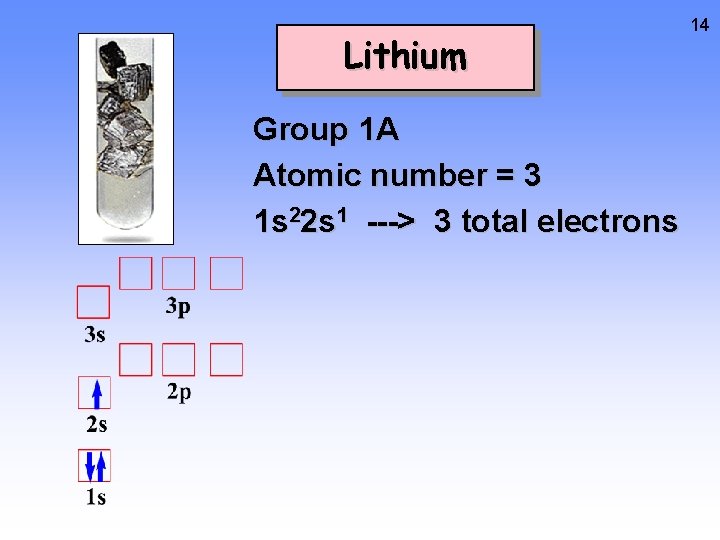
Lithium Group 1 A Atomic number = 3 1 s 22 s 1 ---> 3 total electrons 14

15 Electron Configurations of p -Block Elements

Sodium Group 1 A Atomic number = 11 1 s 2 2 p 6 3 s 1 or “neon core” + 3 s 1 [Ne] 3 s 1 (uses rare gas notation) Note that we have begun a new period. All Group 1 A elements have [core]ns 1 configurations. 16

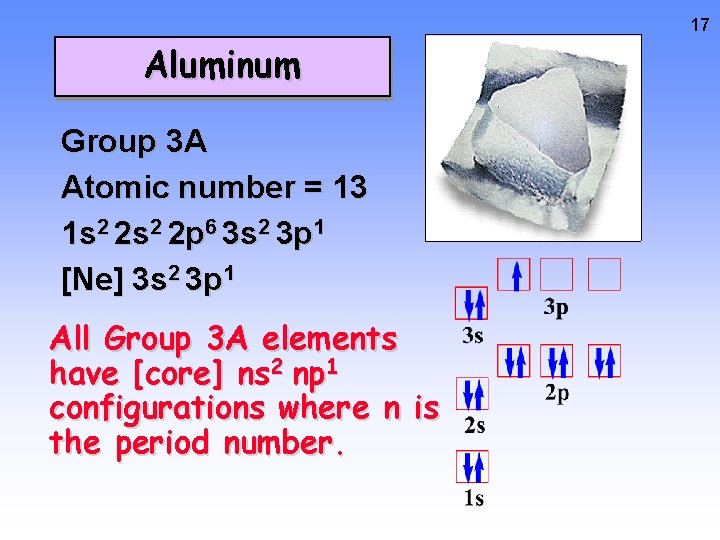
17 Aluminum Group 3 A Atomic number = 13 1 s 2 2 p 6 3 s 2 3 p 1 [Ne] 3 s 2 3 p 1 All Group 3 A elements have [core] ns 2 np 1 configurations where n is the period number.
![Transition Metals Table 8 4 All 4 th period elements have the configuration argon Transition Metals Table 8. 4 All 4 th period elements have the configuration [argon]](https://slidetodoc.com/presentation_image_h2/4deada5f3d3515d269ad47a1e29bcbec/image-18.jpg)
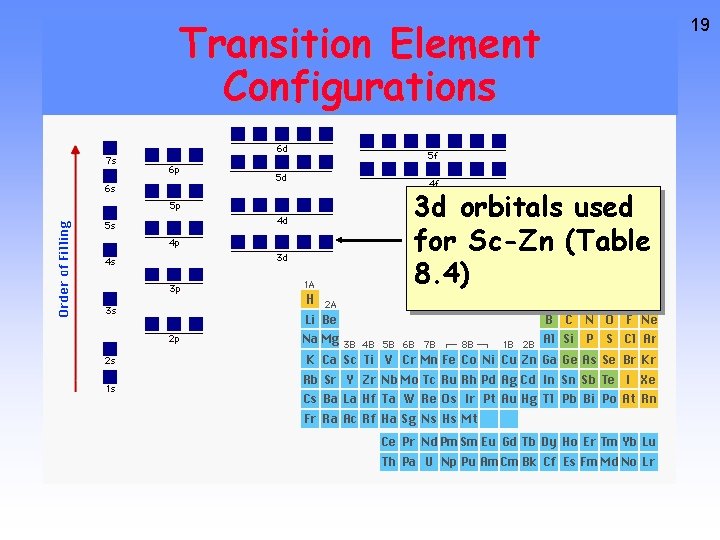
Transition Metals Table 8. 4 All 4 th period elements have the configuration [argon] nsx (n - 1)dy and so are “d-block” elements. Chromium Iron Copper 18

Transition Element Configurations 3 d orbitals used for Sc-Zn (Table 8. 4) 19
![Lanthanides and Actinides 20 All these elements have the configuration core nsx n Lanthanides and Actinides 20 All these elements have the configuration [core] nsx (n -](https://slidetodoc.com/presentation_image_h2/4deada5f3d3515d269ad47a1e29bcbec/image-20.jpg)
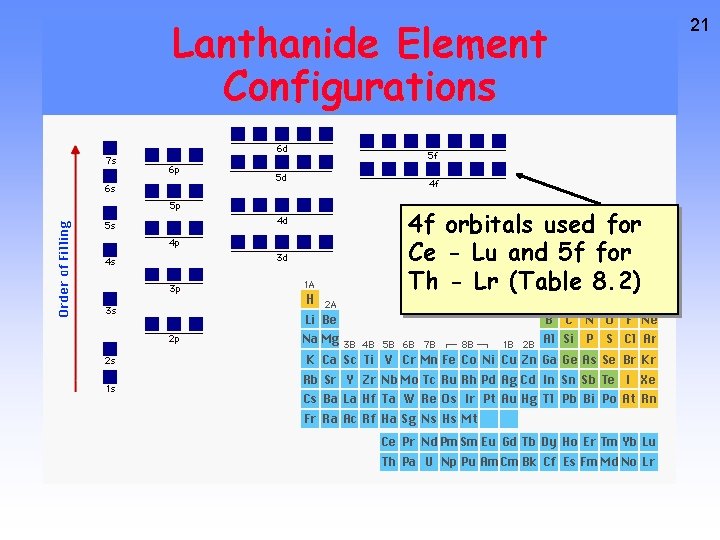
Lanthanides and Actinides 20 All these elements have the configuration [core] nsx (n - 1)dy (n - 2)fz or nsx (n - 2)fz and so are “f-block” elements. Cerium [Xe] 6 s 2 5 d 1 4 f 1 Uranium [Rn] 7 s 2 6 d 1 5 f 3 Only for Ce, Lu, and Gd!!! The rest are like Uranium

Lanthanide Element Configurations 4 f orbitals used for Ce - Lu and 5 f for Th - Lr (Table 8. 2) 21

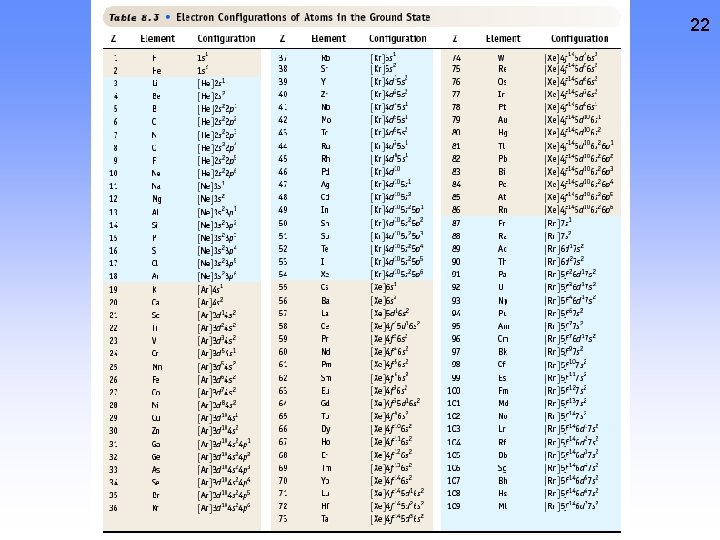
22

23 Transition Metals • Cr, Mo (d 4) – Lose 1 electron from “s” and bump to d 5 • Cu, Ag, Au (d 9) – Lose 1 electron from “s” and bump to d 10

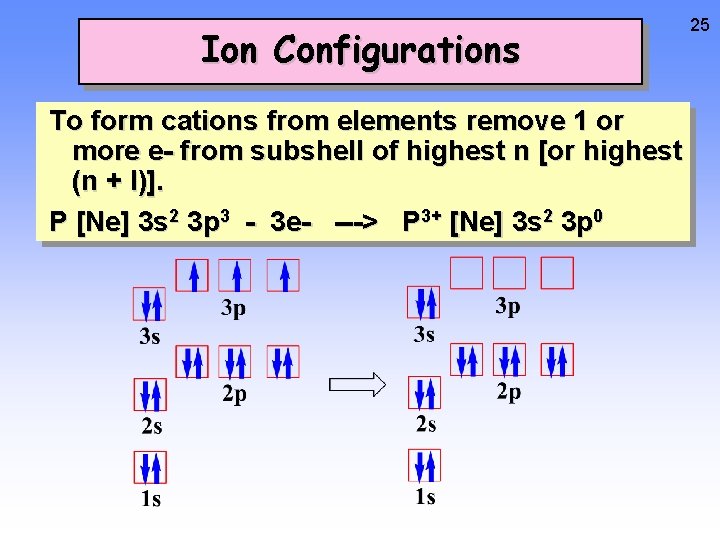
Ion Configurations To form cations from elements remove 1 or more e- from subshell of highest n [or highest (n + l)]. P [Ne] 3 s 2 3 p 3 - 3 e- ---> P 3+ [Ne] 3 s 2 3 p 0 24

Ion Configurations To form cations from elements remove 1 or more e- from subshell of highest n [or highest (n + l)]. P [Ne] 3 s 2 3 p 3 - 3 e- ---> P 3+ [Ne] 3 s 2 3 p 0 25

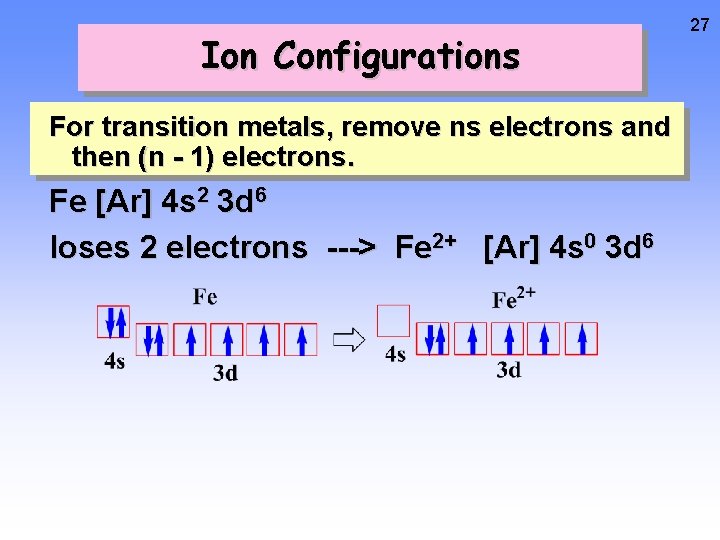
Ion Configurations For transition metals, remove ns electrons and then (n - 1) electrons. Fe [Ar] 4 s 2 3 d 6 loses 2 electrons ---> Fe 2+ [Ar] 4 s 0 3 d 6 26

Ion Configurations For transition metals, remove ns electrons and then (n - 1) electrons. Fe [Ar] 4 s 2 3 d 6 loses 2 electrons ---> Fe 2+ [Ar] 4 s 0 3 d 6 27

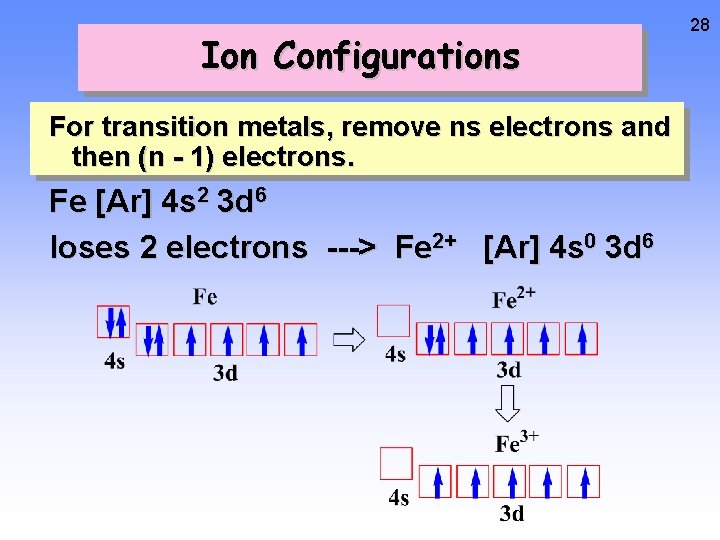
Ion Configurations For transition metals, remove ns electrons and then (n - 1) electrons. Fe [Ar] 4 s 2 3 d 6 loses 2 electrons ---> Fe 2+ [Ar] 4 s 0 3 d 6 28

29 Ion Configurations • Try and determine what charges the following metals will form…. • Ti (2 charges) – +2, +4 • Fe (2 charges) – +2, + 3 • Sn (2 charges) – +2, + 4 • Cu (2 charges) – +1, + 2