CHAPTER 8 Atomic Electron Configurations and Chemical Periodicity

























































- Slides: 63

CHAPTER 8 Atomic Electron Configurations and Chemical Periodicity Outline -Collect homework -Review -Trends -Ions Kull Spring 07 Lesson 23 Ch 8 1

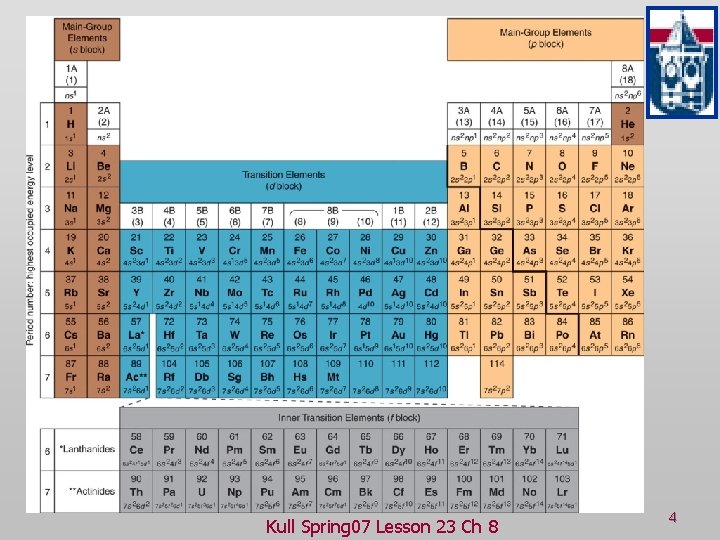
Review Spdf notation v Orbital box notation v Kull Spring 07 Lesson 23 Ch 8 2

Characteristics of Many-Electron Atoms: The Electron-Spin Quantum Number Name Symbol Permitted Values Property principal n positive integers(1, 2, 3, …) orbital energy (size) angular momentum l integers from 0 to n-1 orbital shape (The l values 0, 1, 2, and 3 correspond to s, p, d, and f orbitals, respectively. ) magnetic ml integers from -l to 0 to +l orbital orientation spin ms +1/2 or -1/2 direction of e- spin Kull Spring 07 Lesson 23 Ch 8 3

Kull Spring 07 Lesson 23 Ch 8 4

Practice Problem 23 -1 v Consider this set of quantum numbers: n = 3, ℓ = 2, mℓ = -1, ms = +½ The maximum number of electrons in an atom which can share the above set of quantum numbers is A) 1 B) 14 C) 3 D) 10 E) none of the above Kull Spring 07 Lesson 23 Ch 8 5

Practice Problem 23 -1 Answer v Consider this set of quantum numbers: n = 3, ℓ = 2, mℓ = -1, ms = +½ The maximum number of electrons in an atom which can share the above set of quantum numbers is A) 1 B) 14 C) 3 D) 10 E) none of the above Kull Spring 07 Lesson 23 Ch 8 6

Practice Problem 23 -2 v An atom in its ground state contains 30 electrons. How many of these are in sublevels with ℓ = 2? A) 2 B) 4 C) 6 D) 8 E) 10 Kull Spring 07 Lesson 23 Ch 8 7

Practice Problem 23 -2 Answer v An atom in its ground state contains 30 electrons. How many of these are in sublevels with ℓ = 2? A) 2 B) 4 C) 6 D) 8 E) 10 Kull Spring 07 Lesson 23 Ch 8 8

Practice Problem 23 -3 What are the possible values for the angular momentum quantum number ℓ ? A) integers from -ℓ to 0 to +ℓ B) 1, 2, 3, etc. C) 2, 4, 6, etc. D) +½ , -½ E) integers from 0 to n - 1 Kull Spring 07 Lesson 23 Ch 8 9

Practice Problem 23 -3 Answer v What are the possible values for the angular momentum quantum number ( ℓ) ? A) integers from -ℓ to 0 to +ℓ B) 1, 2, 3, etc. C) 2, 4, 6, etc. D) +½ , -½ E) integers from 0 to n - 1 Kull Spring 07 Lesson 23 Ch 8 10

Practice Problem 23 -4 v The electron configuration of the outermost electrons of atoms of the halogen group is: A) ns 2 np 7 B) ns 1 C) ns 2 np 5 D) ns 2 np 6(n-1)d 7 E) ns 2 np 6 Kull Spring 07 Lesson 23 Ch 8 11

Practice Problem 23 -4 Answer v The electron configuration of the outermost electrons of atoms of the halogen group is: A) ns 2 np 7 B) ns 1 C) ns 2 np 5 D) ns 2 np 6(n-1)d 7 E) ns 2 np 6 Kull Spring 07 Lesson 23 Ch 8 12

Practice Problem 23 -5 v The electronic configuration of the element whose atomic number is 26 is: A) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 0 3 d 8 B) 1 s 2 2 p 6 3 s 2 3 p 6 3 d 6 4 s 2 C) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 6 D) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 4 4 p 2 E) none of the above Kull Spring 07 Lesson 23 Ch 8 13

Practice Problem 23 -5 Answer v The electronic configuration of the element whose atomic number is 26 is: A) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 0 3 d 8 B) 1 s 2 2 p 6 3 s 2 3 p 6 3 d 6 4 s 2 C) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 6 D) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 4 4 p 2 E) none of the above Kull Spring 07 Lesson 23 Ch 8 14

Practice Problem 23 -6 v The set of quantum numbers that correctly describes an electron in a 3 p orbital is A) n = 3; ℓ = 0; ms = 0 B) n = 3; ℓ = 2; mℓ = -2, -1, 0, 1, or 2; ms = +½ or -½ C) n = 3; ℓ = 1; mℓ = -1, 0, or 1; ms = +½ or -½ D) n = 4; ℓ = 0; mℓ = -1 , 0, or 1; ms = +½ or -½ E) none of the above Kull Spring 07 Lesson 23 Ch 8 15

Practice Problem 23 -6 Answer v The set of quantum numbers that correctly describes an electron in a 3 p orbital is A) n = 3; ℓ = 0; ms = 0 B) n = 3; ℓ = 2; mℓ = -2, -1, 0, 1, or 2; ms = +½ or -½ C) n = 3; ℓ = 1; mℓ = -1, 0, or 1; ms = +½ or ½ D) n = 4; ℓ = 0; mℓ = -1 , 0, or 1; ms = +½ or -½ E) none of the above Kull Spring 07 Lesson 23 Ch 8 16

v An Practice Problem 23 - 7 atom in its ground state contains 18 electrons. How many of these are in orbitals with mℓ = 0? A) 2 B) 4 C) 6 D) 8 E) 10 Kull Spring 07 Lesson 23 Ch 8 17

Practice Problem 23 -7 Answer v An atom in its ground state contains 18 electrons. How many of these are in orbitals with mℓ = 0? A) 2 B) 4 C) 6 D) 8 E) 10 Kull Spring 07 Lesson 23 Ch 8 18

Practice Problem 23 -8 v The configuration for the six outer electrons in ground state oxygen atoms is A) 2 s 3 2 p 3 -1 B) 2 p 6 C) 2 s 2 2 p-12 2 p 02 D) 2 s 2 2 p-12 2 p 01 2 p 11 E) 2 s 4 2 p 2 -1 Kull Spring 07 Lesson 23 Ch 8 19

Practice Problem 23 -8 Answer v The configuration for the six outer electrons in ground state oxygen atoms is A) 2 s 3 2 p 3 -1 B) 2 p 6 C) 2 s 2 2 p-12 2 p 02 D) 2 s 2 2 p-12 2 p 01 2 p 11 E) 2 s 4 2 p 2 -1 Kull Spring 07 Lesson 23 Ch 8 20

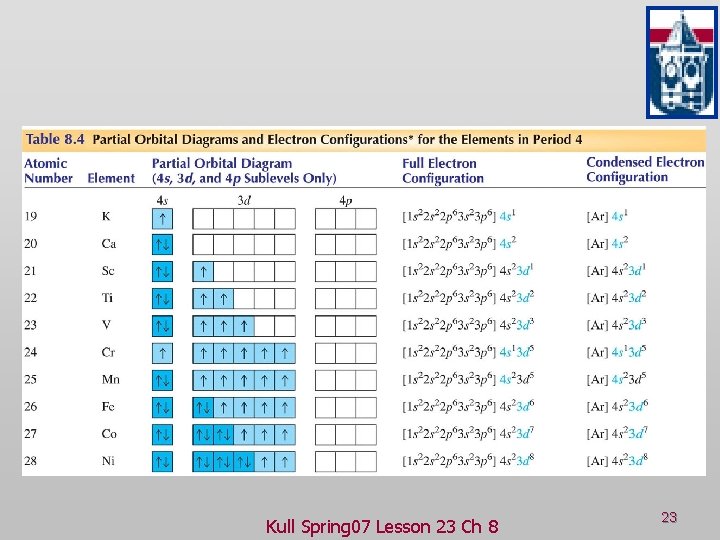
Practice Problem 23 -9 v Which of the following is the electron configuration for chromium, element 24? A) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 B) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 4 C) 1 s 2 2 p 6 3 s 2 3 p 6 3 d 6 D) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 3 d 5 E) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 1 Kull Spring 07 Lesson 23 Ch 8 21

Practice Problem 23 -9 Answer v Which of the following is the electron configuration for chromium, element 24? A) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 B) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 4 C) 1 s 2 2 p 6 3 s 2 3 p 6 3 d 6 D) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 1 3 d 5 E) 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 1 Kull Spring 07 Lesson 23 Ch 8 22

Kull Spring 07 Lesson 23 Ch 8 23

Kull Spring 07 Lesson 23 Ch 8 24

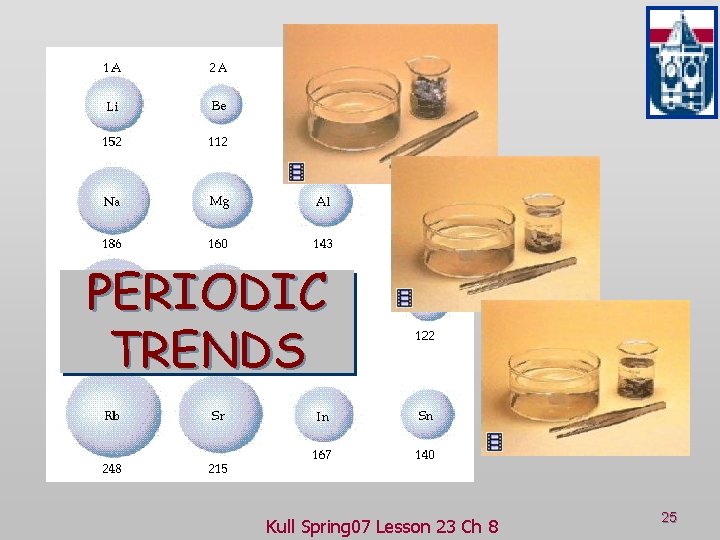
PERIODIC TRENDS Kull Spring 07 Lesson 23 Ch 8 25

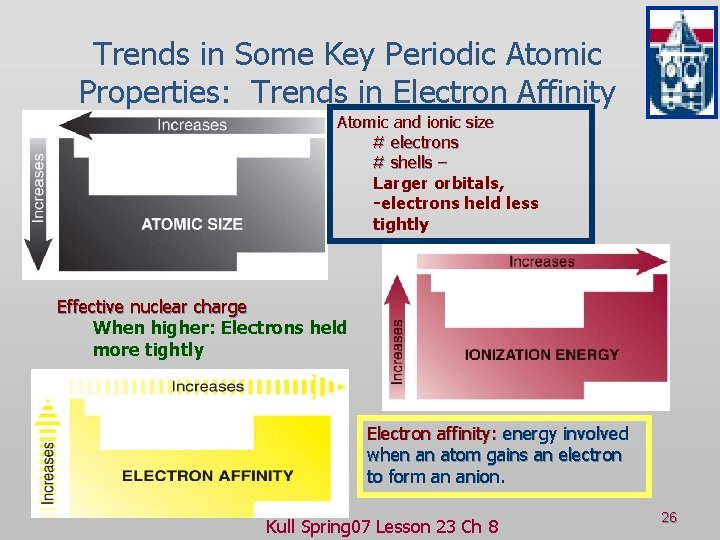
Trends in Some Key Periodic Atomic Properties: Trends in Electron Affinity Atomic and ionic size # electrons # shells – Larger orbitals, -electrons held less tightly Effective nuclear charge When higher: Electrons held more tightly Electron affinity: energy involved when an atom gains an electron to form an anion. Kull Spring 07 Lesson 23 Ch 8 26

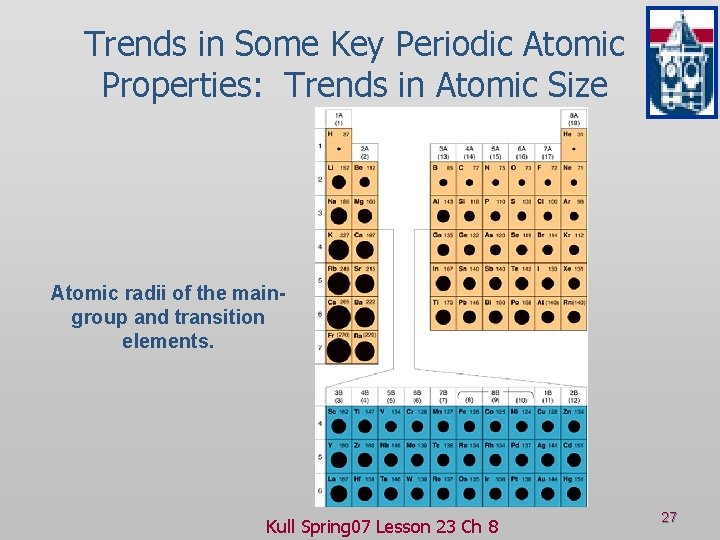
Trends in Some Key Periodic Atomic Properties: Trends in Atomic Size Atomic radii of the maingroup and transition elements. Kull Spring 07 Lesson 23 Ch 8 27

Characteristics of Many-Electron Atoms: Electrostatic Effects and the Splitting of Energy Levels Factors Affecting Atomic Orbital Energies The Effect of Nuclear Charge (Zeffective) Higher nuclear charge lowers orbital energy (stabilizes the system) by increasing nucleus-electron attractions. The Effect of Electron Repulsions (Shielding) Additional electron in the same orbital An additional electron raises the orbital energy through electron-electron repulsions. Additional electrons in inner orbitals Inner electrons shield outer electrons more effectively than do electrons in the same sublevel. Kull Spring 07 Lesson 23 Ch 8 28

Effective Nuclear Charge, Z* v v v v Z* is the nuclear charge experienced by the outermost electrons. Z* increases across a period owing to incomplete shielding by inner electrons. The 2 s electron PENETRATES the region occupied by the 1 s electron. 2 s electron experiences a higher positive charge than expected. Estimate Z* by --> [ Z - (# inner electrons) ] Charge felt by 2 s e- in Li Z* = 3 - 2 = 1 Be Z* = 4 - 2 = 2 B Z* = 5 - 2 = 3 and so on! Kull Spring 07 Lesson 23 Ch 8 29

Ionization Energy IE = energy required to remove an electron from an atom in the gas phase. Mg (g) + 738 k. J ---> Mg+ (g) + e- Mg+ (g) + 1451 k. J ---> Mg 2+ (g) + e- Mg+ has 12 protons and only 11 electrons. Therefore, IE for Mg+ > Mg Kull Spring 07 Lesson 23 Ch 8 30

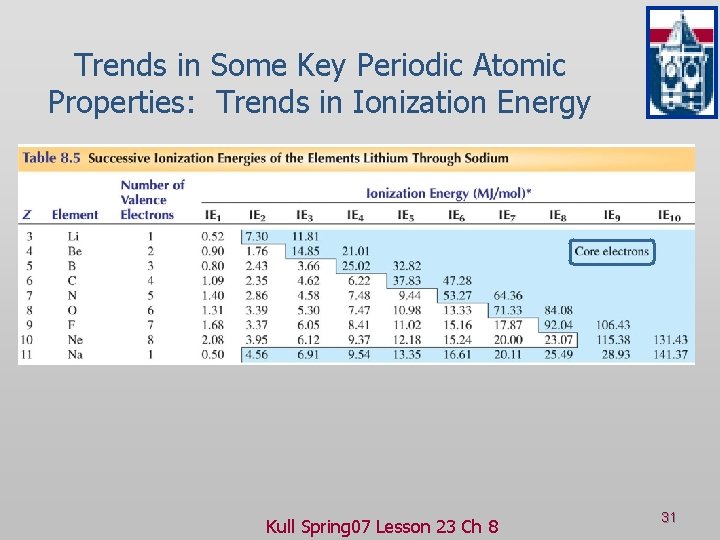
Trends in Some Key Periodic Atomic Properties: Trends in Ionization Energy Kull Spring 07 Lesson 23 Ch 8 31

Ion Sizes + Li, 152 pm 3 e and 3 p Li + , 78 pm 2 e and 3 p Forming a cation. v CATIONS are SMALLER than the atoms from which they come. v The electron/proton attraction has gone UP and so size DECREASES. Kull Spring 07 Lesson 23 Ch 8 32

Ion Sizes F, 71 pm 9 e and 9 p F- , 133 pm 10 e and 9 p Forming an anion. v ANIONS are LARGER than the atoms from which they come. v The electron/proton attraction has gone DOWN and so size INCREASES. v Trends in ion sizes are the same as atom sizes. Kull Spring 07 Lesson 23 Ch 8 33

Ion Configurations To form cations, always remove electrons of highest n value first! P [Ne] 3 s 2 3 p 3 - 3 e- ---> P 3+ [Ne] 3 s 2 3 p 0 Kull Spring 07 Lesson 23 Ch 8 34

Trends in Ion Sizes Active Figure 8. 15 Kull Spring 07 Lesson 23 Ch 8 35

Practice Problem 23 -10 v Which of the following has the largest radius? A) F B) N C) C D) O E) Ne Kull Spring 07 Lesson 23 Ch 8 36

Practice Problem 23 -10 Answer v Which of the following has the largest radius? A) F B) N C) C D) O E) Ne Kull Spring 07 Lesson 23 Ch 8 37

Practice Problem 23 -11 v Which of the following elements has the largest ionization energy? A) Na B) Ne C) F D) K E) Rb Kull Spring 07 Lesson 23 Ch 8 38

Practice Problem 23 -11 Answer v Which of the following elements has the largest ionization energy? A) Na B) Ne C) F D) K E) Rb Kull Spring 07 Lesson 23 Ch 8 39

Practice Problem 23 -12 v Which of the following has the greatest electron affinity (most negative value)? A) Cl B) K C) He D) Na E) Rb Kull Spring 07 Lesson 23 Ch 8 40

Practice Problem 23 -12 Answer v Which of the following has the greatest electron affinity (most negative value)? A) Cl B) K C) He D) Na E) Rb Kull Spring 07 Lesson 23 Ch 8 41

Practice Problem 23 -13 v In the periodic table, the most nonmetallic elements will be found A) at the top of Group 3 A (13) B) at the top of Group 1 A (1) C) at the top of Group 7 A (17) D) at the bottom of Group 1 A (1) E) at the bottom of Group 7 A (17) Kull Spring 07 Lesson 23 Ch 8 42

Practice Problem 23 -13 Answer v In the periodic table, the most nonmetallic elements will be found A) at the top of Group 3 A (13) B) at the top of Group 1 A (1) C) at the top of Group 7 A (17) D) at the bottom of Group 1 A (1) E) at the bottom of Group 7 A (17) Kull Spring 07 Lesson 23 Ch 8 43

Practice Problem 23 -14 v Which ion has the smallest radius? A) Li+ B) Na+ C) K+ D) Be 2+ E) Mg 2+ Kull Spring 07 Lesson 23 Ch 8 44

Practice Problem 23 -14 Answer v Which ion has the smallest radius? A) Li+ B) Na+ C) K+ D) Be 2+ E) Mg 2+ Kull Spring 07 Lesson 23 Ch 8 45

Practice Problem 23 -15 v Which of the following statements about periodic properties is incorrect? A) Both electron affinity and ionization energy decrease down a group. B) Atomic size increases to the right across a period. C) Ionization energy increases to the right across a period. D) Atomic size increases down a group. E) Electron affinity increases to the right across a period. Kull Spring 07 Lesson 23 Ch 8 46

Practice Problem 23. 15 Answer v Which of the following statements about periodic properties is incorrect? A) Both electron affinity and ionization energy decrease down a group. B) Atomic size increases to the right across a period. C) Ionization energy increases to the right across a period. D) Atomic size increases down a group. E) Electron affinity increases to the right across a period. Kull Spring 07 Lesson 23 Ch 8 47

Practice Problem 23. 16 v Which of the following elements has the greatest ionization energy? A) Ga B) As C) K D) Pb E) Na Kull Spring 07 Lesson 23 Ch 8 48

Practice Problem 22. 16 Answer v Which of the following elements has the greatest ionization energy? A) Ga B) As C) K D) Pb E) Na Kull Spring 07 Lesson 23 Ch 8 49

Practice Problem 23. 17 v Which of the following elements has the greatest electron affinity? A) Rb B) Ca C) Li D) Na E) I Kull Spring 07 Lesson 23 Ch 8 50

Practice Problem 23. 17 Answer v Which of the following elements has the greatest electron affinity? A) Rb B) Ca C) Li D) Na E) I Kull Spring 07 Lesson 23 Ch 8 51

Practice Problem 23. 18 v Which of the following ions and atoms has the largest radius? A) Mg B) Na C) Na+ D) Mg 2+ E) Al Kull Spring 07 Lesson 23 Ch 8 52

Practice Problem 23. 18 Answer v Which of the following ions and atoms has the largest radius? A) Mg B) Na C) Na+ D) Mg 2+ E) Al Kull Spring 07 Lesson 23 Ch 8 53

Practice Problem 23. 19 v Which of the following elements has the greatest metallic character? A) Mg B) Al C) Ca D) Ba E) Cs Kull Spring 07 Lesson 23 Ch 8 54

Practice Problem 23. 19 Answer v Which of the following elements has the greatest metallic character? A) Mg B) Al C) Ca D) Ba E) Cs Kull Spring 07 Lesson 23 Ch 8 55

Equations speed of light = wavelength x frequency c = λ X = 3. 00 x 108 m/s v E = nh(c/ ) n= positive integer v Planck’s constant(h) = 6. 626 x 10– 34 J s v Eatom = Eemitted (or absorbed) radiation = nh v Rydberg equation v v =R n 2 > n 1 R = 1. 096776 x 107 m-1 ΔE = Efinal – Einitial = – 2. 18 x 10– 18 J Ephoton = Estate A – Estate B = hν Kull Spring 07 Lesson 23 Ch 8 56

Quantum mechanics Quantum number n– shell/level ℓ - subshell/ sublevel mℓ - orbital ms - spin Values Total number 1, 2, 3, … ∞ 0, 1, … n-1 - ℓ, 0, + ℓ n=# subshells n-1 +½, n 2 = # orbitals in a shell 2 ℓ +1 (orbitals in a subshell) -½ 2 possible Kull Spring 07 Lesson 23 Ch 8 57

Practice Problem 23 -20 v When electrons in helium atoms fall from the 3 s orbital down to the 1 s orbital, how many different energies could be released if all possible pathways of decreasing energy are considered? A) 2 B) 4 C) 5 D) 6 E) 7 Kull Spring 07 Lesson 23 Ch 8 58

Practice Problem 23 -20 Answer v When electrons in helium atoms fall from the 3 s orbital down to the 1 s orbital, how many different energies could be released if all possible pathways of decreasing energy are considered? A) 2 B) 4 C) 5 D) 6 E) 7 Kull Spring 07 Lesson 23 Ch 8 59

Practice Problem 23 -21 v Of the following four electron configurations, which two represent elements that would have similar chemical properties? 1. 1 s 22 p 5 2. 1 s 22 p 6 3. [Ar]4 s 23 d 104 p 4 4. [Ar]4 s 23 d 104 p 5 A) 1 and 3 B) 1 and 4 C) 2 and 3 D) 1 and 2 E) 2 and 4 Kull Spring 07 Lesson 23 Ch 8 60

Practice Problem 23 -21 Answer v Of the following four electron configurations, which two represent elements that would have similar chemical properties? 1. 1 s 22 p 5 2. 1 s 22 p 6 3. [Ar]4 s 23 d 104 p 4 4. [Ar]4 s 23 d 104 p 5 A) 1 and 3 B) 1 and 4 C) 2 and 3 D) 1 and 2 E) 2 and 4 Kull Spring 07 Lesson 23 Ch 8 61

Practice Problem 23 -22 v The electron configuration for Fe 2+ is 1 s 2 2 p 6 3 s 2 3 p 6 3 d 6. Therefore Fe 2+ is A) paramagnetic with two unpaired electrons. B) paramagnetic with one unpaired electron. C) paramagnetic with three unpaired electrons. D) paramagnetic with four unpaired electrons. E) diamagnetic. Kull Spring 07 Lesson 23 Ch 8 62

Practice Problem 23 -22 Answer v The electron configuration for Fe 2+ is 1 s 2 2 p 6 3 s 2 3 p 6 3 d 6. Therefore Fe 2+ is A) paramagnetic with two unpaired electrons. B) paramagnetic with one unpaired electron. C) paramagnetic with three unpaired electrons. D) paramagnetic with four unpaired electrons. E) diamagnetic. Kull Spring 07 Lesson 23 Ch 8 63