to je to upravljanje antimikrobnim lijekovima i kojim

Što je to upravljanje antimikrobnim lijekovima i kojim metodama se može provesti? Prof Bojana Beović UMC Ljubljana Faculty of Medicine, University of Ljubljana Slovenia

• 33 110 (28 480– 38 430) attributable deaths (25, 100 in 2007 = + 32%!!!) • 170 (150– 192) DALYs* per 100 000 population *DALY, disability-adjusted life-years Lancet ID 2018. Published Online November 5, 2018 http: //dx. doi. org/10. 1016/S 1473 -3099(18)30605 -4 ECDC, EMEA doc. ref. EMEA/576176/2009, Stockholm, September 2009
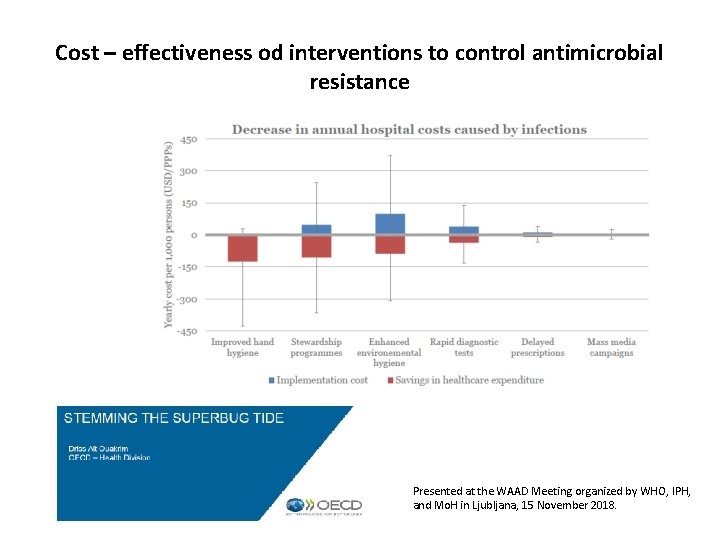
Cost – effectiveness od interventions to control antimicrobial resistance Presented at the WAAD Meeting organized by WHO, IPH, and Mo. H in Ljubljana, 15 November 2018.

IDSA/SHEA: 1997 Shlaes D, et al. Clin Infect Dis. 1997; 25: 584– 99

Definitions of AMS: 2012– 2018 • ESGAP: 1 A set of actions or a programme aimed at ensuring responsible antibiotic use. • IDSA/SHEA: 2 Coordinated interventions designed to improve and measure the appropriate use of antimicrobials by promoting the selection of the optimal antimicrobial drug regimen, dose, duration of therapy, and route of administration. • WHO: 3 Interventions designed to optimise use of antibiotics. 1. Dyar OJ, et al. Clin Microbiol Infect. 2017; 23(11): 793– 98; 2. Fischer N, et al. Infect Control Hosp Epidemiol. 2012; 33(4): 322– 27; 3. Van Dijck C, et al. Bull World Health Organ. 2018; 96: 266– 80

Why do we need AMS? A few simple facts: • Antibiotics cause antimicrobial resistance 1, 2 • Antibiotic resistance decreases efficacy of antibiotics 1 • There are very few new antibiotics that are effective against resistant bacteria 1, 2 • Antibiotics can prescribed by virtually all practising physicians, regardless of speciality 1 • Education on antibiotic resistance 1, 2 and prescribing 2 is deficient 1. Global Action Plan on Antimicrobial Resistance. World Health Organization 2015 http: //www. who. int/antimicrobial-resistance/publications/global-action-plan/en/ [Last accessed September 2018]. , 2. Pulcini C, Gyssens IC. Virulence; 2013: 4(2): 192– 202


Key factors contributing to antimicrobial misuse • Diagnostic uncertainty – Is there a bacterial infection in this wound? • Clinician ignorance – When to treat with antibiotics • Clinician fear – Of failing to treat properly, or of having a bad outcome • Patient demands – For unnecessary antibiotic therapy Lipsky B, et al. J Antimicrob Chemother. 2016; 71(11): 3026– 35.

AMS Interventions in Hospitals Are They Effective?

AMS Studies Can we learn anything from the studies: • • • Differences in antimicrobial resistance Differences in healthcare systems Different group of patients Other interventions that target the same goal (IC) Different sociocultural environment ….

The Efficacy of AMS Interventions in Hospitals The Review includes studies from 1947 to 2015 • • • Better compliance to antibiotic prescribing policy (high certainty evidence) The duration of antibiotic treatment decreased by 1. 95 days (high certainty evidence) Reduced length of stay by 1. 12 days (moderate certainty evidence) reduced Clostridium difficile infections (low certainty evidence) It was not possible to assess the microbial outcome of changed antimicrobial policy Risks Mortality remained unchanged (moderate certainty evidence) Delayed treatment and negative professional culture (low certainty evidence) Davey P, et al. Cochrane Database Syst Rev 2017, Issue 2. Art. No. : CD 003543.

The Efficacy of AMS Interventions by Type • Enablement and restriction increase the compliance with AMS policies. • Enablement increases the effect of restriction. • Enablement with feed-back is probably more effective than enablement alone. Davey P, et al. Cochrane Database Syst Rev 2017, Issue 2. Art. No. : CD 003543.

AMS Interventions Which Intervention?

Do the AS Objectives Have an Impact on AS Goals? (Which Objectives Really Matter? ) ü empirical therapy according to guidelines ü de-escalation of therapy ü switching from intravenous to oral treatment ü therapeutic drug monitoring ü use of a list of restricted antibiotics ü bedside consultations Clinical outcomes Adverse events Costs Resistance rates Current evidence on hospital AMS objectives

Strong recommendations (all moderate level of evidence) Preauthorisation/prospective audit and feeback Reduced use of antibiotics associated with Clostridium difficile infections Increased use of oral antibiotics Shortest effective duration of antimicrobial therapy PK optimization of aminoglycoside therapy Barlam TF, et al. Clin Infect Dis 2016; 62(10): e 51–e 77

Preprescriptional Restrictions or Prospective Audit and Feed-back

Preprescription Authorisation: the prescriber seeks input from an expert (AMS team) before prescribing an antibiotic. Advantages • Reduces initiation of unnecessary/ inappropriate antibiotics • Optimizes empiric choices and influences downstream use • Prompts review of clinical data/prior cultures at the time of initiation of therapy Disadvantages • Impacts use of restricted agents only • Addresses empiric use to a much greater degree than downstream use • Loss of prescriber autonomy • May delay therapy • Decreases antibiotic costs, including those due to high-cost agents • Effectiveness depends on skill of approver • Provides mechanism for rapid response to antibiotic shortages • Real-time resource intensive • Potential for manipulation of system (eg, presenting request in a biased manner to gain approval) • May simply shift to other antibiotic agents and select for different antibiotic-resistance patterns • Direct control over antibiotic use Tamma PD, et al. Clin Infect Dis 2017; 64(5): 537– 43.

Prospective audit (postprescriptional review) and feedback: prescriber decides on antimicrobial therapy himself, the AMS team intervenes afer 48 to 72 hours: adjustment, discontinuation… Advantages • Can increase visibility of antimicrobial stewardship program and build collegial relationships • More clinical data available for recommendations, enhancing uptake by prescribers • Greater flexibility in timing of recommendations • Can be done on less than daily basis if resources are limited • Provides educational benefit to clinicians • Prescriber autonomy maintained • Can address de-escalation of antibiotics and duration of therapy Disadvantages • Compliance voluntary • Typically labor-intensive • Success depends on delivery method of feedback to prescribers • Prescribers may be reluctant to change therapy if patient is doing well • Identification of interventions may require information technology support and/or purchase of computerized surveillance systems • May take longer to achieve reductions in targeted antibiotic use Tamma PD, et al. Clin Infect Dis 2017; 64(5): 537– 43.
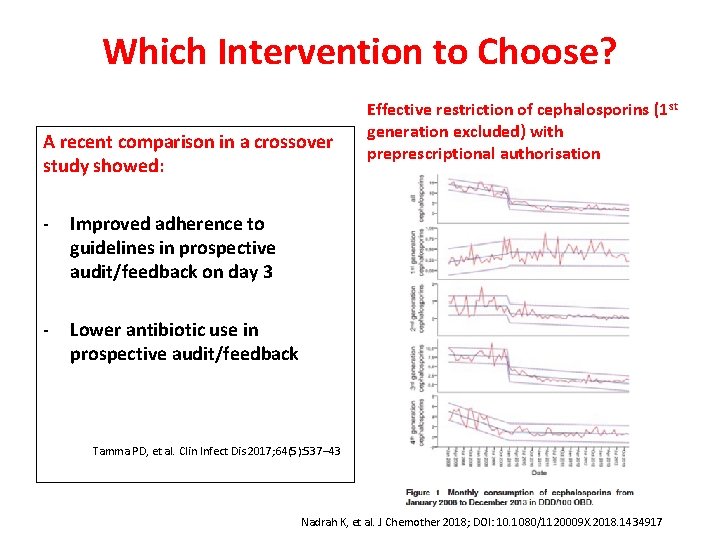
Which Intervention to Choose? A recent comparison in a crossover study showed: - Improved adherence to guidelines in prospective audit/feedback on day 3 - Lower antibiotic use in prospective audit/feedback Effective restriction of cephalosporins (1 st generation excluded) with preprescriptional authorisation Tamma PD, et al. Clin Infect Dis 2017; 64(5): 537– 43 Nadrah K, et al. J Chemother 2018; DOI: 10. 1080/1120009 X. 2018. 1434917

De-escalation
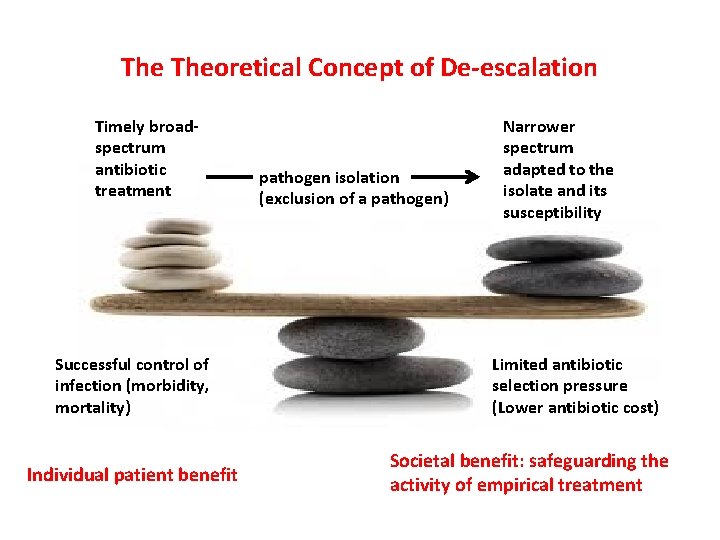
The Theoretical Concept of De-escalation Timely broadspectrum antibiotic treatment Successful control of infection (morbidity, mortality) Individual patient benefit pathogen isolation (exclusion of a pathogen) Narrower spectrum adapted to the isolate and its susceptibility Limited antibiotic selection pressure (Lower antibiotic cost) Societal benefit: safeguarding the activity of empirical treatment

De-escalation: current state of evidence • Very few data and unconvincing effect on antimicrobial resistance. • No effect on Clostridium difficile infections. • Lower mortality in observational studies and controversial results of randomized controlled trials. Paul M, et al. Clin Microbiol Infect 2016; 22: 960 e 967. , Ohji G, et al. Int J Infect Dis 2016; 49: 71– 9. , Guo Y, et al. Heart Lung 2016; 45: 454 e 459. Gutiérrez-Pizarraya A, et al. Expert Rev Clin Pharmacol 2017. DOI: 10. 1080/17512433. 2017. 1293520. , Tabah A, et al. Clin Infect Dis 2016; 62: 1009– 17. , Snyder M, et al. OFID 2017. DOI: 10. 1093/ofid/ofx 226. , Viasus D, et al. J Antimicrob Chemother 2017; 72: 547– 53. , Leon M, et al. Intensive Care Med 2014; 40: 1399– 408. , Bohan JG, et al. OFID 2016. doi: 10. 1093/ofid/ofw 244. , Rattanaumpawan P, et al. BMC Infect Dis 2017; 17: 183. , Falguera M, et al. Thorax 2010; 65: 101 e 106. , Xiao B, et al. Exper Ther Med 2017; 13: 1485 -89. , Kim JW, et al. Critical Care 2012, 16: R 28.

Controversies! Practical Tips for Safe (and Effective) De-escalation • Consider de-escalation when there is high impact of empirical treatment and high need to preserve its efficacy. • Consider de-escalation when second antibiotic therapy with lower resistance selection potential with respect to the setting is available. • Make proper clinical diagnosis of infection. • Assure relevant microbiological diagnostics. • Use rapid diagnostics to accelerate the de-escalation and shorten the exposure to first antibiotic. • Second antibiotic therapy should be given in a dose that attains the PK/PD target and therapeutic concentration in the tissue. • If possible: discontinuation of an antibiotic is more effective than de-escalation. Giantsou E, et al. Intensive Care Med 2007; 33: 1533– 40. , Tabah A, et al. Clin Infect Dis 2016; 62: 1009– 17. , Paul M, et al. Clin Microbiol Infect 2016; 22: 960 e 967, Xiao B, et al. Exper Ther Med 2017; 13: 1485 -89, Carlier M, et al. Antimicrob Agents Chemother 59: 4689 – 94

Duration of Antimicrobial Treatment
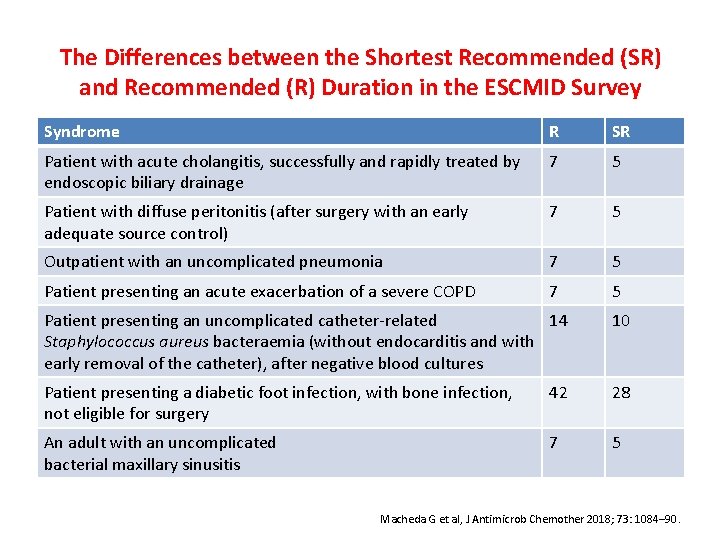
The Differences between the Shortest Recommended (SR) and Recommended (R) Duration in the ESCMID Survey Syndrome R SR Patient with acute cholangitis, successfully and rapidly treated by endoscopic biliary drainage 7 5 Patient with diffuse peritonitis (after surgery with an early adequate source control) 7 5 Outpatient with an uncomplicated pneumonia 7 5 Patient presenting an acute exacerbation of a severe COPD 7 5 Patient presenting an uncomplicated catheter-related 14 Staphylococcus aureus bacteraemia (without endocarditis and with early removal of the catheter), after negative blood cultures 10 Patient presenting a diabetic foot infection, with bone infection, not eligible for surgery 42 28 An adult with an uncomplicated bacterial maxillary sinusitis 7 5 Macheda G et al, J Antimicrob Chemother 2018; 73: 1084– 90.

The Team
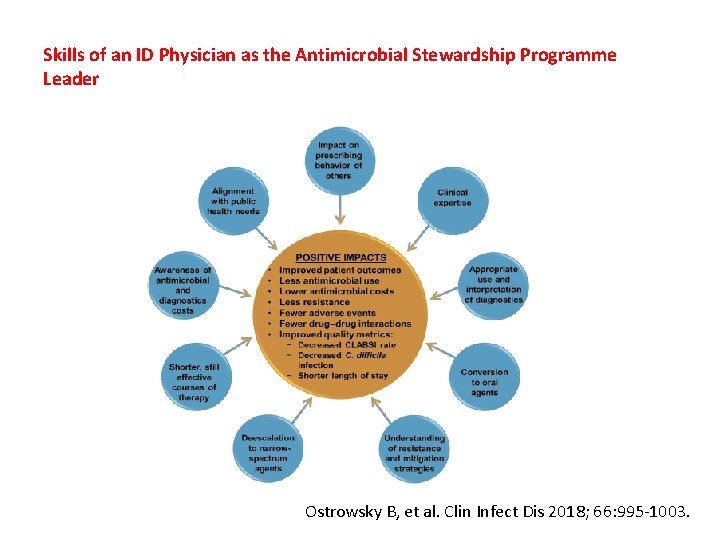
Skills of an ID Physician as the Antimicrobial Stewardship Programme Leader Ostrowsky B, et al. Clin Infect Dis 2018; 66: 995 -1003.

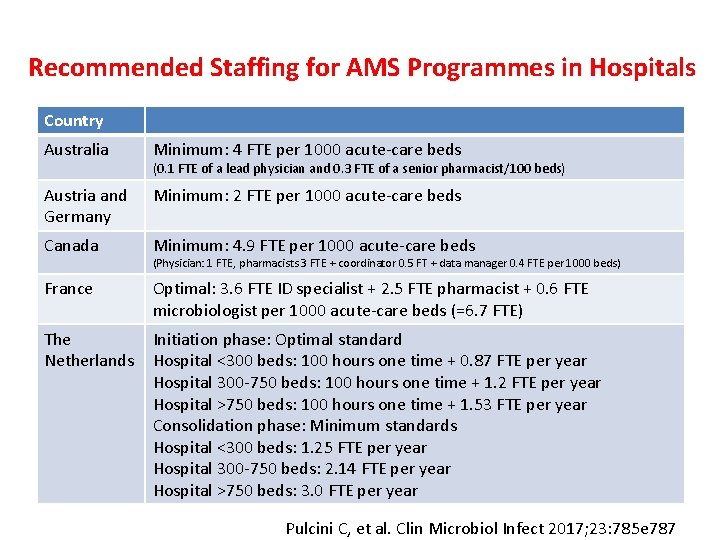
Recommended Staffing for AMS Programmes in Hospitals Country Australia Minimum: 4 FTE per 1000 acute-care beds Austria and Germany Minimum: 2 FTE per 1000 acute-care beds Canada Minimum: 4. 9 FTE per 1000 acute-care beds France Optimal: 3. 6 FTE ID specialist + 2. 5 FTE pharmacist + 0. 6 FTE microbiologist per 1000 acute-care beds (=6. 7 FTE) The Netherlands Initiation phase: Optimal standard Hospital <300 beds: 100 hours one time + 0. 87 FTE per year Hospital 300 -750 beds: 100 hours one time + 1. 2 FTE per year Hospital >750 beds: 100 hours one time + 1. 53 FTE per year Consolidation phase: Minimum standards Hospital <300 beds: 1. 25 FTE per year Hospital 300 -750 beds: 2. 14 FTE per year Hospital >750 beds: 3. 0 FTE per year (0. 1 FTE of a lead physician and 0. 3 FTE of a senior pharmacist/100 beds) (Physician: 1 FTE, pharmacists 3 FTE + coordinator 0. 5 FT + data manager 0. 4 FTE per 1000 beds) Pulcini C, et al. Clin Microbiol Infect 2017; 23: 785 e 787

Education

IDSA & SHEA Guidelines for Implementation of Antimicrobial Stewardship in Hospitals 2007 2016 Education is essential! (strong recommendation, low level of evidence) Dellit TH, et al. Clin Infect Dis 2007; 44: 159– 77. , Barlam TF, et al. Clin Infect Dis 2016; 62(10): e 51–e 77

What Type of Education in AMS is Most Successful? • Systematic review of 28 RTCs • Antibiotic prescription decreased by 9 to 52% • Innapropriate prescriptions decreased by 41% on average • Small group education seems to most effective (52%) followed guidelines and leaflets (42%) Lee CR, et al. Biomed Res Int 2015; Article ID 214021

Competencies in antimicrobial prescribing and AMS Competency is the ability to do something successfully or efficiently. It is a combination of knowledge, skills, motives and personal traits. ESCMID Generic Competencies in Antimicrobial Prescribing and AMS What every independent antibiotic prescriber should know and practice.

Barriers and Facilitators of AMS • Scientific (e. g. , uncertainty of future medical needs) • Economic (e. g. , lack of financial incentives) • Regulatory (e. g. , regulatory harmonisation) • Ethical (e. g. , responsibility for future generations) • Societal (e. g. , ‘invisibility’ of antibacterial resistance) • Political (e. g. , changing political environment) • Professional practice challenges (e. g. , alternatives to antibiotics) Monnier A, et al. Poster P 1713, presented at ECCMID 2018.

Sociocultural aspect Regulatory aspect

Sociocultural dimensions of antibiotic prescribing • Differences in antibiotic use between EU countries can be explained with sociocultural dimensions 1 • Differences in outpatient antibiotic use can be explained by power distance, uncertainty avoidance, hierarchy, masculinity and religion 2 • Differences in antibiotic use for cold, flu and sore throat identified by Eurobarometer are concordant with uncertainty avoidance and masculinity in the society 3 • Frequency of prolonging antibiotic surgical prophylaxis beyond 24 hours is concordant with uncertainty avoidance 4 1. Hulscher M, et al. Lancet Infect Dis. 2010; 10(3): 167– 75, 2. Deschepper R, et al. BMC Health Serv Res. 2008; 8: 123; 3. Borg M. J Antimicrob Chemother. 2012; 67(3): 763– 7; 4. Borg M. J Antimicrob Chemother. 2014; 69(4): 1142– 4.

Legal framework for AMS in Europe (An ESCMID/ESGAP survey) 25 countries and two Italian regions Required component Laws regulating AMS Other regulations on the ministerial level National strategies and/or action plans Funding of AMS Mandatory AMS structures and activities 0 10 20 30 40 50 Proportion of countries with framework in place, % 60 No regulations/strategy/action plan adopted by ministry of health in 5 countries! AMS, antimicrobial stewardship Beović B, et al. Int J Antimicrob Agents. 2018. [Epub ahead of print].

Conclusion • AMS interventions increase the adherence to guidelines in improve the quality of antimicrobial prescribing. • The multidisciplinary team, preferably led by infectious diseases specialist, should have the authority and should be compensated for the time dedicated to AMS. • Carefully chosen interventions should take into account sociocultural characteristics of the setting.
- Slides: 39



























































