THERMODYNAMICS Thermodynamics is the study of energy relationships


















- Slides: 39

THERMODYNAMICS Thermodynamics is the study of energy relationships that involve heat, mechanical work, and other aspects of energy and heat transfer. Central Heating

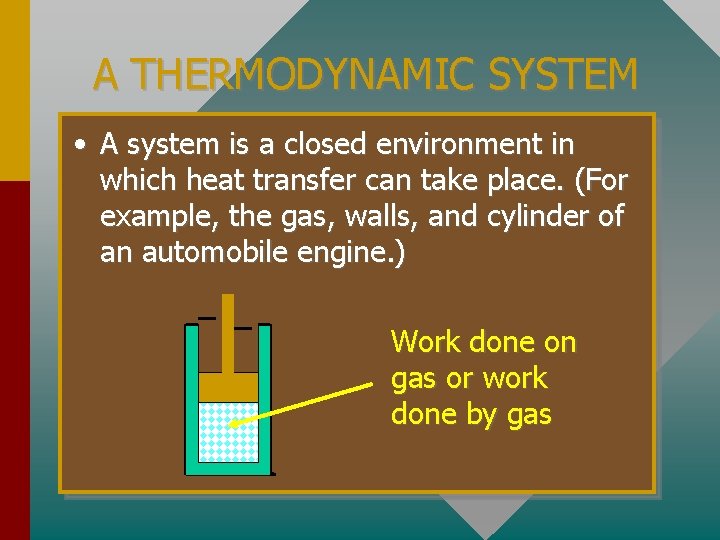
A THERMODYNAMIC SYSTEM • A system is a closed environment in which heat transfer can take place. (For example, the gas, walls, and cylinder of an automobile engine. ) Work done on gas or work done by gas

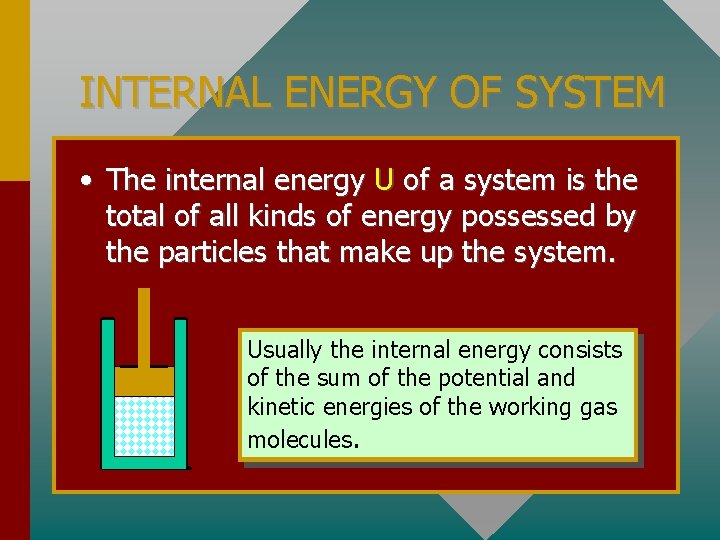
INTERNAL ENERGY OF SYSTEM • The internal energy U of a system is the total of all kinds of energy possessed by the particles that make up the system. Usually the internal energy consists of the sum of the potential and kinetic energies of the working gas molecules.

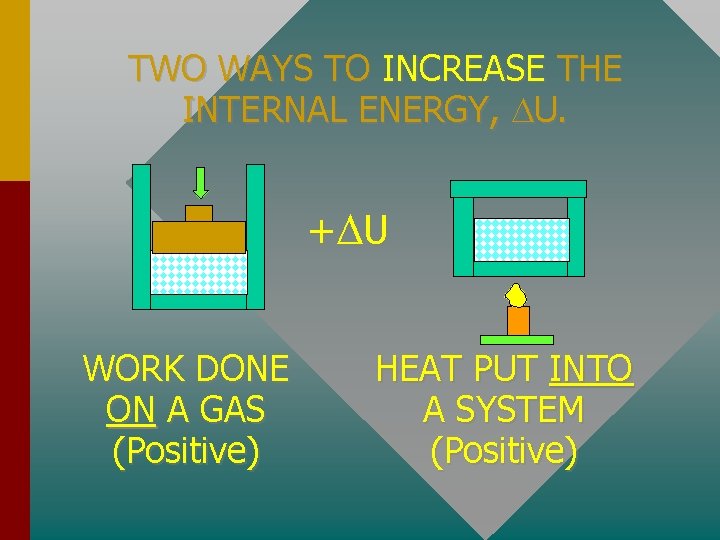
TWO WAYS TO INCREASE THE INTERNAL ENERGY, U. + U WORK DONE ON A GAS (Positive) HEAT PUT INTO A SYSTEM (Positive)

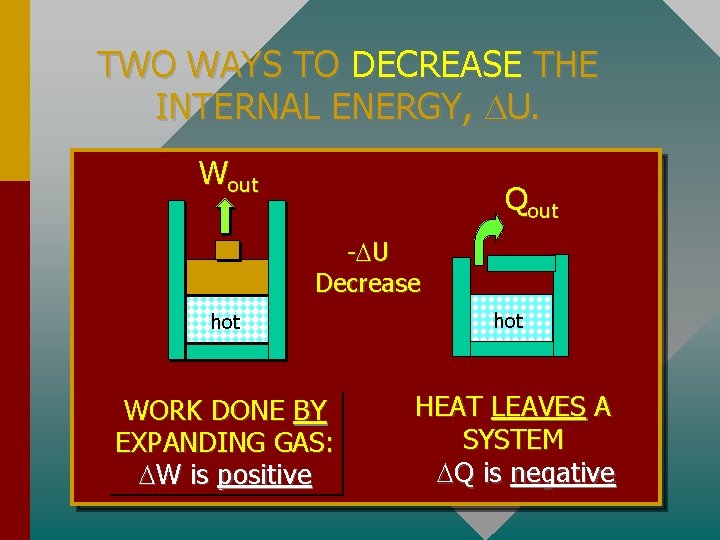
TWO WAYS TO DECREASE THE INTERNAL ENERGY, U. Wout Qout - U Decrease hot WORK DONE BY EXPANDING GAS: W is positive hot HEAT LEAVES A SYSTEM Q is negative

THERMODYNAMIC STATE The STATE of a thermodynamic system is determined by four factors: • Absolute Pressure P in Pascals • Temperature T in Kelvins • Volume V in cubic meters • Number of moles, n, of working gas

THE FIRST LAW OF THERMODYAMICS: • The net heat put into a system is equal to the change in internal energy of the system plus the work done BY the system. Q = U + W final - initial) • Conversely, the work done ON a system is equal to the change in internal energy plus the heat lost in the process.

APPLICATION OF FIRST LAW OF THERMODYNAMICS Example 1: In the figure, the Wout =120 J gas absorbs 400 J of heat and at the same time does 120 J of work on the piston. What is the change in internal energy of the system? Qin Apply First Law: Q = U + W 400 J

Example 1 (Cont. ): Apply First Law Q is positive: +400 J (Heat IN) Wout =120 J W is positive: +120 J (Work OUT) Q = U + W U = Q - W = (+400 J) - (+120 J) = +280 J Qin 400 J U = +280 J

Example 1 (Cont. ): Apply First Law Energy is conserved: The 400 J of input thermal energy is used to perform 120 J of external work, increasing the internal energy of the system by 280 J The increase in internal energy is: Wout =120 J Qin 400 J U = +280 J

FOUR THERMODYNAMIC PROCESSES: • Isochoric Process: V = 0, W = 0 • Isobaric Process: P = 0 • Isothermal Process: T = 0, U = 0 • Adiabatic Process: Q = 0 Q = U + W

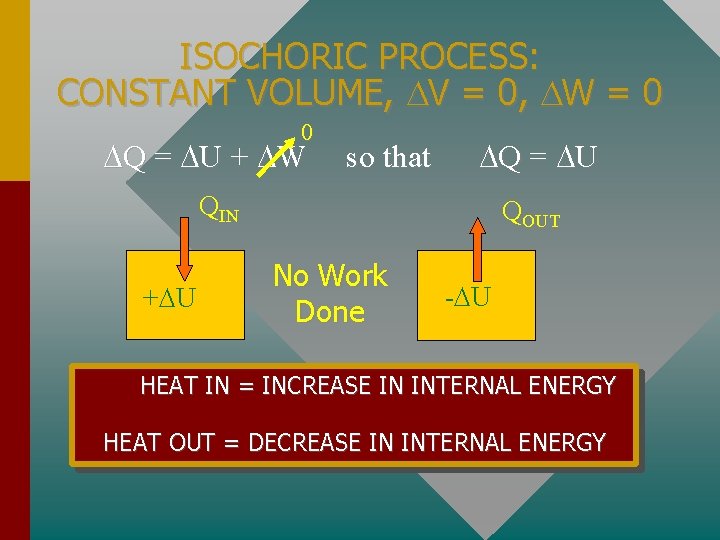
ISOCHORIC PROCESS: CONSTANT VOLUME, V = 0, W = 0 0 Q = U + W so that Q = U QIN + U QOUT No Work Done - U HEAT IN = INCREASE IN INTERNAL ENERGY HEAT OUT = DECREASE IN INTERNAL ENERGY

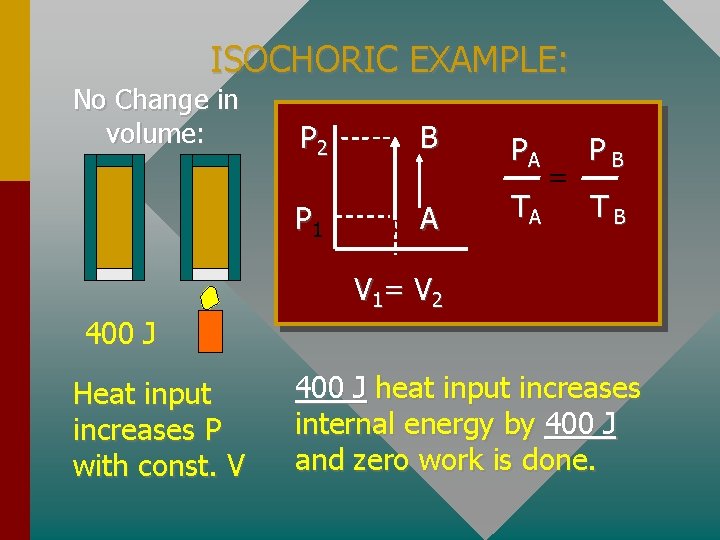
ISOCHORIC EXAMPLE: No Change in volume: P 2 B P 1 A PA TA = PB TB V 1= V 2 400 J Heat input increases P with const. V 400 J heat input increases internal energy by 400 J and zero work is done.

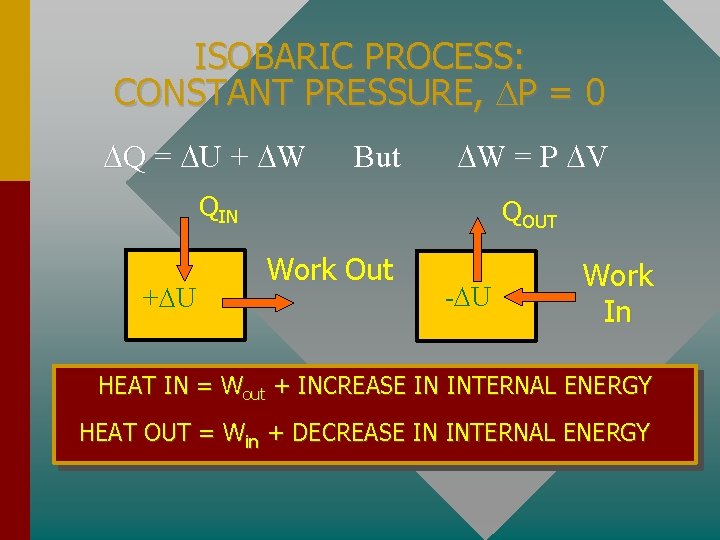
ISOBARIC PROCESS: CONSTANT PRESSURE, P = 0 Q = U + W But W = P V QIN + U QOUT Work Out - U Work In HEAT IN = Wout + INCREASE IN INTERNAL ENERGY HEAT OUT = Win + DECREASE IN INTERNAL ENERGY

ISOBARIC EXAMPLE (Constant Pressure): ( P A B VA TA 400 J Heat input increases V with const. P V 1 = VB TB V 2 400 J heat does 120 J of work, increasing the internal energy by 280 J. J

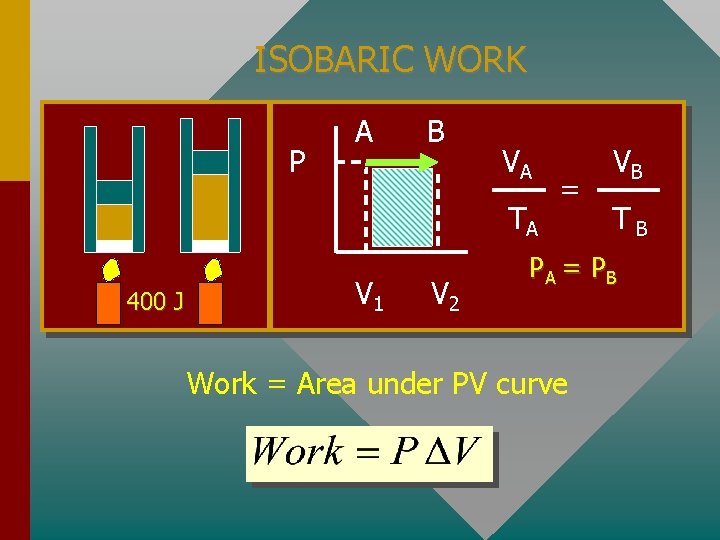
ISOBARIC WORK P A B VA TA 400 J V 1 V 2 = VB TB PA = P B Work = Area under PV curve

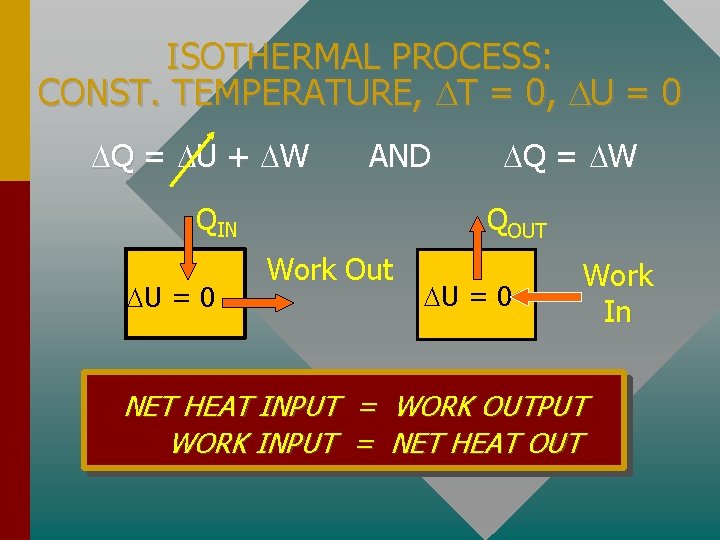
ISOTHERMAL PROCESS: CONST. TEMPERATURE, T = 0, U = 0 Q = U + W AND Q = W QIN U = 0 QOUT Work Out U = 0 Work In NET HEAT INPUT = WORK OUTPUT WORK INPUT = NET HEAT OUT

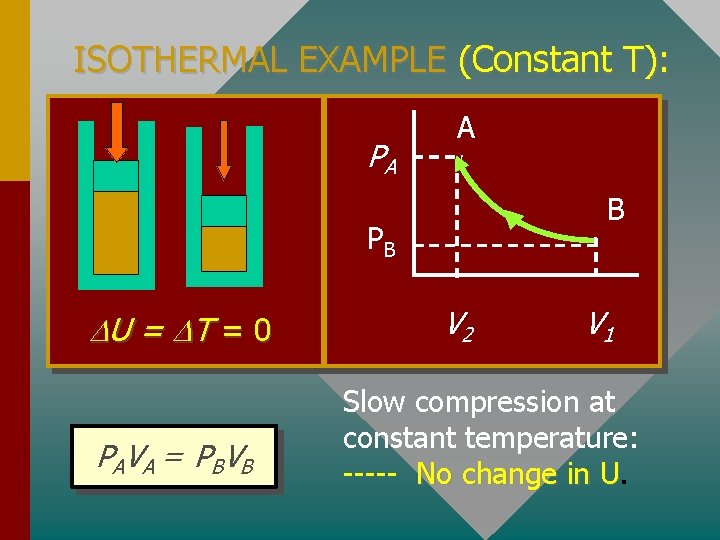
ISOTHERMAL EXAMPLE (Constant T): PA A B PB U = T = 0 P A V A = PBV B V 2 V 1 Slow compression at constant temperature: ----- No change in U. U

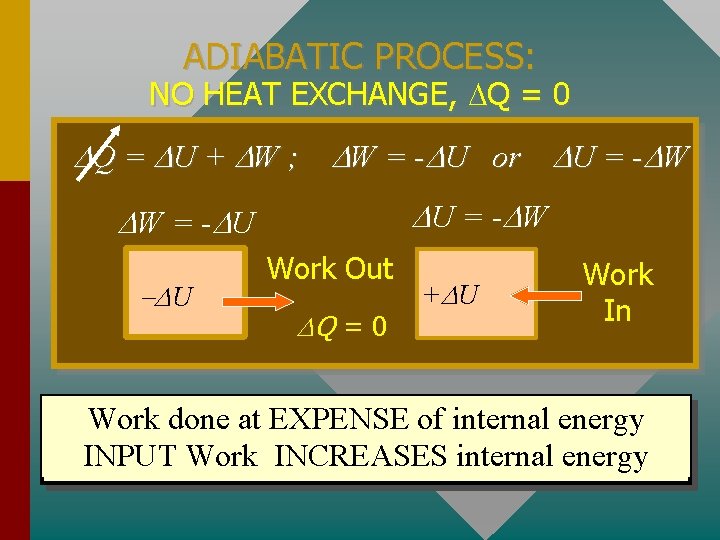
ADIABATIC PROCESS: NO HEAT EXCHANGE, Q = 0 Q = U + W ; W = - U or U = - W W = - U U Work Out Q = 0 + U Work In Work done at EXPENSE of internal energy INPUT Work INCREASES internal energy

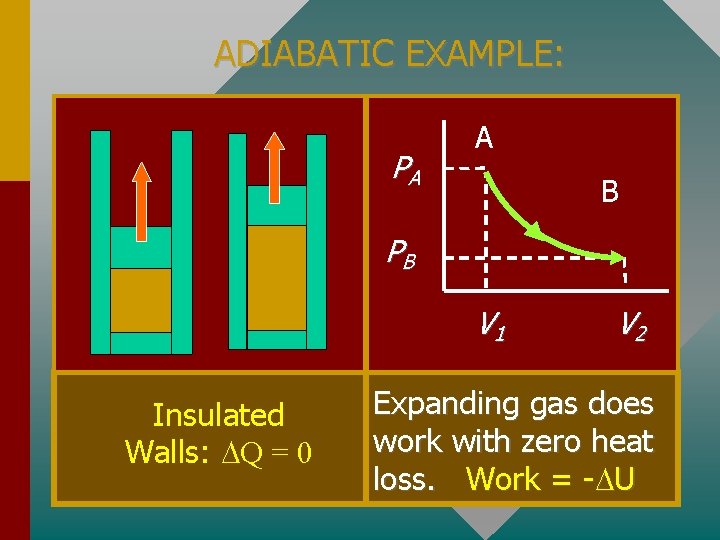
ADIABATIC EXAMPLE: PA A B PB V 1 Insulated Walls: Q = 0 V 2 Expanding gas does work with zero heat loss. Work = - U

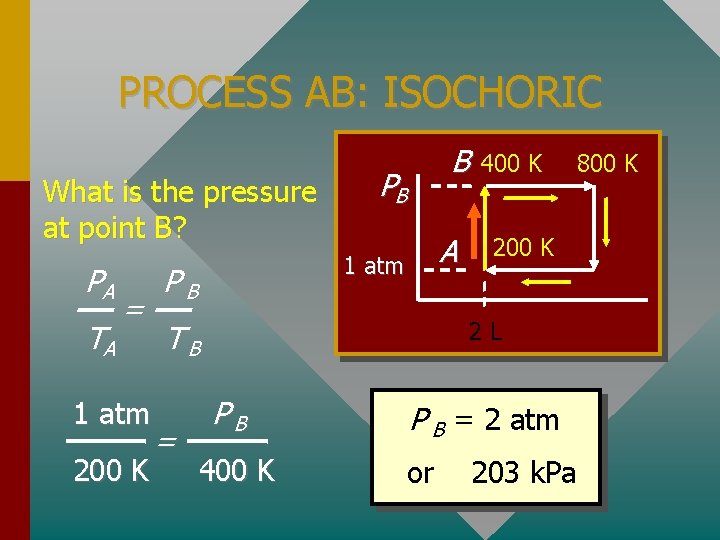
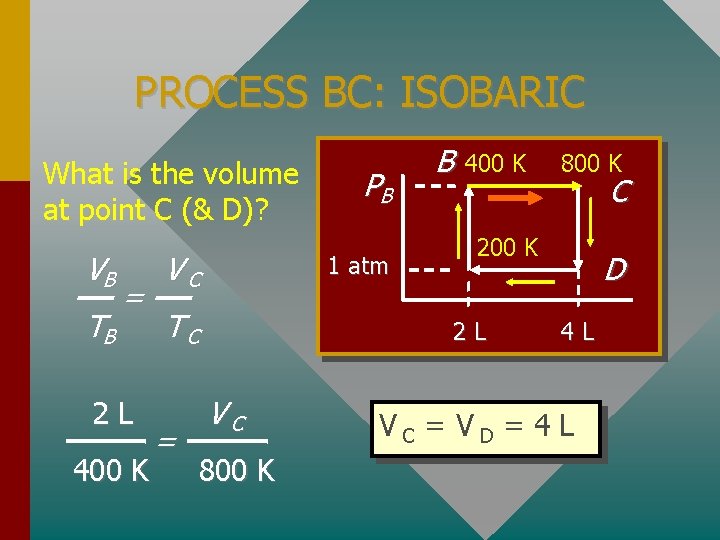
Example Problem: A 2 -L sample of Oxygen gas has an initial temperature and pressure of 200 K and 1 atm. The gas undergoes four processes: • AB: Heated at constant V to 400 K. • BC: Heated at constant P to 800 K. • CD: Cooled at constant V back to 1 atm. • DA: Cooled at constant P back to 200 K.

PV-DIAGRAM FOR PROBLEM How many moles of O 2 are present? Consider point A: PV = n. RT PB 1 atm B A 400 K 2 L 800 K

PROCESS AB: ISOCHORIC What is the pressure at point B? PA TA = 1 atm 200 K B PB A 1 atm PB 200 K 2 L TB = 400 K PB 400 K P B = 2 atm or 203 k. Pa 800 K

PROCESS BC: ISOBARIC What is the volume at point C (& D)? VB TB = 2 L 400 K VC 1 atm TC = PB B 400 K 800 K C 200 K 2 L VC 800 K D 4 L VC = VD = 4 L

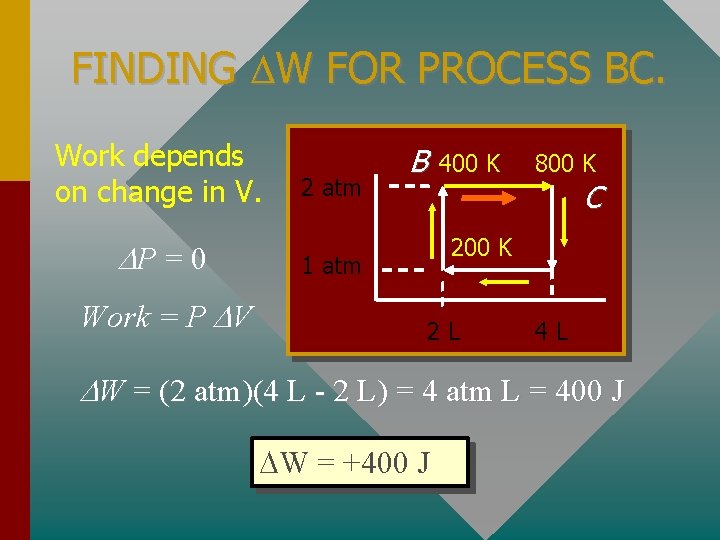
FINDING W FOR PROCESS BC. Work depends on change in V. P = 0 Work = P V 2 atm B 400 K 800 K C 200 K 1 atm 2 L 4 L W = (2 atm)(4 L - 2 L) = 4 atm L = 400 J W = +400 J

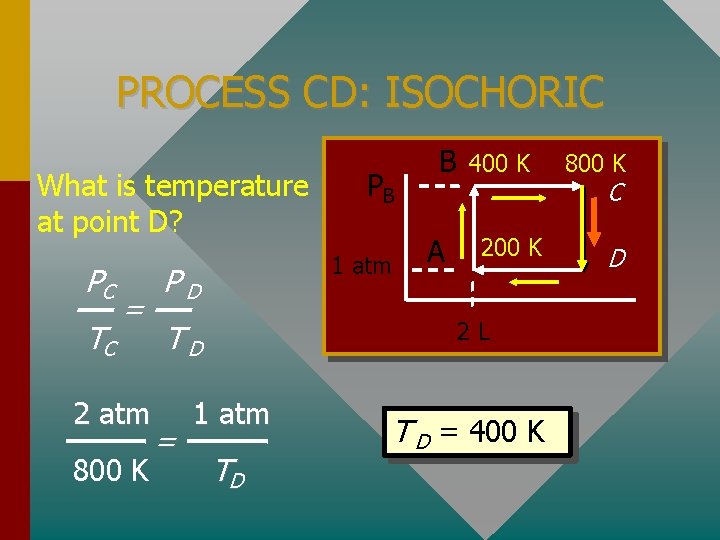
PROCESS CD: ISOCHORIC What is temperature at point D? PC TC = 2 atm 800 K 1 atm PD A 400 K 2 L TD = PB B 1 atm TD T D = 400 K 800 K C D

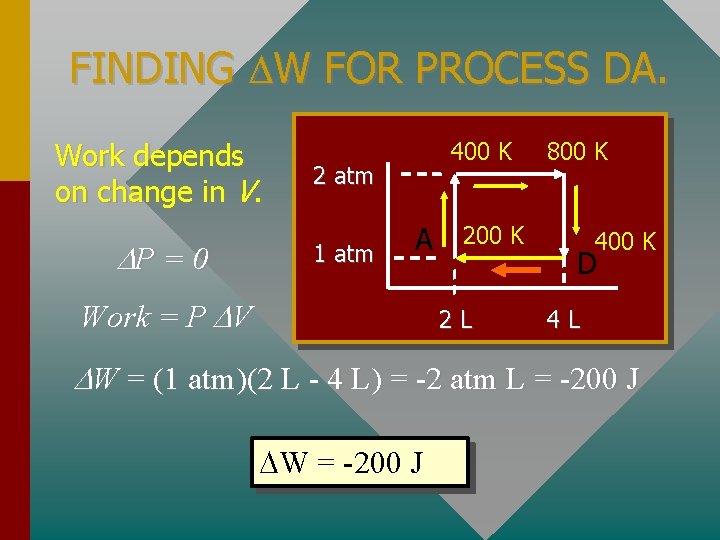
FINDING W FOR PROCESS DA. Work depends on change in V. P = 0 400 K 2 atm 1 atm A Work = P V 200 K 2 L 800 K 400 K D 4 L W = (1 atm)(2 L - 4 L) = -2 atm L = -200 J W = -200 J

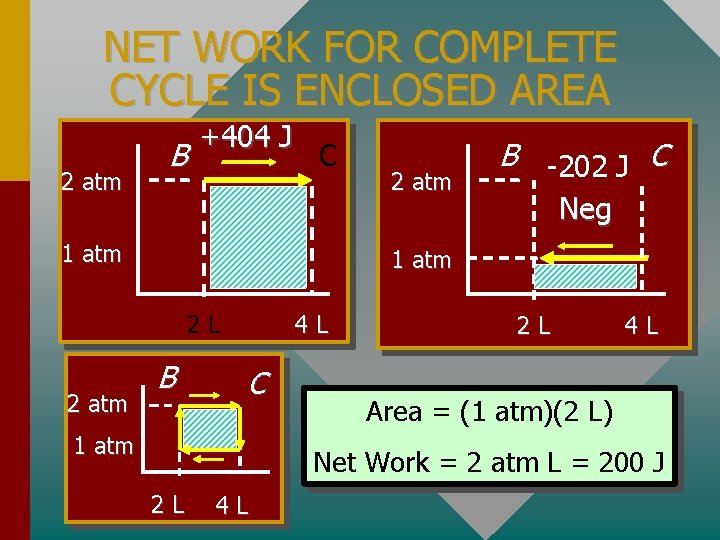
NET WORK FOR COMPLETE CYCLE IS ENCLOSED AREA 2 atm B +404 J C 1 atm Neg 1 atm 2 L 2 atm B -202 J C B 4 L C 1 atm 2 L 4 L Area = (1 atm)(2 L) Net Work = 2 atm L = 200 J 2 L 4 L

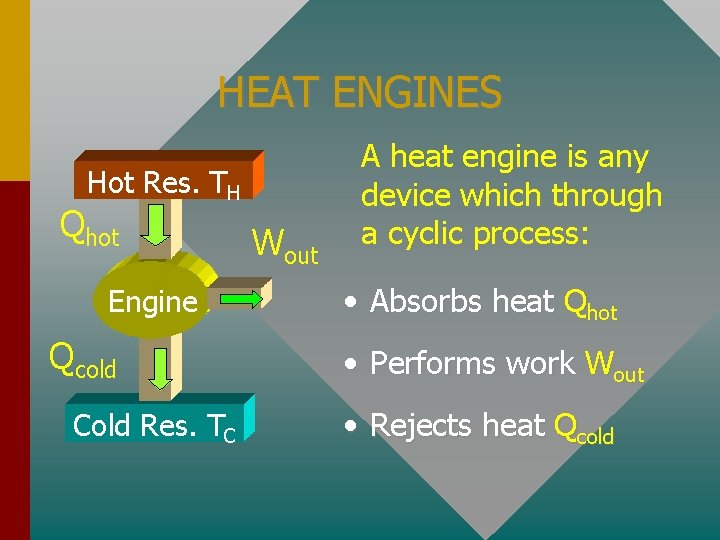
HEAT ENGINES Hot Res. TH Qhot Engine Qcold Cold Res. TC Wout A heat engine is any device which through a cyclic process: • Absorbs heat Qhot • Performs work Wout • Rejects heat Qcold

THE SECOND LAW OF THERMODYNAMICS Hot Res. TH Qhot Engine Wout Qcold Cold Res. TC It is impossible to construct an engine that, operating in a cycle, produces no effect other than the extraction of heat from a reservoir and the performance of an equivalent amount of work.

THE SECOND LAW OF THERMODYNAMICS Hot Res. TH 400 J 100 J Engine Hot Res. TH 400 J Engine 400 J 300 J Cold Res. TC • A possible engine. Cold Res. TC • An IMPOSSIBLE engine.

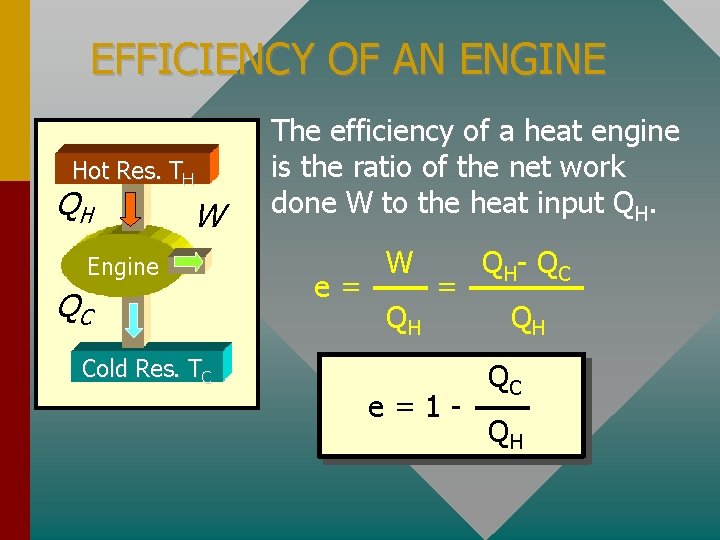
EFFICIENCY OF AN ENGINE Hot Res. TH QH W Engine QC The efficiency of a heat engine is the ratio of the net work done W to the heat input QH. e= W QH = Cold Res. TC e=1 - Q H- Q C QH QC QH

EFFICIENCY EXAMPLE Hot Res. TH 800 J Engine 600 J Cold Res. TC W An engine absorbs 800 J and wastes 600 J every cycle. What is the efficiency? QC e=1 QH e=1 - 600 J 800 J e = 25% Question: How many joules of work is done?

EFFICIENCY OF AN IDEAL ENGINE (Carnot Engine) Hot Res. TH QH Engine QC W For a perfect engine, the quantities Q of heat gained and lost are proportional to the absolute temperatures T. e= T H- T C Cold Res. TC e=1 - TH TC TH

Example 3: A steam engine absorbs 600 J of heat at 500 K and the exhaust temperature is 300 K. If the actual efficiency is only half of the ideal efficiency, how much work is done during each cycle? e=1 e=1 - TC Actual e = 0. 5 ei = 20% TH W 300 K 500 K e = 40% e= QH W = e. QH = 0. 20 (600 J) Work = 120 J

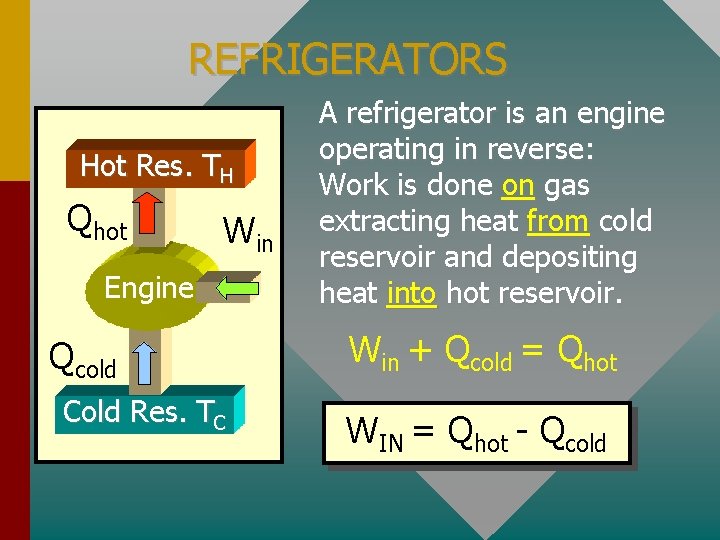
REFRIGERATORS Hot Res. TH Qhot Win Engine Qcold Cold Res. TC A refrigerator is an engine operating in reverse: Work is done on gas extracting heat from cold reservoir and depositing heat into hot reservoir. Win + Qcold = Qhot WIN = Qhot - Qcold

THE SECOND LAW FOR REFRIGERATORS Hot Res. TH Qhot Engine Qcold Cold Res. TC It is impossible to construct a refrigerator that absorbs heat from a cold reservoir and deposits equal heat to a hot reservoir with W = 0. If this were possible, we could establish perpetual motion!

Summary The First Law of Thermodynamics: The net heat taken in by a system is equal to the sum of the change in internal energy and the work done by the system. Q = U + W final - initial) • Isochoric Process: V = 0, W = 0 • Isobaric Process: P = 0 • Isothermal Process: T = 0, U = 0 • Adiabatic Process: Q = 0

Summary (Cont. ) Hot Res. TH Qhot Wout Engine Qcold Cold Res. TC The Second Law of Thermo: It is impossible to construct an engine that, operating in a cycle, produces no effect other than the extraction of heat from a reservoir and the performance of an equivalent amount of work.