Thermodynamics Thermodynamics Vocabulary Thermodynamics The study of the

Thermodynamics

Thermodynamics Vocabulary Thermodynamics The study of the physical transfer of heat. Thermal contact and physical contact are not the same thing. Thermal contact can occur without physical contact. Warming your hands near a fire place. Thermal Contact When two objects are in close enough proximity to transfer heat to one another. Thermal Equilibrium When two objects in thermal contact have the same temperature.

Zeroth Law of Thermodynamics Objects are in thermal equilibrium when they are the same temperature. When objects are in thermal contact the object with the higher temperature transfers heat to the object with the lower temperature.

Temperature A measure of the average kinetic energy of the molecules in a substance. In open air water cannot reach temperatures above boiling because the fastest moving water molecules leave through evaporation. Absolute zero occurs when there is no energy in a substance. The molecules are stationary or there are no molecules present. Vacuum is the absence of matter.

Temperature scales Fahrenheit Freezing 32 o. F Boiling 212 o. F Fahrenheit to Celsius conversion o. C = 5/9(o. F ) + 32 Kelvin Absolute zero Celsius to Kelvin conversion K = o. C + 273 Centigrade (Celsius) Freezing 0 o. C Boiling 100 o. C Celsius to Fahrenheit conversion o. F = 9/5(o. C - 32)

Thermal Expansion Because thermal energy causes molecular motion, substances have a tendency to expand when they are heated. Mercury or alcohol expand inside a glass tube and reflect a rise in temperature. Substances expand in three dimensions: Linear expansion Area expansion Volume expansion

Linear Expansion When substances expand in length. ΔL = coefficient of linear expansion x initial length x ΔT ΔL is a change in length Coefficient of linear expansion depends on the material ΔT is the change in temperature Roads, railroads and pipelines have expansion joints to allow for expansion and prevent cracks and deformation. Bimetallic strips- used to turn heaters on and off. Made of two different types of metal with different coefficients of linear expansion. Uneven expansion causes the strip to bend in one direction or another and is either attached to a dial or may open and close a circuit.

Water a Special Case Water is the only substance on Earth that occurs naturally in all three physical states. Water unlike any other substance is actually less dense as a solid. Due to steric hindrance between the bonds of water molecules the crystalline structure has spaces in it. This means ice floats. When water is heated from 0 o to 4 o it actually shrinks and is densest at 4 o. C.


Biological Implications Because cold dense water sinks to the bottom of a lake in the winter the less dense water freezes at the top to insulate the living organisms at the bottom. When water freezes it releases heat to the lower layers to further warm the water below. Because cold dense water sinks, upwelling occurs to deliver nutrients created from decaying organisms in the sediments below.

Heat and Mechanical Work Count Rumford(1753 -1814) an American born physicist, spy and social reformer supervised the boring of cannon barrels with a drill bit and noticed that the drill heated the metal. Because energy is always conserved, he deduced that some of the mechanical energy from the drill bit was converted thermal energy in the metal.

James Prescott Joule(1818 -1889) Joule observed that when a mass attached to a paddle wheel submerged in water was dropped it produced mechanical work on the paddle wheel which raised the temperature of the water. He was able to calculate the amount of mechanical work converted to heat. Heat is usually measured in Calories. 1 Kilocalorie = the amount of energy necessary to raise the temperature of 1 kg of water 1 o C. 1 Calorie = 1 Kilocalorie 1 calorie = 4. 186 Joules 1 Btu (British unit for heat) = amount of energy to raise 1 pound of water 1 o. F 1 Btu = 0. 252 Kilocalorie= 1055 Joules Q = heat = the amount of energy transferred due to a difference in temperature.

Heat Capacity Heat required to change the temperature of a given substance. C = Q/ΔT SI unit J/K or J/o. C Lead has heat capacity of 128 J/K Heat capacity is always positive Q (heat transferred) is positive if ΔT is positive. That is if heat is added to the system. Q (heat transferred) is negative if ΔT is negative. That is if heat is removed from the system.


Specific Heat The amount of energy required to raise 1 kg of substance 1 OC. C = Q/mΔT SI unit J/kg K or J/kg o. C Substances with a high specific heat require a lot of energy to raise the temperature. Biological significance of a high specific heat. Water has a high specific heat and is therefore biologically significant. Climate moderation. Because the body is primarily composed of water living organisms have the ability to resist large changes in internal temperature.

Calorimetry Calorimeter is a device used to measure the amount of heat generated by a substance when it is burned. It is a closed container that allows the heat to be transferred from the substance to water. From this increase in temperature the number of calories contained within the substance can be calculated. Heat lost by the substance = heat gained by the water. Remember Q =m cΔT m cΔTlost = mc ΔTgained
calorimeter

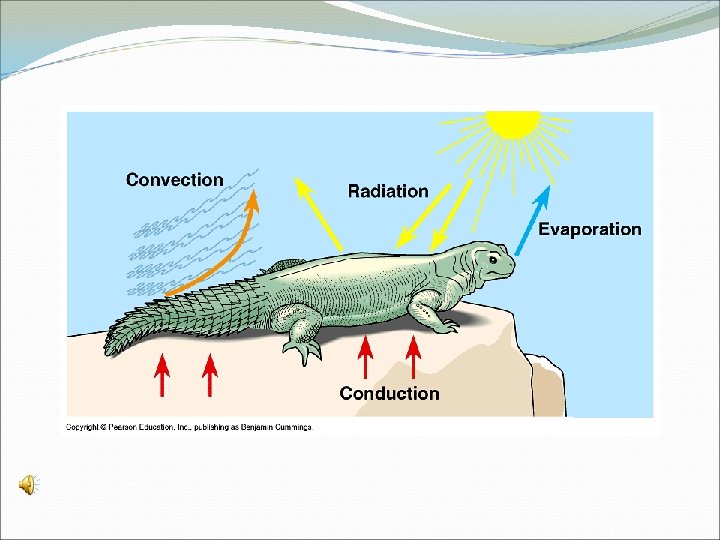
Types of Heat Transfer Conduction Transfer of heat due to the motion of atoms. Requires physical contact A hot plate heats the glass molecules of the beaker. Convection Transfer of heat through the movement of currents within a fluid. Expansion of the fluid causes it to be less dense and rise while cooler denser fluid sinks creating a convective cell. Water above the bottom of the beaker is heated through conduction and causes the water to expand rise, cooler water from above sinks to replace the rising water. Radiation Transfer of heat through a vacuum. Requires no physical contact or matter to present. Place your hands over the top of the beaker filled with hot water and you can feel the heat.


Latent Heat Energy transferred as heat during a phase change. Q = m. L m = mass of the substance latent heat L= Q = Heat transferred Heat of fusion: energy absorbed to go from a solid to a liquid or heat released when a liquid turns into a solid. Heat of Vaporization: Energy absorbed when a substance turns from a liquid to a vapor or energy release when a vapor turns into a liquid.
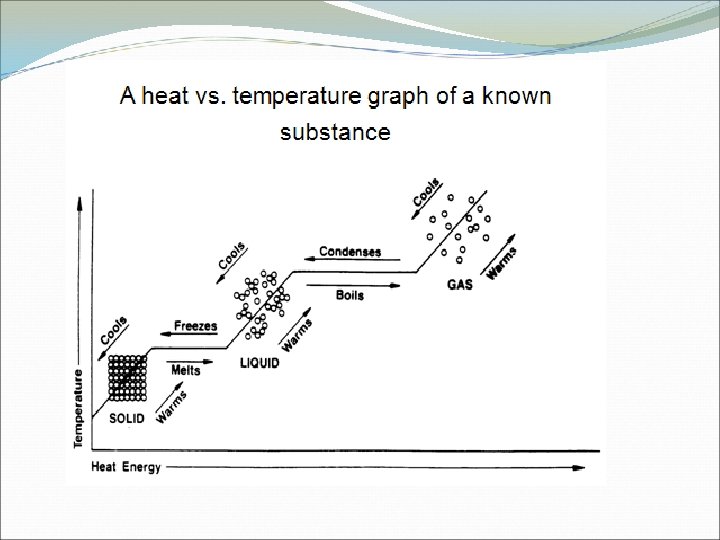

http: //netcamp. prn. bc. ca/nuggets/heatingcurve. swf
- Slides: 23