Topic 2 Thermodynamics Thermodynamics and Energy Thermodynamics The





















- Slides: 22

Topic 2: Thermodynamics

Thermodynamics and Energy • Thermodynamics – The science of heat and work • work = force x distance – Force: any push or pull – Distance: distance over which force is exerted • Heat: energy transferred from one object to another based upon a temperature difference • Energy: capacity to do work or transfer heat – Energy used to cause an object that has mass to move is called work. – Energy used to cause the temperature of an object to rise is called heat.

Types of Energy • Kinetic Energy – Energy of an object by virtue of its motion 1 KE = mv 2 2 • m= kg • v = m/s • E = J = kgm 2/s 2

Types of Energy • Kinetic Energy – the motion of atoms, molecules, or ions at the submicroscopic level (thermal energy). All matter has thermal energy. – The motion of macroscopic objects like a moving tennis ball or automobile (mechanical energy). – The movement of electrons through a conductor (electrical energy) – The compression and expansion of the spaces between molecules in the transmission of sound (acoustic energy)

Types of Energy • Potential Energy – Energy of position – Stored energy based upon position of one object with respect to another

Types of Energy • Potential Energy – Energy possessed by a ball held above the floor and by water at the top of a waterfall (gravitational energy) – Energy stored in fuels (chemical energy). All chemical reactions involve a change in chemical energy. – The energy associated with the separation of two electrical charges (electrostatic energy)

Question: • What’s the kinetic energy of a 7. 5 kg bowling ball traveling at 1. 5 m/s? 1 E = (7. 5 kg)(1. 5 m/s)2 2 E = 8. 44 J

Energy Units • • • 2 m kg / 1 J=1 s 2 1000 J = 1 kilojoule (k. J) 1 calorie = 4. 184 J 1000 calories = 1 kilocalorie (kcal) 1 Calorie (dietary) = 1 kcal

Conservation of Energy • Law of Conservation of Energy – Energy can be neither created or destroyed – Total energy of the universe is constant

Temperature and Heat • Temperature is a measure of an object’s ability to transfer energy as heat. – Temperature determines the direction of thermal energy transfer – The higher the temperature of a given object, the greater thermal energy (energy associated with molecular motion) of its atoms, ions or molecules – Heating and cooling are processes by which energy is transferred as heat from an object at a higher temperature to one at a lower temperature. Heat is not a substance.

System vs. Surroundings • System: portion of space singled out for study – Or you can think of it as the object(s) being studied • Surroundings: everything else in the universe

Thermal Equilibrium • Thermal equilibrium – When two objects reach the same temperature • Distinguishing feature is that on the macroscale, no further temperature change occurs

Example: • Placing a hot metal bar into a beaker of water – Lets us observe 3 important principles 1. Energy transfer as heat will occur spontaneously from an object at a higher temperature to an object at a lower temperature 2. Transfer of energy as heat continues until both objects are at the same temperature (thermal equilibrium) 3. After thermal equilibrium is attained, the object whose temperature increased has gained thermal energy, and the object whose temperature decreased has lost thermal energy

Directionality • We have terms to describe the direction of thermal energy transfer: – Exothermic process • Energy is transferred as heat from a system to its surroundings • Energy of the system decreases as the energy of the surroundings increases – Endothermic process • Energy is transferred as heat to a system from its surroundings • Energy of the system increases as the energy of the surroundings decreases

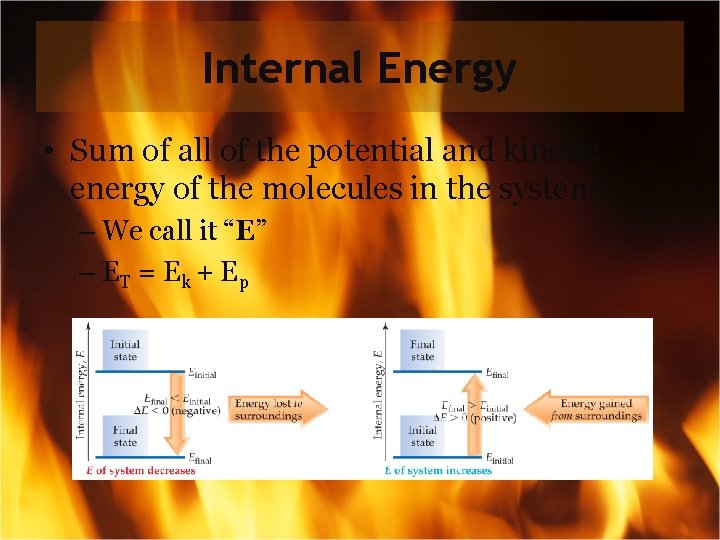
Internal Energy • Sum of all of the potential and kinetic energy of the molecules in the system – We call it “E” – ET = E k + E p

Internal Energy By definition, the change in internal energy, E, is the final energy of the system minus the initial energy of the system: E = Efinal − Einitial … This lead us into the First Law of Thermodynamics…

First Law of Thermodynamics • Energy is conserved – It is neither created nor destroyed. • In other words, the total energy of the universe is a constant; if the system loses energy, it must be gained by the surroundings, and vice versa

Changes in Internal Energy • If E > 0, Efinal > Einitial – Therefore, the system absorbed energy from the surroundings. – This energy change is called endergonic

Changes in Internal Energy • If E < 0, Efinal < Einitial – Therefore, the system released energy to the surroundings. – This energy change is called exergonic.

Changes in Internal Energy • When energy is exchanged between the system and the surroundings, it is exchanged as either heat (q) or work (w). • That is, E = q + w.

E, q, w and Their Signs – E (+) = energy moves from surroundings to system – E (-) = energy moves from system to surroundings – q(+) = when heat is absorbed (energy absorbed by the system) – q(-) = when heat is released (energy released by the system) – w(+) = work done on the system by the surroundings – w(-) = work done on the surroundings by the system

Question • Calculate the total energy change for a system that gives off 200 k. J of heat and does 175 k. J of work on the surroundings? E = q + w E = (-200 k. J) + (-175 k. J) E = -375 k. J